29: Hidrocele Congênita e Hérnia Inguinal
Este capítulo levará aproximadamente 11 minutos para ler.
Introdução
O capítulo a seguir procurará descrever, de forma completa, porém concisa e resumida, a história, embriologia e desenvolvimento, diagnóstico e tratamento da hérnia inguinal congênita e da hidrocele.
Histórico
A cronologia descrita abaixo destaca marcos históricos e avanços ao longo do tempo relacionados à hérnia inguinal congênita e à hidrocele (Figura 1)
No ano 25 AC, Celsus fez a primeira referência à correção de hérnia em crianças.
Os avanços na medicina, assim como a assistência pós-operatória ao paciente, melhoraram as técnicas cirúrgicas e os desfechos cirúrgicos ao longo do tempo. Hoje em dia, o procedimento cirúrgico é realizado de forma semelhante em todo o mundo, com variações mínimas, dependendo das preferências do cirurgião e da abordagem escolhida (laparoscópica vs convencional) para o reparo.
Figura 1 Linha do tempo
Embriologia
Hidrocele e hérnia inguinal congênita surgem como resultado de falha no processo de obliteração do processo vaginal (PV), uma evaginação peritoneal derivada do epitélio celômico, que normalmente se fecha durante o primeiro ano de vida.
A hidrocele pode ser definida como um acúmulo de líquido entre as camadas parietal e visceral da túnica vaginal (TV), que se forma quando o PV permanece pérvio. Podem ser classificadas em comunicante, não comunicante e do cordão. Se o lúmen for suficientemente amplo, o conteúdo abdominal pode protruir para dentro dele e originar uma hérnia inguinal.1
No estágio final da vida fetal, os testículos e os ovários ocuparão uma posição diferente da que tinham no embrião. A mudança de posição dos testículos é muito mais acentuada do que a dos ovários e inclui sua trajetória através da espessura da parede abdominal até o escroto.2
Os eventos que regulam o desenvolvimento testicular e a subsequente descida dos testículos nos indivíduos do sexo masculino são:
3ª semana
As células germinativas primordiais (gâmetas extra-embrionários), que estão na parede do saco vitelino, migram em direção às cristas gonadais (Figura 2)
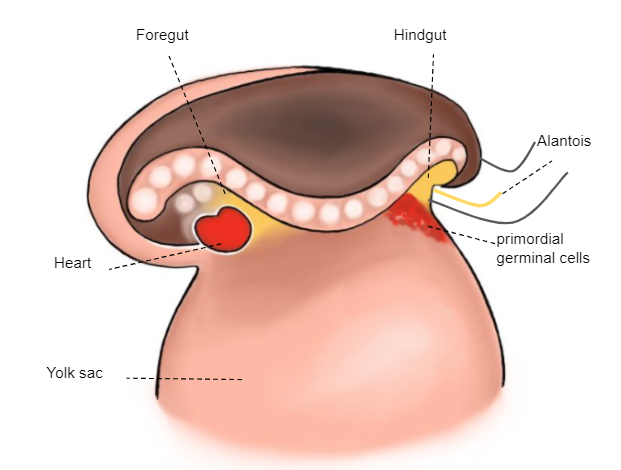
Figura 2 Embrião de 3ª semana
6ª Semana
A gônada indiferenciada está fixada à parede posterior pelo mesoperitônio que, cranialmente, forma o ligamento diafragmático e, caudalmente, o ligamento inguinal (gubernáculo) (Figura 3)
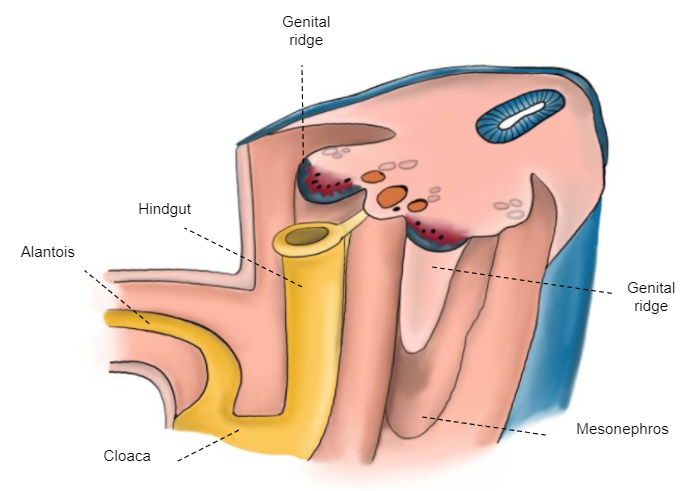
Figura 3 Embrião de 6 semanas
7ª–8ª semana
Os cordões sexuais penetram na medula, originando os cordões testiculares, que alcançam o mesórquio e formam a rete testis (rede de Haller). Posteriormente, estes são transformados em cordões eferentes. (Figura 4)
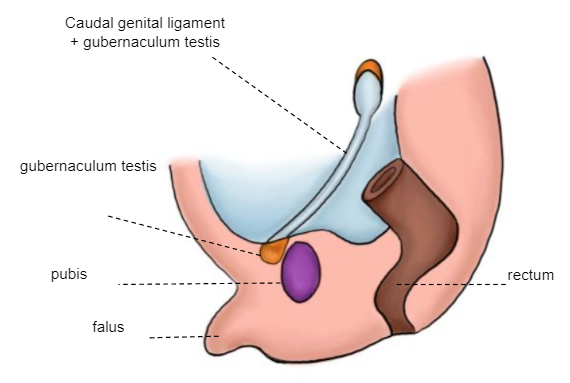
Figura 4 Embrião de 7.ª–8.ª semana
10ª–12ª Semana
O gubernáculo testicular está fixado distalmente à crista lábio-escrotal, que servirá como eixo em torno do qual o mesoderma é modelado, dando origem ao canal inguinal. Forma-se uma evaginação peritoneal, o processus vaginalis (epitélio celômico), seguindo a trajetória do gubernáculo em direção à crista lábio-escrotal. Ocorre a primeira fase da descida testicular, na qual o testículo passa de sua posição lombar para a proximidade do orifício inguinal externo (Figura 5)
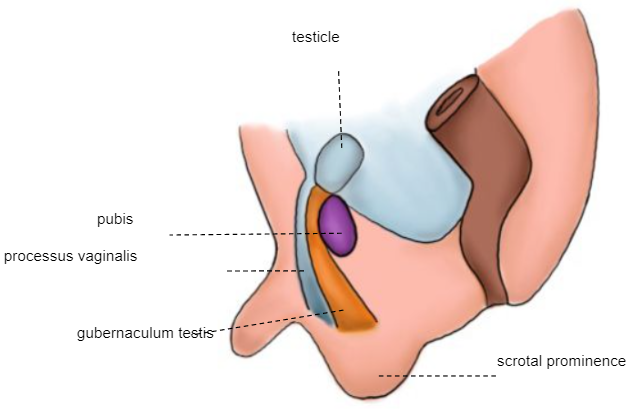
Figura 5 Embrião de 10–12 semanas
28ª semana
Segunda fase da descida: o testículo atravessa o canal inguinal já formado, alcançando o escroto entre as semanas 32 e 35 (Figura 6)
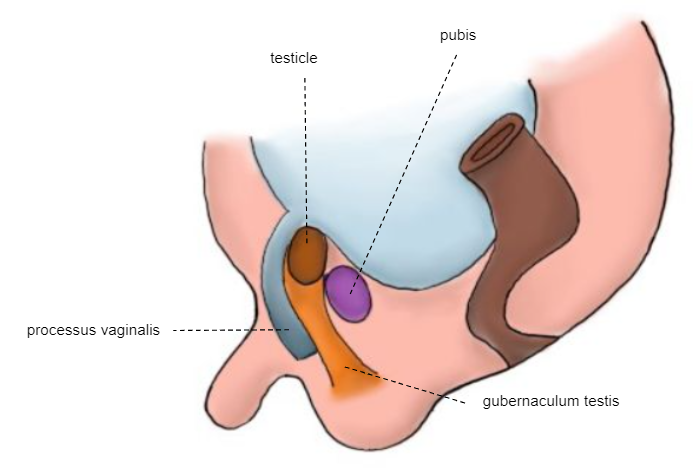
Figura 6 Embrião da 28ª semana
Recém-nascido a termo
O testículo é fixado ao escroto (Figura 7)
Os fatores mecânicos que poderiam desencadear a descida testicular ao escroto são:
- Tração distal gerada pelo gubernáculo testicular.
- Pressão intra-abdominal.
Outros possíveis fatores determinantes incluem fatores hormonais e neurológicos.2
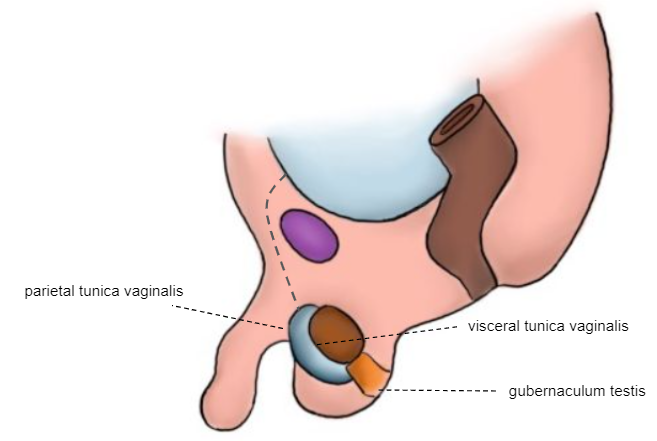
Figura 7 Recém-nascido
Como mostrado nas ilustrações embriológicas sequenciais anteriores, o processo vaginal (PV) começa a obliterar-se pouco antes do nascimento. O fechamento completo é normalmente obtido durante o primeiro ano de vida. A túnica vaginal (TV) permanece envolvendo o testículo. A falha no processo de obliteração do PV está então associada a hérnia, hidrocele e hidrocele encistada do cordão espermático.
Epidemiologia
A incidência de hérnia inguinal indireta congênita em recém-nascidos a termo é de 3.5–5%, enquanto em prematuros é consideravelmente mais alta e varia de 9–11%. Aproxima-se de 60% à medida que o peso ao nascer diminui de 500 a 750 gramas. A maioria das séries relata uma predominância do sexo masculino sobre o feminino variando de 5:1 a 10:1.3
Por outro lado, as hidroceles comunicantes são comuns em recém-nascidos, com uma incidência de 2–5%; 90% destas se resolvem durante o primeiro ano de vida como resultado do fechamento espontâneo.4
Hérnias inguinais congênitas são mais comuns no lado direito (60%), 30% ocorrem no lado esquerdo e 10% são bilaterais. Essa distribuição não se altera com o sexo, pois mesmo em meninas existe predominância do lado direito.5
Etiologia / Patogênese
A anatomia do canal inguinal varia ligeiramente com a idade. Em adultos e crianças, os orifícios inguinais interno e externo estão amplamente separados, ao passo que em lactentes jovens eles praticamente se sobrepõem. Nas meninas, a anatomia é semelhante, exceto pela ausência dos elementos espermáticos, que são substituídos pelo ligamento redondo.3 Os diferentes tipos de hidroceles estão ilustrados na imagem abaixo (Figura 8)
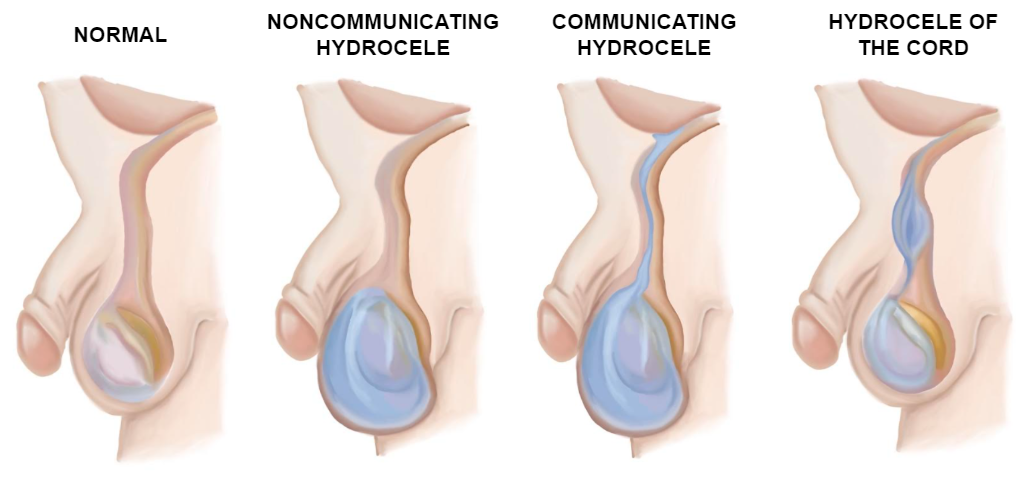
Figura 8 Tipos de hidroceles
Hérnia inguinal indireta congênita pode formar-se, como já foi mencionado, a partir de um PV permeável, permitindo a extrusão de omento, intestinos, apêndice ou até mesmo ovários ou trompa de Falópio em indivíduos do sexo feminino pelo canal inguinal. Um saco herniário também pode estender-se da cavidade abdominal, através do anel inguinal interno até a TV, resultando na chamada hérnia inguinoescrotal.
Hérnias inguinais diretas são muito incomuns na população pediátrica; são observadas principalmente em adultos, causadas por fraqueza da musculatura abdominal posterior que permite que os intestinos deslizem para a região inguinal.
Diagnóstico e Avaliação
Hérnia inguinal e hidrocele podem ser diagnosticadas por exame físico sem a necessidade estrita de exames de imagem. A presença de um abaulamento firme, redutível e assintomático na virilha ou no escroto em meninos, ou de uma massa nos grandes lábios em meninas, é sugestiva de hérnia inguinal.
Deve-se suspeitar de encarceramento do saco herniário em paciente que apresenta dor intensa de início súbito e uma massa dura, dolorosa à palpação e fixa na região inguinal. Se não for reduzido manualmente, deve-se proceder à redução cirúrgica.6,7
O exame físico deve incluir a avaliação do paciente em decúbito dorsal, reforçando, se possível, a manobra de Valsalva para identificar hérnias transitórias; e a avaliação em ortostatismo, na qual o médico deve palpar e comparar ambas as virilhas em busca de espessamento do cordão espermático ou de um abaulamento. Nos pacientes do sexo masculino, deve-se verificar a correta descida dos testículos, e a transiluminação (fonte de luz sobre o escroto mostrando ausência da sombra testicular) pode, às vezes, ajudar a confirmar a hidrocele.
Dicas
- Durante a palpação, é fundamental sentir o cordão espermático deslizar sob as polpas digitais.
- Sugerimos palpar o cordão espermático com a mão mais hábil.
- Em recém-nascidos, as hérnias podem ser mais frequentemente reduzidas. Isso deve ser feito comprimindo o conteúdo herniado para cima com a mão mais hábil, enquanto se exerce pressão para baixo com a outra mão, de modo a alinhar ambos os anéis inguinais e dirigir os elementos herniados para a cavidade abdominal (Figura 9)
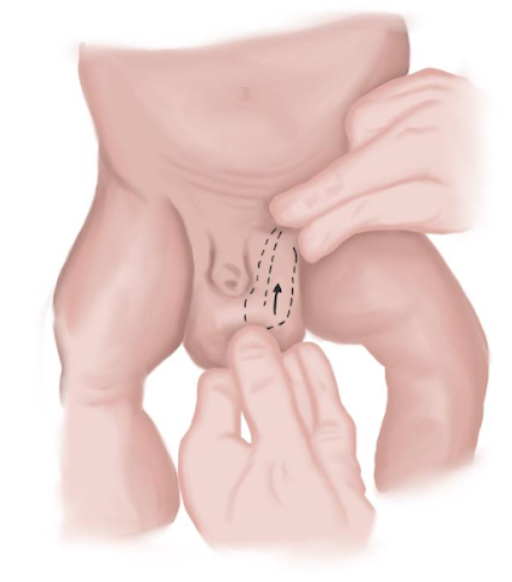
Figura 9 Manobra de redução de hérnia em neonatos.
Se o exame físico não for conclusivo, a ultrassonografia inguinal pode acrescentar informações ao diagnóstico suspeito.8 Ainda assim, o exame físico continua sendo o método diagnóstico padrão-ouro. A sensibilidade da ultrassonografia depende principalmente da habilidade do operador e da cooperação do paciente (imobilidade e manobra de Valsalva). O conteúdo herniário pode ser hiperecogênico devido à gordura omental, anecogênico na presença de líquido, ou de ecogenicidade mista com reverberações, o que se correlaciona com a existência de ar no lúmen das alças intestinais.9 A US também pode excluir uma hérnia inguinal contralateral, ou PV que pode ser reparado na mesma cirurgia.
Diagnóstico diferencial
O diagnóstico diferencial das lesões inguinais é frequentemente desafiador devido às semelhanças semiológicas no exame físico. Uma vez detectada uma massa palpável dolorosa, o médico deve ter em mente os seguintes diagnósticos possíveis, valendo-se de outros exames complementares caso o exame físico não seja suficiente:
- Testículo não descido.
- Hematoma / Inflamação.
- Abscesso.
- Tumores benignos ou malignos / linfadenomegalia metastática ou benigna.
- Varicosidades do ligamento redondo.
- Cisto mesotelial.
Opções de Tratamento
O tratamento da hérnia inguinal é sempre cirúrgico, e o aspecto-chave do tratamento é realizar uma ligadura alta do saco herniário.
Em lactentes e crianças pequenas, a herniotomia pode ser realizada pelo orifício inguinal externo, sem abrir a aponeurose, pois ambos os orifícios geralmente se sobrepõem. Em pacientes mais velhos, os orifícios encontram-se separados entre si. Nesses casos, é aconselhável abrir a aponeurose do oblíquo externo para se obter uma ligadura alta do saco.3 O uso de tela parietal em pacientes pediátricos está reservado para casos especiais.
Tanto para a hidrocele quanto para as hérnias inguinais, os princípios e as técnicas cirúrgicas são muito semelhantes. Quando a hidrocele é tratada cirurgicamente, além da ligadura alta do saco, a porção distal dele também deve ser ressecada. Essa etapa não é obrigatória para as hérnias inguinais.
As hérnias inguinais podem ser abordadas por meio de uma herniorrafia inguinal aberta (OIH) ou de uma correção laparoscópica de hérnia inguinal (LIHR). Os componentes mais importantes do procedimento envolvem garantir que o saco herniário seja esvaziado e completamente fechado após a cirurgia, preservando a integridade do ducto deferente, dos vasos testiculares e do nervo ilioinguinal. Em meninas, a fixação do ligamento redondo deve ser realizada para assegurar a fixação uterina adequada, prevenindo dispareunia futura.
Herniorrafia inguinal aberta
As principais etapas da OIH são detalhadas abaixo com desenhos esquemáticos das manobras.
- Incisão inguinal transversa seguindo a prega cutânea natural sobre o cordão (Figura 10)
- Dissecção do tecido celular subcutâneo, incisando a fáscia de Scarpa até alcançar a aponeurose muscular.
- Incisão sobre a aponeurose muscular (nem sempre necessária) (Figura 11)
- Extensão da incisão da aponeurose em direção ao orifício inguinal superficial.
- O cordão inguinal é então dissecado em ambos os lados e por sua face inferior com manobras rombas. Em seguida, o cordão é laçado com um laço elástico.z
- O músculo cremáster é aberto por dissecção romba na face ânteromedial do cordão e afastado para expor o peritônio brilhante do saco herniário indireto (Figura 12)
- O cordão e seu conteúdo (vasos, ducto deferente) são dissecados delicadamente, separando-o do saco, que estará na situação ânteromedial.
- Os conteúdos do cordão são cuidadosamente afastados do saco, que é descolado deste (Figura 13)
- O saco herniário é aberto e seu conteúdo é reduzido para a cavidade abdominal.
- O saco herniário é torcido, garantindo que não haja conteúdo em seu interior. Um nó de poliglactina absorvível é dado em sua base e o saco é seccionado e solto na cavidade abdominal (Figura 14), (Figura 15), e (Figura 16)
- A aponeurose muscular é fechada com uma sutura contínua de poliglactina absorvível, se for aberta (Figura 17)
- Nós separados são usados para aproximar o tecido subcutâneo e uma sutura intradérmica é usada para aproximar o tecido cutâneo (Figura 18)
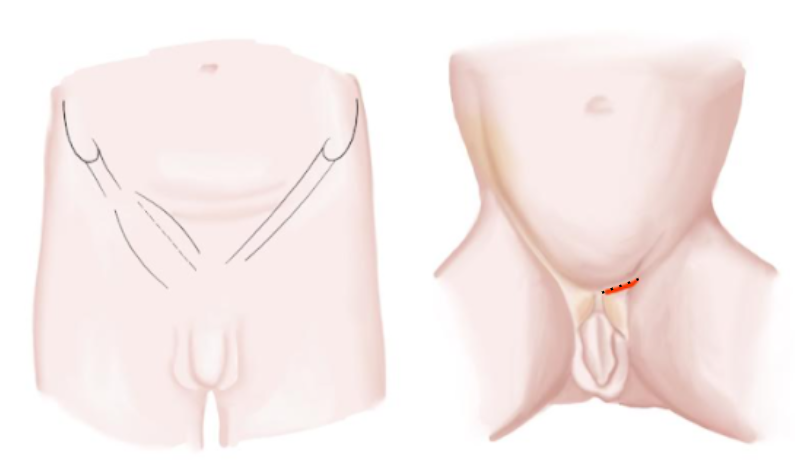
Figura 10 Incisão inguinal em lactentes e adolescentes
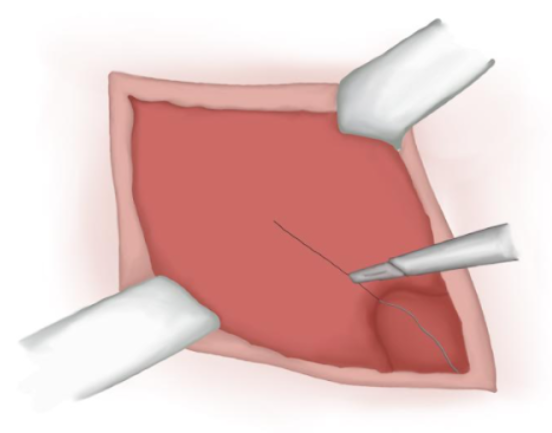
Figura 11 Incisão sobre a aponeurose muscular.
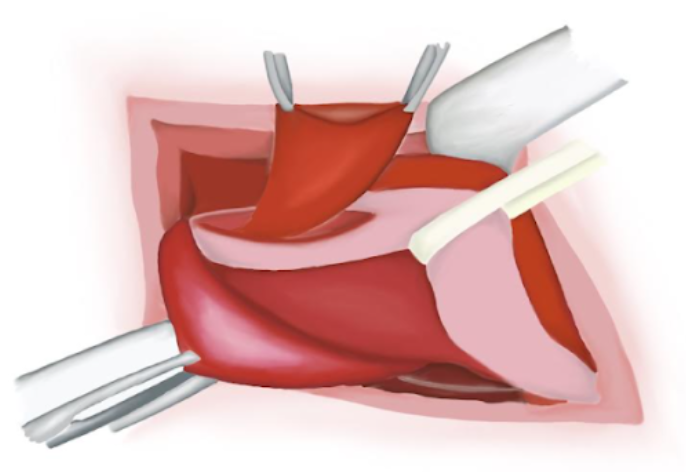
Figura 12 Identificação do cordão espermático. Dissecção do saco herniário.
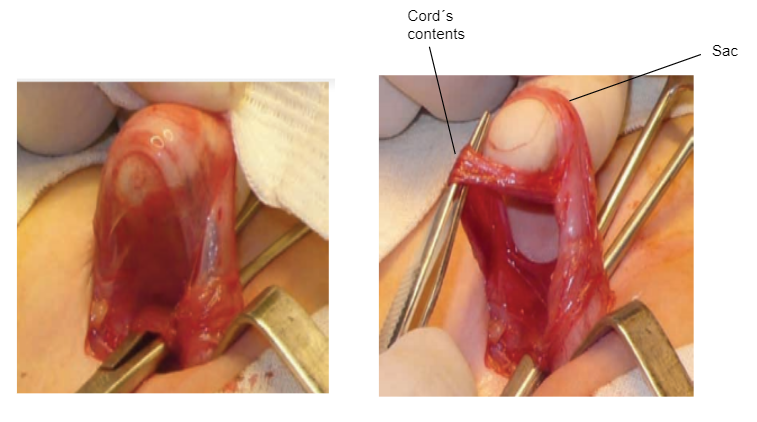
Figura 13 Dissecção do saco herniário.
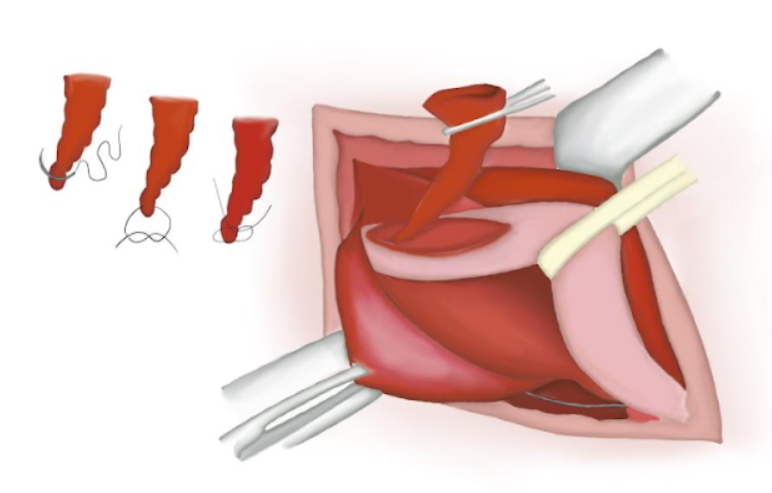
Figura 14 O saco herniário é torcido, garantindo que não haja conteúdo no interior.
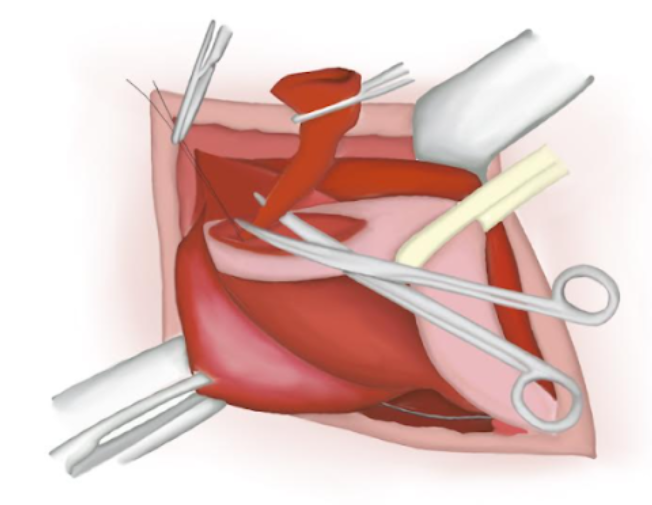
Figura 15 Saco herniário ressecado.
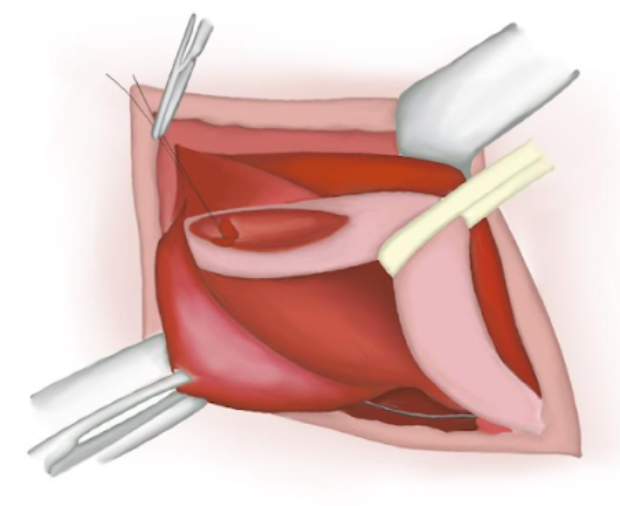
Figura 16 Extremidade proximal suturada perdida na cavidade abdominal.
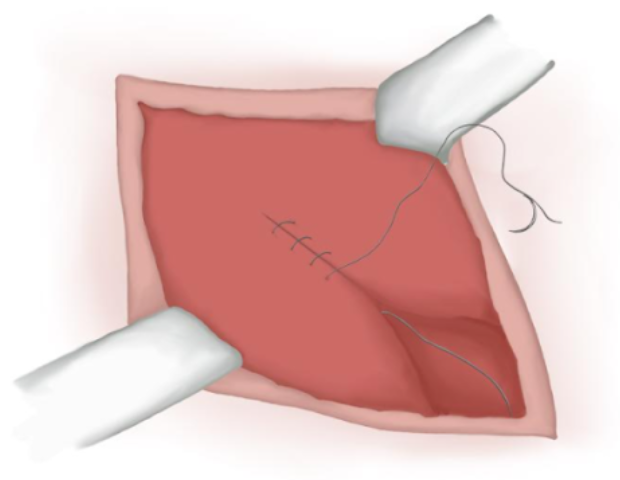
Figura 17 Fechamento da aponeurose muscular.
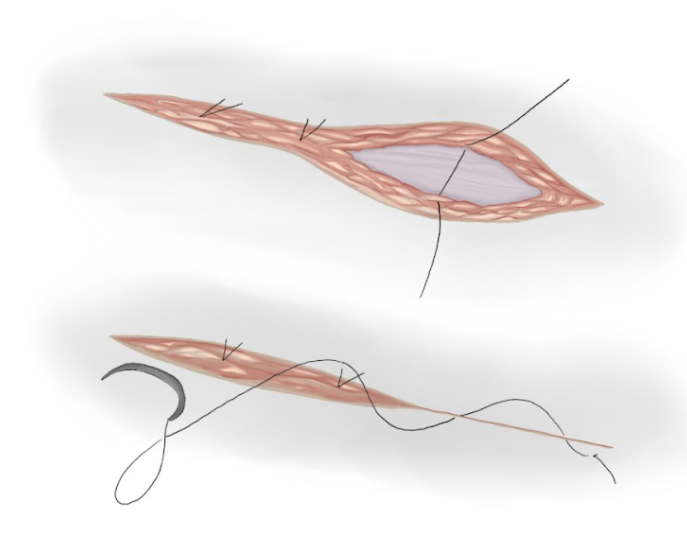
Figura 18 Fechamento do subcutâneo e da pele.
Dicas
- Os autores recomendam uma incisão cutânea transversa seguindo a prega cutânea.
- Localizar a arcada crural no plano aponeurótico em casos mais complexos (meninas com hérnia bilateral, crianças obesas) para localizar o orifício inguinal superficial.
- Dissecar o saco herniário sempre do mesmo lado do cordão de maneira regrada
Correção laparoscópica da hérnia inguinal
O papel da correção laparoscópica das hérnias inguinais não está totalmente estabelecido. A abordagem é criticada por transformar um procedimento extraperitoneal em um transperitoneal. Os detratores sustentam que essa técnica implica uma cirurgia tecnicamente mais complexa e que resulta em uma taxa de recidiva maior do que a abordagem convencional.
Em contraste, os defensores da abordagem laparoscópica enfatizam que essa técnica assegura menor invasividade, sem manipulação das estruturas do funículo espermático, e pode ser útil para o diagnóstico correto de hérnias diretas e femorais associadas. Além disso, o anel interno contralateral pode ser claramente visualizado e reparado, se necessário. Finalmente, os defensores endossam a melhor cosmese resultante após essa cirurgia em comparação com uma incisão convencional (Tabela 1).10
Tabela 1 A tabela a seguir apresenta algumas vantagens e desvantagens relativas à LIHR:
| VANTAGENS | DESVANTAGENS |
|---|---|
| Técnica simples. | Custos. |
| Procedimento ambulatorial. | A duração da cirurgia pode ser mais longa. |
| As estruturas do cordão permanecem intocadas. | É necessária anestesia geral com intubação endotraqueal. |
| O tipo de hérnia é evidente, assim como o anel inguinal contralateral. | Maior possibilidade de lesão de órgãos intra-abdominais. |
| Visualização clara da anatomia. | Curva de aprendizado mais longa. |
| Inspeção dos anexos em meninas. | Possível lesão do cordão no longo prazo |
Cuidados Pós-Operatórios e Acompanhamento
Como se trata de uma cirurgia de um dia, no pós-operatório, alimentos e líquidos podem ser iniciados após a recuperação da anestesia. Lactentes com displasia broncopulmonar, anemia, prematuridade, ou aqueles que necessitaram de suporte ventilatório ao nascimento devem ser observados após a correção cirúrgica por pelo menos 24 horas, monitorados quanto a episódios de apneia e/ou bradicardia. Os pacientes devem receber alta com analgésicos orais.
A atividade deve ser limitada por 48 horas. Retornar às atividades normais assim que a criança se sentir capaz. Em crianças com mais de 6 anos, as restrições de atividade devem ser mais rigorosas, sem atividade vigorosa por um período de 1 mês. Os banhos podem ser iniciados no terceiro dia após a cirurgia.
O seguimento deve ser realizado 1 semana após a cirurgia para avaliar complicações precoces, seguido de controles clínicos em 1 mês, 3 e 6 meses após a cirurgia, antes da alta.
Desfechos, Complicações e Manejo
Durante a cirurgia, as complicações locais incluem sangramento e lesão dos vasos espermáticos e do canal deferente. O uso de lupas de aumento durante a reconstrução de lesões intraoperatórias tornará o reparo mais preciso, especialmente em recém-nascidos.
As complicações precoces após a cirurgia podem incluir edema local, hematoma e infecção da ferida.
Complicações tardias observadas durante o seguimento podem incluir hidrocele ou hérnia recorrentes, deslocamento iatrogênico do testículo e atrofia testicular. Para evitar hidrocele pós-operatória, as porções anterior e lateral do saco podem ser parcialmente ressecadas, ou o saco distal pode ser aberto completamente.
A recorrência é maior em pacientes do sexo masculino operados antes do primeiro ano de vida e naqueles tratados por abordagem laparoscópica. A recorrência reduz-se significativamente quando o procedimento é realizado por um cirurgião pediátrico ou urologista pediátrico experiente e habilidoso, ou se for uma cirurgia programada.
A hérnia inguinal recorrente em crianças pode incluir:
- Um saco herniário não identificado ou laceração peritoneal não reconhecida.
- Ligadura inadequada do saco herniário.
- Falha em reparar um grande anel inguinal interno.
- Defeito não reconhecido do assoalho do canal inguinal (hérnia inguinal direta).
- Infecção.
- Aumento da pressão intra-abdominal (pacientes com derivações ventrículo-peritoneais ou diálise peritoneal ambulatorial contínua).
- Pacientes com fibrose cística e tosse crônica.
- Distúrbios do tecido conjuntivo (isto é, síndrome de Ehlers-Danlos).
Pontos-chave
- A literatura sobre hérnia inguinal abrange mais de 20 séculos. Galeno, em 176 d.C., descreveu pela primeira vez a patogênese da hérnia inguinal indireta ao retratar pela primeira vez o “Processus Vaginalis Peritonei”.
- A incidência de hérnia inguinal indireta congênita em neonatos a termo é de 3,5–5%, enquanto em prematuros é de 9–11%.
- Epidemiologia: Observada principalmente no sexo masculino, com uma razão masculino:feminino variando de 5:1 a 10:1.
- Diagnóstico tanto da hérnia inguinal quanto do hidrocele pode ser feito por exame físico e confirmado sem necessidade obrigatória de exames de imagem.
- Tratamento da hérnia inguinal é sempre cirúrgico. Pode ser abordada por herniorrafia inguinal aberta (OIH) ou reparo laparoscópico de hérnia inguinal (LIHR).
- O tratamento cirúrgico é geralmente ambulatorial. Lactentes com displasia broncopulmonar, anemia, prematuridade, ou aqueles que necessitaram de suporte ventilatório ao nascimento devem ser observados após o reparo cirúrgico por pelo menos 24 horas.
- Seguimento deve ser feito 1 semana após a cirurgia para complicações precoces, seguido de controles clínicos com 1 mês, 3 e 6 meses pós-operatório.
- Complicações precoces após a cirurgia podem incluir edema local, hematoma e infecção de ferida operatória.
- Complicações tardias observadas no pós-operatório durante o seguimento incluem hidrocele ou hérnia recorrentes, deslocamento iatrogênico do testículo e atrofia testicular.
Conclusões
Processo vaginal patente (PV) leva a hidrocele congênita e hérnia inguinal. Massa palpável na região inguinal deve ser avaliada quanto a possíveis condições associadas ao PV. O exame físico é o método diagnóstico padrão-ouro para essas condições. O tratamento da hérnia inguinal e da hidrocele comunicante com hérnia associada é sempre cirúrgico. O sucesso do tratamento cirúrgico depende de uma ligadura alta do saco, bem como de sua ressecção distal nas hidroceles. As abordagens incluem herniorrafia inguinal aberta, assim como laparoscópica.
Complicações podem ser observadas durante a cirurgia ou em fase pós-operatória precoce ou tardia, incluindo, respectivamente, edema, hematoma ou hidrocele recorrente, ou hérnia. Várias estratégias são descritas por especialistas para evitá-las. O seguimento deve ser realizado periodicamente por pelo menos 6 meses.
Persistem controvérsias quanto ao manejo da hérnia e da hidrocele, especialmente no que tange à técnica cirúrgica com base nas taxas de recorrência e complicações, buscando procedimentos minimamente invasivos com melhores desfechos.
Referências
- Gearhart JP, Rink RC, Mouriqand PDE, editors. Pediatric Urology. 2nd ed., Philadelphia, PA: Elsevier; 2010, DOI: 10.1097/00042307-200011000-00007.
- Palermo M. Hernias de la pared abdominal - Conceptos clásicos, evidencias y nuevas técnicas. Actualidades Médicas, C.A: AMOLCA; 2012.
- J. T. Chapter 59: Inguinal Hernia. In: Puri P, editor. Newborn Surgery. 2nd ed. Boca Raton, FL: CRC Press; 2003. DOI: 10.5005/jp/books/12263_1.
- Godbole P.P. MNP. Chapter 18: Testis, Hydrocele and Varicocele. In: Wilcox DT, Thomas DFM, editors. Essentials of Pediatric Urology. 2nd ed. Boca Raton, FL: CRC Press; 2021. DOI: 10.1201/9781003182023.
- Rowe MI, Clatworthy HW. The other side of the pediatric inguinal hernia. Surg Clin North Am 1971; 51: 1371. DOI: 10.1016/s0039-6109(16)39592-5.
- Choi KH, Baek HJ. Incarcerated ovarian herniation of the canal of Nuck in a female infant: Ultrasonographic findings and review of literature. Annals of Medicine and Surgery 2016; 9: 38–40. DOI: 10.1016/j.amsu.2016.06.003.
- Stringer MD, Oldham KT, P.D.E. M, editors. Pediatric Surgery and Urology Long-term Outcomes. 2nd ed., Cambridge, UK: Cambridge University Press; 2010.
- Park HR, Park SB, Lee ES, Park HJ. Sonographic evaluation of inguinal lesions. Clinical Imaging 2016; 40 (5): 949–955. DOI: 10.1016/j.clinimag.2016.04.017.
- Yang DM, Kim HC, Lim JW, Jin W, Ryu CW, Kim GY. Sonographic findings of groin masses. J Ultrasound Med 2007; 26: 605–614. DOI: 10.7863/jum.2007.26.5.605.
- Holcomb GW, Georgeson KE, Rothenberg SE, editors. Atlas of Pediatric Laparoscopy and Thoracoscopy. Amsterdam, Netherlands: Elsevier; 2021, DOI: 10.1007/978-3-030-84467-7_61.
Ultima atualização: 2025-09-21 13:35