18: Divertículos, Anomalias do Úraco e Utrículos
Este capítulo levará aproximadamente 24 minutos para ler.
Anomalias do úraco
Embriologia e Anatomia
O úraco é uma estrutura tubular que conecta o alantóide, no umbigo, à cúpula da bexiga e permanece pérvio durante a gestação.1 O lúmen normalmente se fecha por volta da 12ª semana de gestação e se oblitera completamente. Após a obliteração, o que tipicamente permanece é um cordão fibroso que se estende do aspecto inferior do umbigo até a cúpula da bexiga. O úraco é extraperitoneal e facilmente visualizado durante a visualização laparoscópica da pelve (Figura 1).
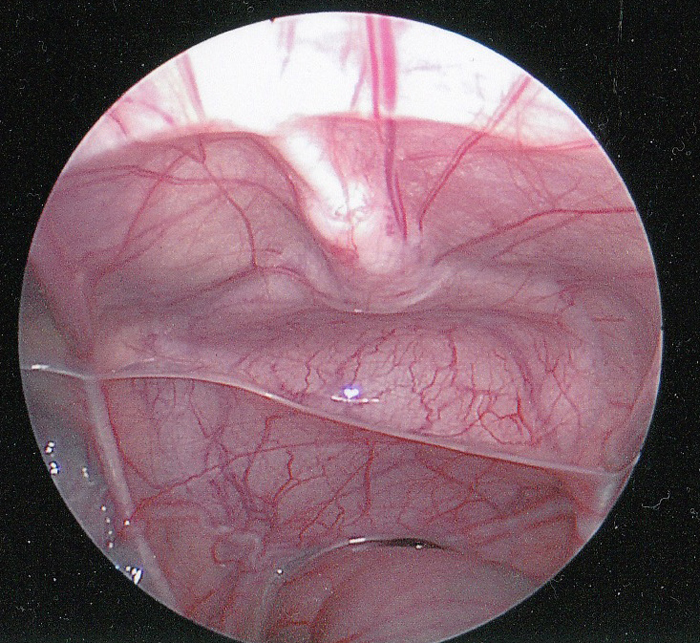
Figura 1 Visão laparoscópica intraperitoneal do úraco. O cordão fibroso do úraco (ligamento umbilical mediano) é mostrado na linha média, estendendo-se até a cúpula da bexiga. O reto encontra-se inferiormente. O ducto deferente pode ser visto estendendo-se lateralmente logo acima do reto.
O úraco é recoberto pelas pregas do peritônio, formando o ligamento umbilical mediano. Raramente, o úraco pode apresentar falhas no cordão fibroso ou até obliteração completa do cordão. É um marco cirúrgico importante para marcar a cúpula da bexiga e garantir a colocação adequada de uma vesicostomia.
Classificação das anomalias do úraco
Anomalias do úraco resultam de falha na obliteração completa do lúmen durante a gestação.2 Sua classificação anatômica baseia-se no grau de persistência da permeabilidade do úraco e varia desde permeabilidade completa com livre fluxo de urina até um pequeno trajeto sinusal com fundo cego a partir da pele (Figura 2). Um cisto uracal pode estar presente em qualquer local ao longo do úraco, mas é mais comumente encontrado próximo à cúpula vesical. Um divertículo uracal—a anomalia mais rara descrita—é uma permeabilidade parcial do úraco que drena para a cúpula vesical.
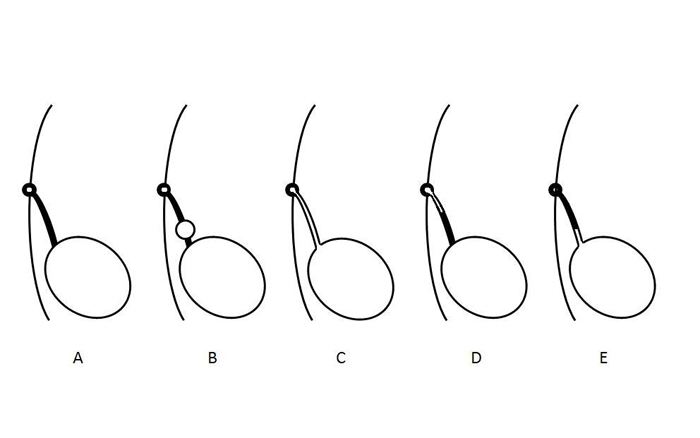
Figura 2 Diagrama que ilustra diferentes tipos de anomalias do úraco. A- Obliteração normal do lúmen do úraco. B- Cisto uracal. C- Úraco pérvio. D- Seio uracal. E- Divertículo uracal. Essas anomalias não são mutuamente exclusivas e podem ocorrer em combinação.
A incidência relativa dos diferentes tipos de anomalias do úraco provenientes de várias séries clínicas é mostrada na Tabela 1.3,4,5,6,7,8,9
Tabela 1 Resumo dos tipos e da incidência das anomalias do úraco que se apresentam em crianças.
| Autor | Nº de pts | Cisto | Pérvio | Seio | Divertículo |
|---|---|---|---|---|---|
| Naiditch | 103 | 38 | 21 | 11 | 13 |
| Fox | 66 | 34 | 14 | 7 | 10 |
| Ashley | 46 | 25 | 6 | 14 | 1 |
| Cilento | 45 | 16 | 7 | 22 | 0 |
| Rich | 35 | 12 | 19 | 4 | 0 |
| Yiee | 31 | 19 | 7 | 5 | 0 |
| Copp | 29 | 7 | 3 | 17 | 2 |
| Total | 355 | 151 (43) | 108 (30) | 80 (23) | 26 (7) |
Curiosamente, há um único relato de caso de úraco pérvio que se fechou espontaneamente após 2 semanas de drenagem por cateter e persistiu como um divertículo uracal.10
Apresentação Clínica
Os sintomas de apresentação mais comuns em crianças com anomalias do úraco são drenagem umbilical persistindo por semanas após o parto ou uma massa e/ou dor decorrentes de infecção.3,6,8,11 A drenagem umbilical pode ser clara, serosa, purulenta ou sanguinolenta e pode orientar quanto à sua causa: vazamento persistente de líquido claro (provavelmente urina) em um lactente é sugestivo de úraco pérvio, enquanto líquido turvo, seroso ou sanguinolento é indicativo de um seio uracal ou cisto uracal. Há uma distribuição etária bimodal com apresentação em média aos 1-3 meses de idade para aqueles com seio uracal ou úraco pérvio versus 3 anos para os que se apresentam com cisto uracal.8 O diagnóstico diferencial da drenagem umbilical também inclui onfalite, remanescente do ducto onfalomesentérico, ou um granuloma umbilical.6
Tabela 2 Sintomas de Apresentação em Crianças com Anomalias do Úraco.
| Autor | # Pacientes | Drenagem | Massa/Infecção | Dor | Assintomático | Outros |
|---|---|---|---|---|---|---|
| Gleason | 721 | 26 | 19 | 17 | 667 | 0 |
| Naiditch | 103 | 60 | 7 | 4 | 18 | 12 |
| Stopak | 85 | 43 | 36 | 0 | 4 | 1 |
| Dethlefs | 68 | 52 | 32 | 0 | 8 | 0 |
| Cilento | 45 | 19 | 15 | 10 | 0 | 1 |
| Yiee | 37 | 20 | 8 | 4 | 2 | 3 |
| Total | 1059 | 220 (21) | 117 (11) | 35 (3) | 699 (66) | 17 (2) |
O exame físico também pode ser útil. Um úraco pérvio ou seio uracal pode se apresentar como uma fosseta ou depressão na base do umbigo (Figura 3).
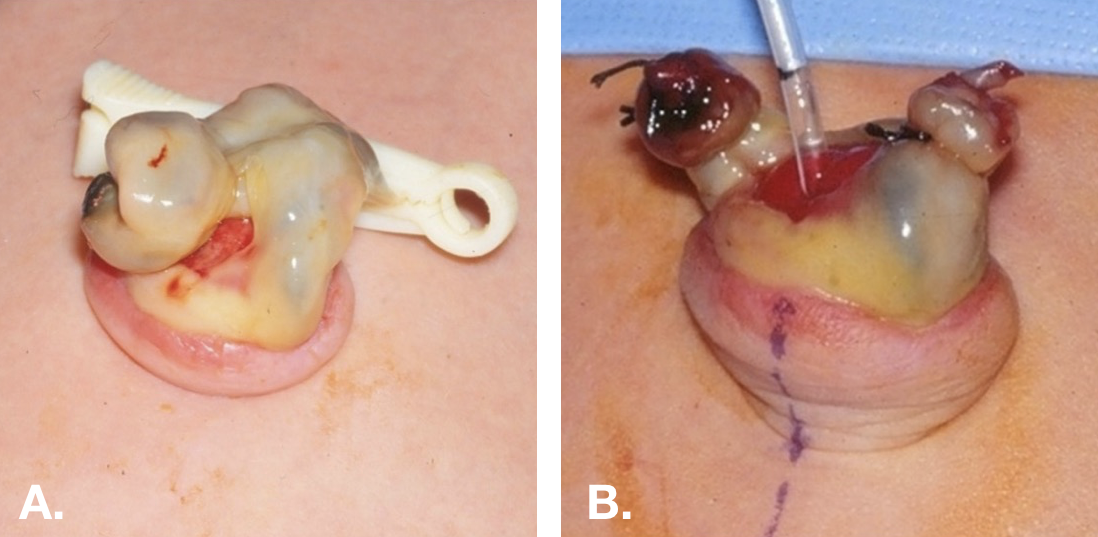
Figura 3 Úraco pérvio em um recém-nascido. A. Demonstra o aspecto vermelho vivo da extremidade umbilical de um úraco pérvio. B. A pele umbilical foi evertida e uma pequena sonda de alimentação foi passada através do úraco pérvio para dentro da bexiga.
Algumas anomalias do úraco são descobertas incidentalmente durante a avaliação radiográfica de rotina de outras afecções, como infecções do trato urinário ou hidronefrose antenatal. Cistos uracais provavelmente são identificados durante a ultrassonografia da bexiga (Figura 4).
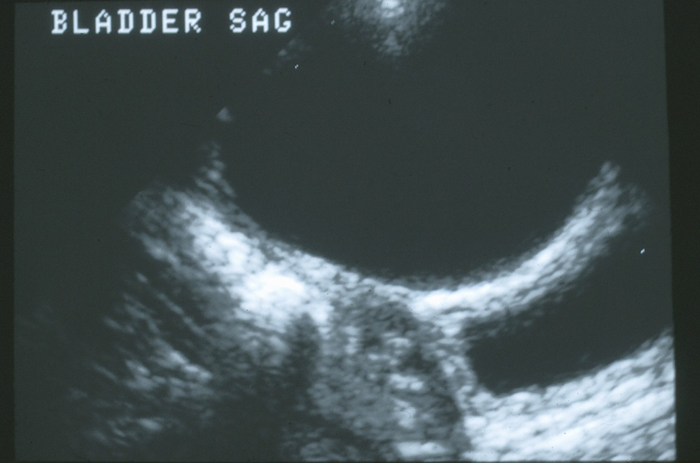
Figura 4 Visão sagital da cúpula vesical na linha média. Há um grande cisto uracal anecogênico anterior à parede vesical.
Carcinoma do úraco
Como o carcinoma uracal não é uma doença relatada como ocorrendo em crianças ou adolescentes, o tratamento dessa afecção não será incluído neste capítulo.5,12 Como será discutido na seção sobre manejo, o risco de uma anomalia uracal assintomática desenvolver carcinoma e, portanto, o valor da excisão profilática são desconhecidos. Fatores de risco para carcinoma em uma anomalia uracal incluem tamanho maior que 4 cm e idade > 55 anos. Calcificações são uma característica comum no carcinoma; no entanto, o significado de calcificações em um remanescente uracal benigno assintomático é desconhecido.5,12 Parece prudente remover lesões com calcificações, visto que calcificações podem estar associadas à inflamação crônica, que está associada à carcinogênese.
Avaliação Radiológica
A realização de exames de imagem é orientada pelos sintomas de apresentação e pelo grau de suspeita clínica.6,8 Um úraco pérvio que permite que a urina drene livremente pelo umbigo pode ser demonstrado por imagem com alta sensibilidade por meio de uma cistouretrografia miccional (VCUG) ou sinograma. Tradicionalmente, uma VCUG era realizada para identificar o úraco pérvio e também fornecer uma avaliação anatômica da bexiga e avaliar a presença de obstrução da via de saída vesical ou refluxo vesicoureteral (Figura 5). No entanto, a ultrassonografia abdominal é adequada para o diagnóstico na maioria dos pacientes, mais amplamente disponível, menos invasiva e menos custosa; assim, tornou-se o padrão para o diagnóstico, e a VCUG é reservada para casos complicados.3,13 É importante informar ao ultrassonografista sobre a suspeita clínica de anomalia uracal para que a parede abdominal na linha média seja examinada minuciosamente. Cistos uracais infectados apresentam-se como grandes massas heterogêneas com coleções líquidas complexas (Figura 6).
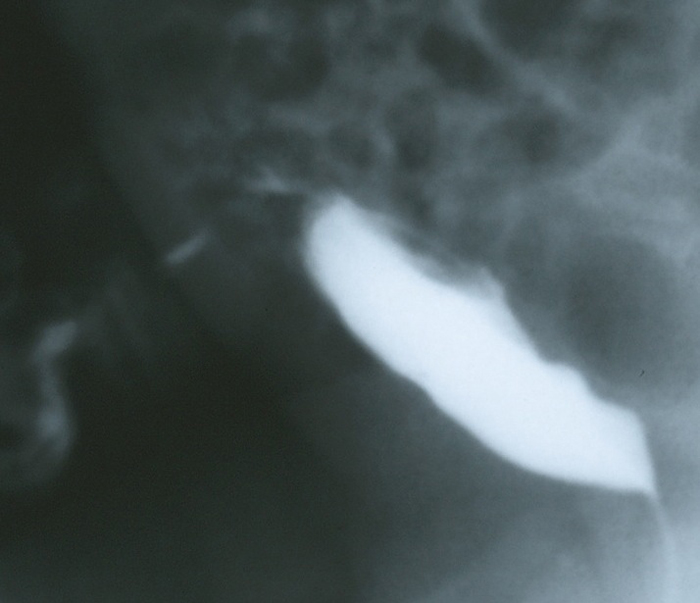
Figura 5 Projeção lateral durante o enchimento de uma VCUG em um lactente com grandes quantidades de líquido claro drenando pelo umbigo. A imagem demonstra contraste drenando através da parede abdominal anterior a partir da cúpula da bexiga por meio do úraco pérvio.
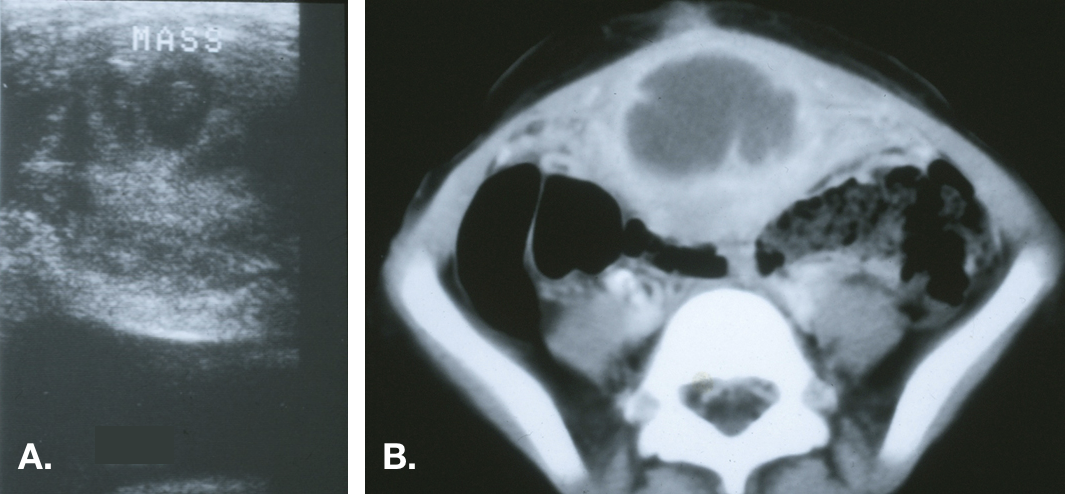
Figura 6 Cisto do úraco infectado. A. Vista transversal do domo vesical na linha média demonstrando uma massa ecogênica heterogênea acima da bexiga. B. Tomografia computadorizada mostrando a massa na linha média com densidade central de líquido consistente com um abscesso e estriações inflamatórias da parede abdominal anterior adjacente.
As lesões podem ter vários centímetros de diâmetro e podem, ocasionalmente, estender-se além do espaço pré-peritoneal e perfurar a cavidade peritoneal.14,15
A TC não deve ser considerada parte integrante da investigação diagnóstica de rotina mas pode detectar anomalias não vistas pela ultrassonografia e, assim, é útil em casos de alta suspeita clínica sem diagnóstico claro (Figura 7).
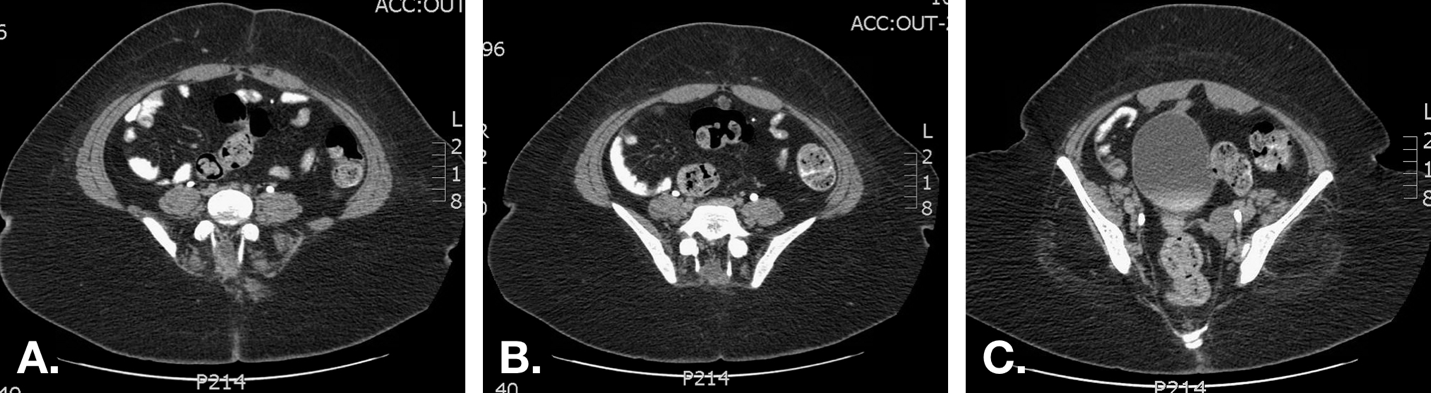
Figura 7 TC em paciente com drenagem umbilical sanguinolenta recorrente. A. Imagem logo abaixo do umbigo demonstra um úraco de aspecto normal (cordão fibroso) logo inferior aos músculos retos abdominais na linha média. B. Um cisto do úraco é demonstrado mais inferiormente. C. Demonstra-se a proximidade entre a extensão inferior do cisto e a cúpula da bexiga.
A avaliação também deve incluir uma ultrassonografia renal para assegurar a ausência de hidronefrose ou outras anomalias renais congênitas.6,8,9,16 A incidência de anomalias renais concomitantes tem variado amplamente nas séries publicadas, mas, dada a ausência de morbidade e de risco com a ultrassonografia, é prudente incluir a avaliação por imagem dos rins como parte da investigação diagnóstica.4
Tratamento
Em geral, remanescentes uracais sintomáticos devem ser tratados com excisão cirúrgica. Isso deve incluir excisão completa do úraco desde o umbigo até a cúpula vesical (Figura 8).
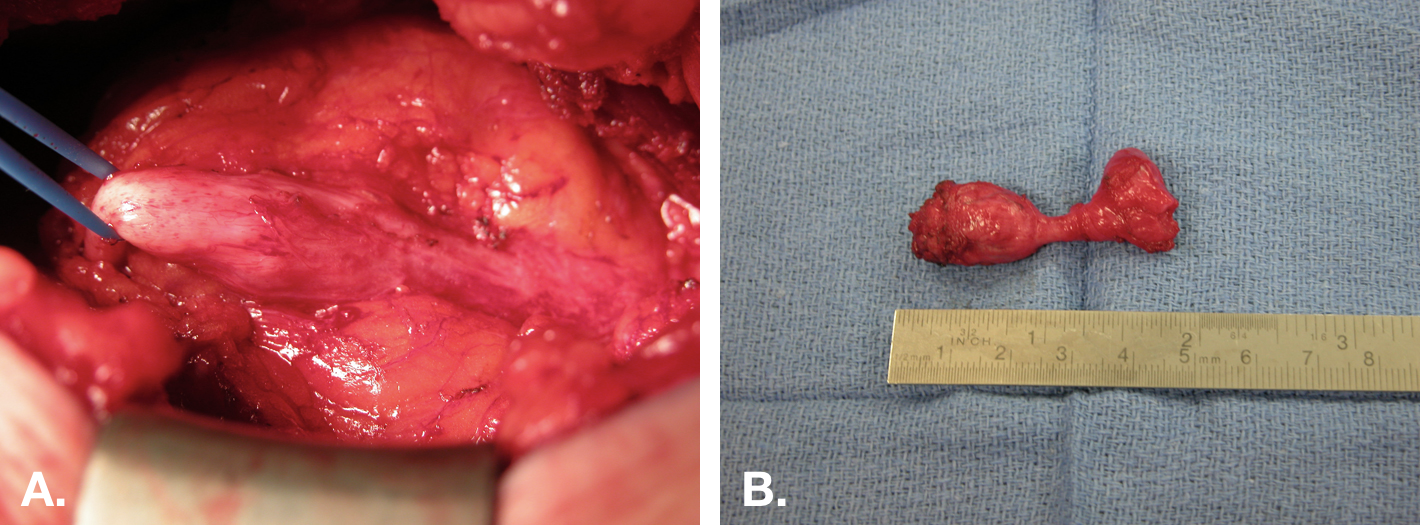
Figura 8 Excisão aberta de cisto do úraco. A. Vista intraoperatória de uma excisão de cisto do úraco por meio de uma incisão mediana infraumbilical. O cisto está ao centro, com a inserção na bexiga à direita e o cordão fibroso estendendo-se até o umbigo à esquerda. B. A peça (cisto) após a excisão, com um pequeno manguito vesical (extremidade direita da peça).
A necessidade de ressecar a mucosa vesical em cistos ou seios uracais que não têm continuidade com a parede vesical é controversa.2,6,7,17,18 Cistos uracais infectados representam outro dilema, pois o acentuado aumento do cisto devido à infecção e à inflamação circundante torna a excisão simples mais difícil e pode aumentar o risco de complicações. Pode-se abordar o cisto uracal infectado por meio de uma abordagem em tempo único ou em estágios.7,17,19 Os méritos e riscos relativos das duas abordagens são discutidos mais adiante.
A excisão cirúrgica do remanescente do úraco é curativa e não há sequelas funcionais decorrentes de sua excisão, pois se trata de um remanescente vestigial. O principal dilema cirúrgico ocorre em pacientes que se apresentam com uma lesão assintomática incidentalmente descoberta em exames de imagem.
Manejo conservador
Existem dados limitados sobre as sequelas a longo prazo—a saber, infecção recorrente e desenvolvimento de carcinoma—de anomalias do úraco mantidas in situ. Historicamente, favorecia-se a excisão devido ao risco presumido de transformação maligna, porém, publicações recentes defendem uma abordagem conservadora.10,20 O fechamento do úraco pérvio e a regressão de cistos uracais—mesmo no contexto de infecção—foram relatados.21,22,23 A recorrência de sintomas após a drenagem percutânea de cistos infectados parece ser rara.23 Em uma série, 92% dos lactentes < 6 meses que foram observados tiveram resolução da infecção, ao passo que 60% dos que foram submetidos à excisão desenvolveram infecção pós-operatória, indicando que o manejo conservador até pelo menos 6 meses de idade pode ser prudente.21
Para cistos uracais infectados, devem ser obtidas urocultura e cultura da drenagem da ferida, se presente. O microrganismo mais comum cultivado a partir de cistos uracais infectados é o Staphylococcus aureus (Tabela 3).5,21,18,20 Se o paciente estiver estável, afebril, não tóxico, sem sinais peritoneais, sem celulite disseminada ou sinais de fascite, pode-se considerar o manejo inicial apenas com antibióticos.
Tabela 3 Espécies microbianas cultivadas a partir de remanescentes uracais infectados. * E Coli-3, Citrobacter, Enterococcus e Proteus.
| Autor | # Pacientes | S. Aureus | Strep sp | Outros * |
|---|---|---|---|---|
| Stopak | 16 | 9 | 0 | 2 |
| McCollum | 9 | 6 | 1 | 2 |
| Minevich | 9 | 9 | 0 | 0 |
| Ashley | 9 | 6 | 1 | 2 |
| Galati | 5 | 5 | 0 | 0 |
| Newman | 5 | 3 | 0 | 2 |
| Total | 53 | 38 (72) | 2 (4) | 8 (15) |
Para pacientes instáveis ou aqueles que não respondem ao tratamento conservador, o cisto infectado pode ser tratado por excisão cirúrgica completa inicial ou por drenagem percutânea (guiada por US ou TC) ou aberta. Na excisão em um único estágio do úraco infectado, as margens de ressecção serão maiores, resultando em um defeito maior de partes moles, e a inflamação pode estender-se ao intraperitônio, o que coloca as vísceras abdominais em risco. Há um risco pequeno, porém significativo, de fístula enterocutânea e uma maior taxa de complicações da ferida operatória após a excisão primária de um cisto uracal infectado, em comparação à drenagem.21,18,19,24 Drenos percutâneos podem drenar de forma incompleta coleções líquidas multiloculadas. Tradicionalmente, todos os cistos eram ressecados após a resolução da inflamação, em um procedimento em estágios. Vários estudos recentes demonstraram que a infecção recorrente é incomum e, portanto, a excisão tardia pode não ser necessária.
Excisão Laparoscópica do Úraco
Nesta era da cirurgia minimamente invasiva, surgiram múltiplos relatos de ressecção laparoscópica e, mais recentemente, laparoscópica assistida por robô, de remanescentes uracais em crianças.25,26,27 A cirurgia laparoscópica de sítio único também foi relatada.28 A principal vantagem da técnica robótica é a facilidade de sutura em comparação com a laparoscopia pura.27 A visualização é excelente com a abordagem laparoscópica para a bexiga, mas pode ser mais desafiadora no umbigo se os portos não forem cuidadosamente posicionados. O úraco deve ser ressecado desde a base do umbigo até a cúpula da bexiga. Novamente, há controvérsia quanto a se a ressecção completa requer excisão da cúpula da bexiga.25,26
O posicionamento dos portais é uma consideração importante.26 Como o úraco se origina no umbigo, este não pode ser usado como local de portal. O local mais comum para a câmera é supra-umbilical (geralmente 1–2 cm). Isso fornece espaço de trabalho suficiente para visualizar a dissecção do umbigo até a cúpula da bexiga. Os portais de trabalho devem ser posicionados lateralmente em ambos os lados, geralmente ao nível do umbigo. Alternativamente, a colocação lateral dos portais de trabalho e da câmera (no lado direito ou esquerdo do abdome) oferece visualização lateral do úraco e mantém todos os portais em localização infra-umbilical. A configuração lateral dos portais tornará a sutura do fechamento vesical mais difícil devido ao ângulo.

Figura 9 Diagrama do possível posicionamento dos portais para excisão do remanescente do úraco. Ao contrário da configuração para a maioria das cirurgias laparoscópicas, o umbigo não pode ser utilizado como local de portal, pois é a origem do úraco e deve ser visualizado durante a dissecção.
Excisão aberta do úraco
Em lactentes e crianças pequenas, a ressecção completa do úraco pode ser realizada com facilidade por meio de uma pequena incisão. Ela pode ser orientada transversalmente ou como uma incisão mediana vertical. Para lactentes, uma pequena incisão de 1-1.5 cm a meio caminho entre a púbis e o umbigo dará acesso ao úraco e permitirá a ressecção completa do umbigo até a cúpula vesical, com excelente exposição da cúpula vesical para o fechamento. Essa pequena incisão é comparável ao tamanho da incisão necessária para o portal de câmera de 12 mm do robô cirúrgico e mantém o procedimento inteiramente extraperitoneal, eliminando potenciais complicações intra-abdominais. Em crianças mais velhas ou obesas, ou em adolescentes, é prudente realizar uma incisão mediana vertical. Se houver dificuldade com a exposição, a incisão pode ser estendida para cima em direção ao umbigo ou para baixo em direção à bexiga para facilitar a remoção completa do úraco nesses pacientes. As taxas de complicações da excisão simples são muito baixas, e a cirurgia pode ser realizada em regime ambulatorial ou com breve internação se um cateter for deixado no local. Fugas de urina ou complicações da ferida estão mais comumente associadas à excisão em tempo único de cistos infectados, e não à excisão simples.18,19
Pontos-chave
- As anomalias do úraco apresentam-se mais comumente como drenagem umbilical persistindo por semanas após o parto ou como massa umbilical e/ou dor devido à infecção.
- O diagnóstico diferencial da drenagem umbilical inclui anomalia do úraco, onfalite, remanescente do ducto onfalomesentérico ou granuloma umbilical.
- O risco absoluto de uma anomalia do úraco assintomática desenvolver carcinoma é desconhecido. Fatores de risco para carcinoma em uma anomalia do úraco incluem tamanho maior que 4 cm e idade >55 anos.
- A ultrassonografia abdominal é adequada para o diagnóstico de anomalia do úraco na maioria dos pacientes. A investigação deve incluir ultrassonografia renal para avaliar anomalias renais congênitas.
- Remanescentes do úraco sintomáticos devem ser excisados por abordagem aberta, laparoscópica ou assistida por robô. Se for utilizada uma abordagem laparoscópica ou robótica, o umbigo não pode ser usado como ponto de acesso.
- Infecções não complicadas de cistos do úraco podem ser tratadas de forma conservadora com antibióticos ± drenagem percutânea. A excisão imediata acarreta risco significativo de infecção pós-operatória. A excisão tardia é razoável, mas pode não ser necessária, pois o fechamento dos remanescentes/cisto é possível e a infecção recorrente é rara, especialmente em crianças <6 meses de idade.
Utrículo Prostático
Embriologia e Anatomia
Em fetos masculinos, a produção de substância inibidora de Müller (MIS) pelas células de Sertoli sinaliza a rápida regressão dos ductos paramesonéfricos (de Müller)—que nas fêmeas se tornam as trompas de Falópio, oviduto, útero e os 2/3 superiores da vagina—nas 8-10ª semanas de gestação. O utrículo prostático é um pequeno remanescente dos ductos paramesonéfricos que persiste nos homens como uma expansão da uretra prostática.1 O utrículo origina-se na linha média da próstata, no aspecto cranial do verumontanum.
Apresentação Clínica
Os utrículos prostáticos (UP) não são clinicamente significativos, a menos que a regressão completa tenha sido prejudicada e o utrículo mantenha um aspecto dilatado e cístico, atuando como um divertículo da uretra prostática. O aumento do utrículo está associado a hipospádia, criptorquidia, distúrbios da diferenciação sexual e agenesia renal unilateral.29,30,31,32,33,34 Séries de casos indicam que a dilatação dos UP ocorre em 10-33% dos pacientes com hipospádia proximal, com a incidência aumentando com a gravidade do defeito de hipospádia.30,32 Em uma série de meninos com genitália externa normal e UP sintomático, agenesia renal unilateral foi encontrada em quase um terço dos casos.33
PU pode ser assintomático, causar sintomas relacionados à obstrução ou à estase urinária—infecções do trato urinário recorrentes, epidídimo-orquite, secreção uretral, retenção, formação de cálculos, ou gotejamento pós-miccional—ou pode apresentar-se com dor abdominal, hematúria, ou hematospermia.31,33,35 Raramente, foi relatada transformação maligna.33,34,36
A dilatação do utrículo prostático pode dificultar a cateterização de pacientes do sexo masculino com hipospádia—a cateterização difícil pode ser o sintoma de apresentação ou o único sintoma de utrículo prostático nesses pacientes. Utrículos volumosos podem ser palpáveis inferiormente ou posteriormente à bexiga ao exame digital retal.31,33,35,37
O diagnóstico diferencial dos utrículos prostáticos inclui cisto do ducto mülleriano (não contínuo com a uretra prostática), divertículo vesical, cisto do úraco e cisto da vesícula seminal.33,38
Tabela 4 Sintomas de apresentação de pacientes com utrículos prostáticos
| Autor | N.º de pacientes | ITU | STUI | Retenção | Epididimite/ dor escrotal | Outros |
|---|---|---|---|---|---|---|
| Desautel | 26 | 6 | 1 | 7 | 2 | 1 |
| Liu | 22 | 8 | 7 | 6 | 3 | 6 |
| Dai | 15 | 6 | 2 | 1 | 7 | 4 |
| Jia | 14 | 8 | 1 | 1 | 5 | 3 |
| Babu | 7 | 3 | 2 | 1 | 3 | 1 |
| Total | 84 | 31 (37) | 13 (15) | 16 (19) | 20 (24) | 5 (6) |
Avaliação Radiológica
O diagnóstico é frequentemente estabelecido por uretrografia retrógrada (RUG) ou cistouretrografia miccional (VCUG), pois essas modalidades permitem a visualização do orifício do utrículo em relação à uretra e à bexiga. Entretanto, a ultrassonografia transabdominal é quase tão sensível quanto a RUG e pode ser adequada para detectar utrículos grandes o suficiente para justificar intervenção.30,39 Os utrículos podem não ser identificados nos exames de imagem devido a preenchimento incompleto. Exames de imagem seccionais podem ser realizados e podem auxiliar no planejamento operatório, mas não são necessários para o diagnóstico. Os utrículos podem ser classificados nos exames de imagem da seguinte forma:29
- Grau 0 – restrito ao colículo seminal
- Grau 1 – estende-se até ou abaixo do colo vesical (Imagem 1)
- Grau 2 – estende-se proximalmente ao colo vesical (Imagem 2)
- Grau 3 – abre-se distalmente ao esfíncter uretral externo
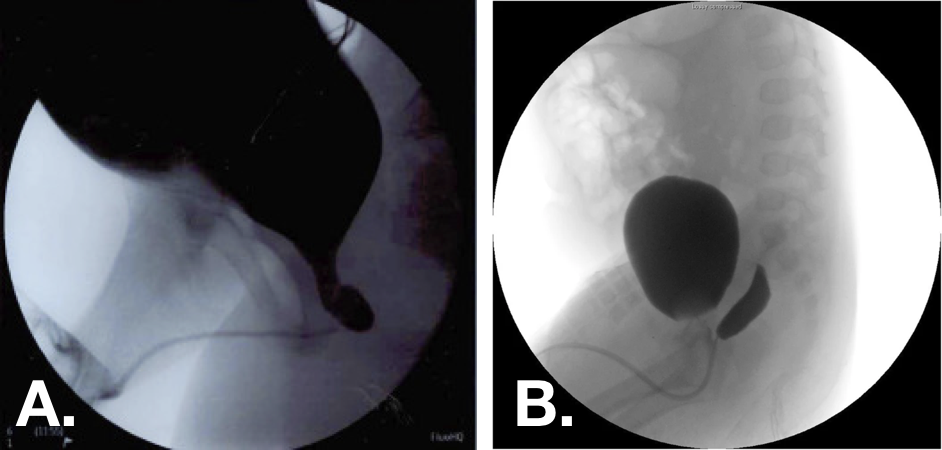
Figura 10 Imagens de cistouretrografia miccional (VCUG) demonstrando utrículos prostáticos grau 1 (esquerda) e grau 2 (direita).30,33
Manejo
O manejo conservador bem-sucedido de utrículos sintomáticos não foi bem descrito e, em geral, exigiria profilaxia antibiótica frequente ou de longo prazo para infecções recorrentes.33 Uma variedade de abordagens cirúrgicas bem-sucedidas foi descrita, com a excisão completa do utrículo oferecendo manejo definitivo. Para utrículos com orifícios muito pequenos, foram relatadas aspiração guiada por ultrassonografia transretal e cateterização e aspiração transuretrais, incisão do orifício e fulguração.31,33,35,40 Essas abordagens são minimamente invasivas, mas conferem maior incidência de recorrência de sintomas do que a excisão. Abordagens abertas, laparoscópicas e assistidas por robô para excisão foram todas descritas, e não há consenso quanto à abordagem ideal. A abordagem robótica pode ser vantajosa devido à localização difícil do utrículo. Os utrículos podem ser acessados por abordagens extraperitoneais, extravesicais, transvesicais, sagital posterior ou perineal.31,33,35,37,41 A cistoscopia concomitante pode ajudar a definir os limites da dissecção em procedimentos laparoscópicos.37,41 As conversões relatadas de abordagens laparoscópicas para abertas na excisão do utrículo são incomuns.
Os princípios fundamentais da excisão, independentemente da abordagem, são dissecção adequada, excisão e fechamento estanque do óstio do utrículo e a prevenção de lesão aos nervos pélvicos, aos ductos deferentes, às vesículas seminais, aos colículos seminais e ao reto.
Tabela 5 Abordagens cirúrgicas para excisão do utrículo prostático e recorrência pós-operatória dos sintomas de apresentação
| Abordagem | Endoscópica | Endoscópica | Aberta | Aberta | Laparoscópica | Laparoscópica |
|---|---|---|---|---|---|---|
| Autor | Casos | Recorrência | Casos | Recorrência | Casos | Recorrência |
| Liu | 3 | 0 | 11 | 0 | 4 | 0 |
| Desautel | 2 | 1 | 13 | 0 | – | – |
| Dai | 13 | 5 | – | – | 4 | 0 |
| Jia | – | – | 8 | 2 | 6 | 0 |
| Babu | 7 | 2 | 2 | 2 | 1 | 1 |
| Yeung | – | – | – | – | 4 | 0 |
| Bayne | – | – | – | – | 4 | 0 |
| Total | 25 | 8 (32) | 34 | 4 (12) | 23 | 1 (4) |
Pontos-chave
- O utrículo prostático é um pequeno remanescente dos ductos paramesonéfricos que persiste nos homens como uma expansão da uretra prostática.
- O aumento do utrículo está associado a hipospádia, criptorquidia, distúrbios da diferenciação sexual e agenesia renal unilateral.
- Os utrículos prostáticos podem ser assintomáticos, causar sintomas relacionados à obstrução ou estase urinária, ou podem se apresentar com dor abdominal, hematúria ou hematospermia.
- A dilatação do utrículo prostático pode impedir a cateterização de pacientes do sexo masculino com hipospádia.
- Utrículos prostáticos sintomáticos podem ser excisados por via aberta, laparoscópica ou assistida por robô.
Divertículos da Bexiga
Divertículos vesicais são raros na população pediátrica, ocorrendo em 1,7% dos pacientes.42 Os divertículos podem ser congênitos ou adquiridos; os divertículos vesicais congênitos são muito mais comuns na população pediátrica.43
Divertículos Congênitos
Acredita-se que os divertículos congênitos resultem de um defeito no músculo detrusor. Noventa por cento se desenvolvem ao redor da bainha de Waldeyer, que contém o uréter, e são conhecidos como divertículos de Hutch.44 Podem ser classificados como periureterais (divertículo próximo, mas sem envolver o orifício ureteral) ou paraureterais (orifício ureteral envolvido pelo divertículo).44 A distorção da parede vesical e do túnel muscular do uréter predispõe as crianças com esses divertículos ao refluxo vesicoureteral. Divertículos congênitos não associados ao uréter desenvolvem-se na região póstero-lateral da bexiga.43
Divertículos vesicais congênitos estão associados a síndromes genéticas que afetam o desenvolvimento do tecido conjuntivo: síndrome de Ehlers-Danlos tipos IV, V, IX; síndrome de Menkes; síndrome de Williams-Beuren e síndrome alcoólica fetal.41,44,45 Ao contrário dos divertículos adquiridos, os divertículos congênitos são encontrados em bexigas de parede lisa que tipicamente apresentam mecânica miccional normal, são defeitos solitários e não estão associados a aumento do risco de malignidade.43 Os divertículos congênitos ocorrem com mais frequência em indivíduos do sexo masculino e são geralmente maiores do que os adquiridos por disfunção vesical neurogênica.43
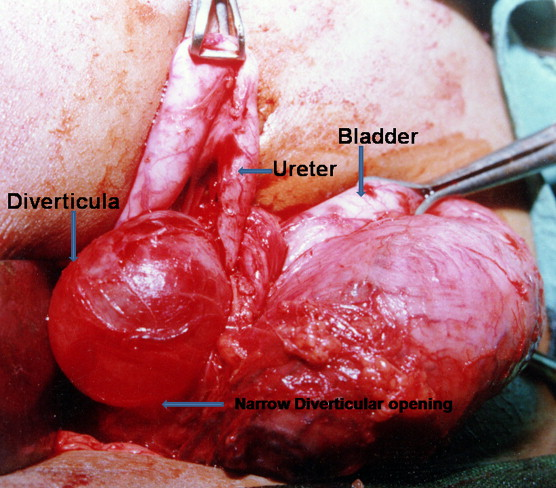
Figura 11 Demonstração intraoperatória de grande divertículo vesical paraureteral, com dilatação proximal e deslocamento medial do uréter.46
Divertículos Adquiridos
Divertículos adquiridos ou secundários se desenvolvem devido a patologias neurogênicas ou obstrutivas no trato urinário inferior. Embora sejam o tipo mais comum de divertículo em adultos, os divertículos secundários são raros em pacientes pediátricos e indicam disfunção grave do trato urinário inferior.43 Eles são mais frequentemente encontrados próximos à cúpula vesical, podem ser múltiplos e são mais comumente observados em uma bexiga trabeculada. Divertículos adquiridos, em geral, estão associados à bexiga neurogênica e à obstrução da via de saída vesical e, em pacientes pediátricos, podem ser observados em associação com válvulas uretrais posteriores e síndrome de Prune Belly.42,43
Apresentação Clínica
Divertículos vesicais em crianças geralmente se manifestam com ITU secundária à estase urinária dentro do divertículo ou são descobertos durante a investigação de hidronefrose ou ITU febril e refluxo vesicoureteral suspeito.44,46 Anomalias do trato urinário superior podem ser encontradas em 16-30% dos casos.44 Os sintomas de apresentação menos comuns incluem sintomas miccionais irritativos, hematúria, distensão e dor abdominal, enurese, cálculos vesicais e obstrução da via de saída devido à distorção do colo vesical.46,47,48 Raramente, divertículos vesicais podem se apresentar com obstrução ureteral completa ou perfuração intra- ou extraperitoneal.49,50,51,52,53,54
A maioria dos pacientes com divertículos vesicais congênitos é assintomática, e—embora muito menos frequentemente do que com os divertículos adquiridos—pode ser diagnosticada incidentalmente em adultos.
Dada a associação com várias condições genéticas, a frequência de anomalias do trato urinário superior e a possibilidade de doença neurogênica ou obstrutiva subjacente, foi proposto por alguns que todas as crianças com divertículos vesicais sejam testadas quanto a anomalias cromossômicas.42 A testagem genética de rotina não é atualmente recomendada para pacientes com divertículo vesical congênito único.44
Tabela 6 Sintomas de apresentação e achados associados em crianças com divertículos vesicais
| Autores | Nº Pacientes | ITU | STUI | Retenção | Outros | RVU | Hidronefrose |
|---|---|---|---|---|---|---|---|
| Alexander | 47 | 26 | 2 | 0 | 2 | 32 | 19 |
| Evangilidis | 21 | 19 | 12 | 0 | – | – | – |
| Marte | 16 | 15 | 9 | 0 | 6 | 7 | – |
| Bhat | 12 | 5 | – | 12 | 4 | 9 | 5 |
| Macedo | 10 | 5 | 3 | 1 | – | – | 4 |
| Total | 106 | 70 (66) | 26 (25) | 13 (12) | 12 (11) | 48 (45) | 28 (26) |
Avaliação Radiológica
Divertículos vesicais são frequentemente detectados inicialmente na ultrassonografia vesical como um cisto hipoecoico que se projeta da parede póstero-lateral da bexiga. O diagnóstico diferencial para divertículo suspeito na imagem inclui cisto uracal, ureterocele evertida, ureter ectópico, herniação vesical em hérnia inguinal e duplicação vesical.44 A cistouretrografia miccional com incidências laterais e oblíquas é o padrão-ouro para estabelecer o diagnóstico de divertículo vesical e permite a avaliação simultânea para RVU.43 Particularmente nos casos sem RVU, pielograma intravenoso ou urografia por TC ou por RM podem ser utilizados para esclarecer a relação anatômica entre o divertículo e o ureter.44 Os tratos urinários superiores de crianças com divertículos vesicais devem ser avaliados quanto a hidroureteronefrose.
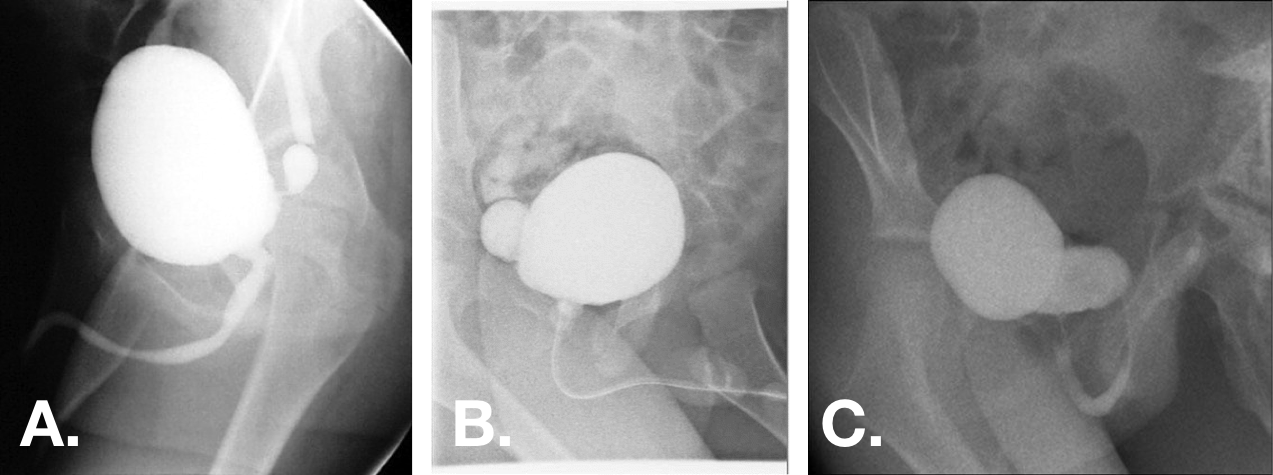
Figura 12 Divertículos vesicais congênitos observados nas projeções oblíquuas da VCUG. Os divertículos podem variar desde pequenos, com colos muito estreitos, até maiores do que a bexiga, com aberturas de base ampla.44,47,55
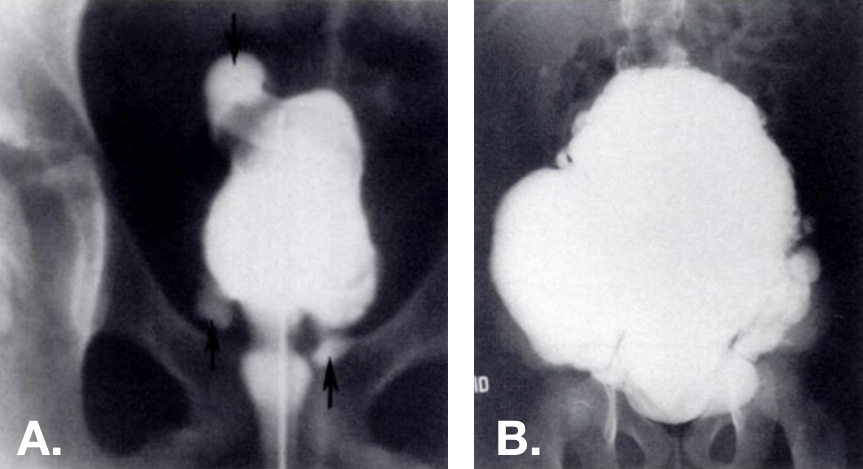
Figura 13 Divertículos adquiridos vistos na cistouretrografia miccional (VCUG). Múltiplos divertículos presentes em vários locais—incluindo na cúpula vesical—em bexigas trabeculadas. A foto à direita é de um paciente com síndrome de Ehlers-Danlos.42
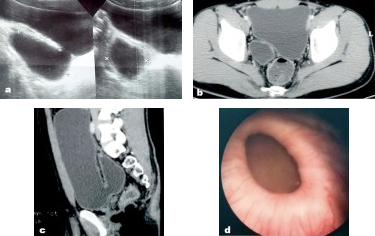
Figura 14 Divertículo vesical congênito como visto na ultrassonografia, tomografia computadorizada pélvica e cistoscopia.56
Estudos urodinâmicos podem ser considerados em pacientes com divertículos vesicais se houver suspeita de disfunção subjacente do trato urinário inferior ou se a presença de um divertículo não explicar adequadamente os sintomas apresentados pelo paciente. Hiperatividade do detrusor pode ser observada em cerca de metade desses pacientes, possivelmente secundária à hipotonia dentro e ao redor do divertículo e à estase urinária crônica.44,48 Os divertículos complicam a interpretação do cistometrograma, pois reduzem a pressão intravesical. A estase de urina no interior do divertículo e o RVU associado também podem complicar os resultados.44
A avaliação endoscópica pode ajudar a esclarecer a anatomia e é recomendada durante a intervenção cirúrgica, mas não é necessária para o diagnóstico de divertículo vesical.
Indicações para correção
Embora os divertículos vesicais não se resolvam sem intervenção, nem todos os pacientes necessitam de cirurgia. O tratamento adequado da infecção do trato urinário (ITU) e da disfunção miccional pode levar à resolução dos sintomas em pacientes com divertículos pequenos e não complicados.44 Para aqueles com sintomas refratários ou divertículos complicados, a excisão cirúrgica é geralmente curativa, e o reimplante ureteral pode ser realizado simultaneamente, quando necessário. As indicações geralmente aceitas para cirurgia incluem as seguintes:43,44
- Sintomas miccionais refratários ao manejo clínico
- Divertículo > 3cm
- ITU frequentes
- RVU complicado
- Retenção urinária
- Obstrução ou deterioração do trato urinário superior
- Cálculos vesicais
- Ruptura de divertículo
Ao contrário do que ocorre em adultos, a degeneração maligna do epitélio associada a divertículos vesicais pediátricos geralmente não é observada; no entanto, qualquer suspeita de malignidade exigiria intervenção cirúrgica.43
Antes de qualquer intervenção cirúrgica, a disfunção miccional deve ser avaliada e tratada para isolar os sintomas relacionados ao divertículo e sua evolução no pós-operatório. Patologias associadas, como subatividade do detrusor, obstrução da via de saída vesical ou disrafismo espinhal, devem ser abordadas antes da diverticulectomia.
A intervenção cirúrgica deve ser cuidadosamente considerada em pacientes com distúrbios subjacentes do tecido conjuntivo, pois a recorrência é provável nessa população.44,53
Abordagens operatórias
A diverticulectomia vesical pode ser realizada endoscopicamente ou por abordagens trans- ou extravesicais, abertas ou minimamente invasivas. As abordagens transvesical e extravesical reproduzem aquelas utilizadas para o reimplante ureteral, e o reimplante pode ser realizado como parte do procedimento se necessário devido à localização do divertículo ou se indicado devido ao RVU associado grave. Recomenda-se a avaliação cistoscópica da abertura diverticular em relação ao óstio ureteral como parte do planejamento operatório. Várias séries de casos relataram resolução completa dos sintomas—ou redução acentuada dos sintomas de disfunção miccional—bem como do RVU após a excisão do divertículo, independentemente da abordagem.57,55 A oclusão da abertura diverticular parauréterica por meio de injeção subureteral de agentes preenchedores foi relatada, embora os dados sobre a eficácia a longo prazo sejam limitados.58,59 Uma comparação entre as abordagens endoscópica, intra- e extravesical demonstrou resolução bem-sucedida do RVU em 79%, 91% e 80% dos casos, respectivamente.59
Considerações cirúrgicas importantes, independentemente da abordagem, são a profilaxia antimicrobiana perioperatória orientada por cultura, a prevenção de lesão ou desvascularização do ureter ou do ducto deferente, a prevenção de lesão retal no reparo de grandes divertículos posteriores e a descompressão vesical no pós-operatório.44
Pontos-chave
- Os divertículos vesicais em crianças podem ser congênitos—devido a defeito no músculo detrusor, mais frequentemente ao redor da bainha de Waldeyer (divertículos de Hutch)—ou adquiridos devido a patologia neurogênica ou obstrutiva do trato urinário inferior.
- A distorção da parede vesical e do túnel muscular do ureter pelos divertículos de Hutch predispõe as crianças ao refluxo vesicoureteral.
- Os divertículos vesicais em crianças com mais frequência se apresentam com ITU secundária à estase urinária dentro do divertículo ou são descobertos durante a investigação de hidronefrose ou ITU febril e suspeita de refluxo vesicoureteral. A maioria dos divertículos é assintomática.
- A cistouretrografia miccional com incidências laterais e oblíquas é o padrão-ouro para estabelecer o diagnóstico de divertículo vesical e permite avaliação simultânea para RVU.
- Para aqueles com sintomas refratários ou divertículos complicados, a excisão cirúrgica pode ser realizada por via transvesical ou extravesical. O reimplante ureteral pode ser realizado simultaneamente, conforme necessário.
- Antes de qualquer intervenção operatória, a disfunção miccional deve ser avaliada e tratada.
Resumo
As anomalias do úraco podem apresentar-se de várias formas e, mais frequentemente, causam drenagem pelo umbigo ou infecção. As anomalias podem ser tratadas de forma conservadora e podem regredir espontaneamente. Cistos infectados podem ser drenados percutaneamente, e qualquer anomalia sintomática pode ser excisada por via aberta ou laparoscópica. O risco de transformação maligna de anomalias do úraco assintomáticas em crianças é desconhecido.
A dilatação do utrículo prostático está associada a hipospádia e a outras anomalias geniturinárias e pode ser assintomática ou causar sintomas relacionados à obstrução da saída vesical e à estase urinária. Os utrículos podem ser diagnosticados por RUG, VCUG ou US. Para pacientes sintomáticos, a excisão pode ser realizada por via endoscópica, aberta ou laparoscópica, com o manejo endoscópico conferindo um risco mais elevado de recorrência.
Os divertículos vesicais pediátricos podem ser congênitos ou adquiridos. Divertículos congênitos decorrem de defeitos do músculo detrusor próximos à bainha ureteral e estão associados ao RVU, bem como a distúrbios do tecido conjuntivo. Os divertículos são frequentemente assintomáticos, mas podem causar infecção, sintomas miccionais irritativos, retenção urinária ou dor abdominal e estão frequentemente associados à hiperatividade do detrusor. Divertículos adquiridos são secundários à obstrução do trato urinário inferior ou à bexiga neurogênica. As indicações para correção cirúrgica incluem sintomas refratários ou graves, tamanho >3cm e disfunção do trato urinário associada. A intervenção deve ser precedida pelo manejo de qualquer outra patologia do trato urinário inferior. Não há consenso quanto à abordagem operatória, e o reimplante ureteral é frequentemente realizado simultaneamente.
Referências
- Jm P. Embryology of the genitourinary tract. Elsevier: Philadelphia; 2016, DOI: 10.1201/b13795-15.
- MacNeily AE, Koleilat N, Kiruluta HG, Homsy YL. Urachal abscesses: Protean manifestations, their recognition, and management. Urology 1992; 40 (6): 530–535. DOI: 10.1016/0090-4295(92)90409-p.
- Naiditch JA, Radhakrishnan J, Chin AC. Current diagnosis and management of urachal remnants. J Pediatr Surg 2013; 48 (10): 2148–2152. DOI: 10.1016/j.jpedsurg.2013.02.069.
- Fox JA, M.S. R, JC G, CF A, RA H, JC V, et al.. Vesicoureteral reflux in children with urachal anomalies. J Pediatr Urol 2011; 7 (6): 632–635. DOI: 10.1016/j.jpurol.2011.04.001.
- Ashley RA, Inman BA, Routh JC, Rohlinger AL, Husmann DA, Kramer SA. Urachal Anomalies: A Longitudinal Study of Urachal Remnants in Children and Adults. J Urol 2007; 178 (4s): 1615–1618. DOI: 10.1016/j.juro.2007.03.194.
- Cilento BG, Bauer SB, Retik AB, Peters CA, Atala A. Urachal anomalies: defining the best diagnostic modality. Urology 1998; 52 (1): 120–122. DOI: 10.1016/s0090-4295(98)00161-7.
- Rich RH, Hardy BE, Filler RM. Surgery for Anomalies of the Urachus. J Urol 1983; 131 (3): 616–616. DOI: 10.1016/s0022-5347(17)50535-2.
- Yiee JH, Garcia N, Baker LA, Barber R, Snodgrass WT, Wilcox DT. A diagnostic algorithm for urachal anomalies. J Pediatr Urol 2007; 3 (6): 500–504. DOI: 10.1016/j.jpurol.2007.07.010.
- Copp HL, Wong IY, Krishnan C, Malhotra S, Kennedy WA. Clinical Presentation and Urachal Remnant Pathology: Implications for Treatment. J Urol 2009; 182 (4s): 1921–1924. DOI: 10.1016/j.juro.2009.03.026.
- Cuda SP, Vanasupa BP, Sutherland RS. Nonoperative management of a patent urachus. Urology 2005; 66 (6): 1320.e7–1320.e9. DOI: 10.1016/j.urology.2005.06.121.
- Gleason JM, Bowlin PR, Bagli DJ, Lorenzo AJ, Hassouna T, Koyle MA, et al.. A Comprehensive Review of Pediatric Urachal Anomalies and Predictive Analysis for Adult Urachal Adenocarcinoma. J Urol 2015; 193 (2): 632–636. DOI: 10.1016/j.juro.2014.09.004.
- Stopak JK, Azarow KS, Abdessalam SF, Raynor SC, Perry DA, Cusick RA. Trends in surgical management of urachal anomalies. J Pediatr Surg 2015; 50 (8): 1334–1337. DOI: 10.1016/j.jpedsurg.2015.04.020.
- Dethlefs CR, Abdessalam SF, Raynor SC, Perry DA, Allbery SM, Lyden ER, et al.. Conservative management of urachal anomalies. J Pediatr Surg 2019; 54 (5): 1054–1058. DOI: 10.1016/j.jpedsurg.2019.01.039.
- Sheldon CA, Clayman RV, Gonzalez R, Williams RD, Fraley EE. Malignant Urachal Lesions. J Urol 1984; 131 (1): 1–8. DOI: 10.1016/s0022-5347(17)50167-6.
- Oestreich AE. Urachal anomalies in children: the vanishing relevance of the preoperative voiding cystourethrogram. Yearbook of Diagnostic Radiology 2005; 2007 (12): 174–175. DOI: 10.1016/s0098-1672(08)70123-1.
- Lewis JB, Morse JW, Eyolfson MF, Schwartz SL. Spontaneous Rupture of a Vesicourachal Diverticulum Manifesting as Acute Abdominal Pain. Acad Emerg Med 1996; 3 (12): 1140–1143. DOI: 10.1111/j.1553-2712.1996.tb03375.x.
- OGBEVOEN JUSTINO, JAFFE DAVIDM, LANGER JACOBC. Intraperitoneal rupture of an infected urachal cyst: A rare cause of peritonitis in children. Pediatr Emerg Care 1996; 12 (1): 41–43. DOI: 10.1097/00006565-199602000-00012.
- GONZ??LEZ RICARDO, De FILIPPO ROGER, JEDNAK ROMAN, BARTHOLD JULIASPENCER. Urethral Atresia: Long-term Outcome In 6 Children Who Survived The Neonatal Period. J Urol 2001: 2241–2244. DOI: 10.1097/00005392-200106001-00006.
- Mesrobian H-GO, Zacharias A, Balcom AH, Cohen RD. Ten Years of Experience With Isolated Urachal Anomalies in Children. J Urol 1997; 158: 1316–1318. DOI: 10.1097/00005392-199709000-00173.
- McCollum MO, MacNeily AE, Blair GK. Surgical implications of urachal remnants: Presentation and management. J Pediatr Surg 2003; 38 (5): 798–803. DOI: 10.1016/jpsu.2003.50170.
- Minevich E, W.J. L, A.G.. The infected urachal cyst: Primary excision versus a staged approach. J Pediatr Surg 1997; 33 (1): 147. DOI: 10.1016/s0022-3468(98)90408-0.
- Newman BM, Karp MP, Jewett TC, Cooney DR. Advances in the management of infected urachal cysts. J Pediatr Surg 1986; 21 (12): 1051–1054. DOI: 10.1016/0022-3468(86)90006-0.
- Yoo KH, Lee S-J, Chang S-G. Treatment of Infected Urachal Cysts. Yonsei Med J 2006; 47 (3): 423. DOI: 10.3349/ymj.2006.47.3.423.
- Galati VG, D.B. R, F.. Management of Urachal Remnants in Early Childhood. Yearbook of Diagnostic Radiology 2008; 2009: 154–155. DOI: 10.1016/s0098-1672(09)79312-9.
- Nogueras-Ocaña M, Rodríguez-Belmonte R, Uberos-Fernández J, Jiménez-Pacheco A, Merino-Salas S, Zuluaga-Gómez A. Urachal anomalies in children: Surgical or conservative treatment? J Pediatr Urol 2014; 10 (3): 522–526. DOI: 10.1016/j.jpurol.2013.11.010.
- Lipskar AM, Glick RD, Rosen NG, Layliev J, Hong AR, Dolgin SE, et al.. Nonoperative management of symptomatic urachal anomalies. J Pediatr Surg 2010; 45 (5): 1016–1019. DOI: 10.1016/j.jpedsurg.2010.02.031.
- Stone NM, Garden RJ, Webert H. Laparoscopic excision of a urachal cyst. Urology 1995; 45 (1): 161–164. DOI: 10.1016/s0090-4295(95)97824-0.
- OKEGAWA TAKATSUGU, ODAGANE AKIHIRO, NUTAHARA KIKUO, HIGASHIHARA EIJI. Laparoscopic management of urachal remnants in adulthood. Int J Urol 2005; 13 (12): 1466–1469. DOI: 10.1111/j.1442-2042.2006.01613.x.
- Yamzon J, K.P. DF, RE C, AY H, BE K, C.J.. V1710 Pediatric Robotic-assisted Laparoscopic Urachal Cyst And Bladder Cuff Excision. J Urol 2008; 185 (4s): 2385–2388. DOI: 10.1016/j.juro.2011.02.2035.
- Patrzyk M, Glitsch A, Schreiber A, Bernstorff W von, Heidecke C-D. Single-incision laparoscopic surgery as an option for the laparoscopic resection of an urachal fistula: first description of the surgical technique. Surg Endosc 2010; 24 (9): 2339–2342. DOI: 10.1007/s00464-010-0922-4.
- IKOMA F, SHIMA H, YABUMOTO H. Classification of Enlarged Prostatic Utricle in Patients with Hypospadias. Br J Urol 1985; 57 (3): 334–337. DOI: 10.1111/j.1464-410x.1985.tb06356.x.
- Aktuğ T, Ekberli\. G. Re: The prostatic utricle: an under-recognized condition resulting in significant morbidity in boys with both hypospadias and normal external genitalia. J Pediatr Urol 2017; 15 (4): 425. DOI: 10.1016/j.jpurol.2019.03.030.
- Dai L-N, He R, Wu S-F, Zhao H-T, Sun J. Surgical treatment for prostatic utricle cyst in children: A single-center report of 15 patients. Int J Urol 2021; 28 (6): 689–694. DOI: 10.1111/iju.14543.
- Devine CJ, Gonzalez-Serva L, Stecker JF, Devine PC, Horton CE. Utricular Configuration in Hypospadias and Intersex. J Urol 1980; 123 (3): 407–411. DOI: 10.1016/s0022-5347(17)55959-5.
- Liu B, He D, Zhang D, Liu X, Lin T, Wei G. Prostatic utricles without external genital anomalies in children: our experience, literature review, and pooling analysis. BMC Urol 2019; 19 (1): 21. DOI: 10.1186/s12894-019-0450-z.
- Farikullah J, Ehtisham S, Nappo S, Patel L, Hennayake S. Persistent Müllerian duct syndrome: lessons learned from managing a series of eight patients over a 10-year period and review of literature regarding malignant risk from the Müllerian remnants. BJU Int 2012; 110 (11c): E1084–e1089. DOI: 10.1111/j.1464-410x.2012.11184.x.
- Desautel MG, Stock J, Hanna MK, REMNANTS MULLERIANDUCT. Müllerian Duct Remnants. J Urol 1999; 162 (3, Part 2): 1014. DOI: 10.1097/00005392-199909000-00013.
- Jia W, L.G. Z, L W, Y F, W H, J X, et al.. Comparison of laparoscopic excision versus open transvesical excision for symptomatic prostatic utricle in children J Pediatr Surg. 2016; 51 (10): 1597–1601. DOI: 10.1016/j.jpedsurg.2016.06.004.
- Babu R, Chandrasekharam VVS. Cystoscopic Management of Prostatic Utricles. Urology 2021; 149: e52–e55. DOI: 10.1016/j.urology.2020.09.005.
- Nallabothula AK, Yatam LS, Ayapati T, Bodagala VD. Large Prostatic Utricle With Transitional Cell Carcinoma in an Adult. Urology 2017; 102: e5–e6. DOI: 10.1016/j.urology.2016.12.048.
- Yeung CK, Sihoe JDY, Tam YH, Lee KH. Laparoscopic excision of prostatic utricles in children. BJU Int 2001; 87 (6): 505–508. DOI: 10.1046/j.1464-410x.2001.00132.x.
- Johnson D, Parikh K, Schey W, Mar W. MRI in Diagnosis of a Giant Prostatic Utricle. Case Rep Radiol 2014; 2014: 1–3. DOI: 10.1155/2014/217563.
- Kojima Y, Hayashi Y, Maruyama T, Sasaki S, Kohri K. Comparison between ultrasonography and retrograde urethrography for detection of prostatic utricle associated with hypospadias. Urology 2001; 57 (6): 1151–1155. DOI: 10.1016/s0090-4295(01)00954-2.
- Bayne AP, Austin JC, Seideman CA. Robotic assisted retrovesical approach to prostatic utricle excision and other complex pelvic pathology in children is safe and feasible. J Pediatr Urol 2021; 17 (5): 710–715. DOI: 10.1016/j.jpurol.2021.08.004.
- Blane CE, Zerin JM, Bloom DA. Bladder diverticula in children. Radiology 1994; 190 (3): 695–697. DOI: 10.1148/radiology.190.3.8115613.
- Es R. Bladder and female urethral diverticula. Elsevier: Philadelphia; 2017, DOI: 10.1016/b978-1-4160-6911-9.00078-5.
- Psutka SP, Cendron M. Bladder diverticula in children. J Pediatr Urol 2013; 9 (2): 129–138. DOI: 10.1016/j.jpurol.2012.02.013.
- Sammour Z, Gomes C, Bessa J, Pinheiro M, Kim C, Sacomani C, et al.. 1622 Congenital Genitourinary Abnormalities In Children With Williams-beuren Syndrome. J Urol 2014; 187 (4s): 804–809. DOI: 10.1016/j.juro.2012.02.1417.
- Bhat A, Bothra R, Bhat MP, Chaudhary GR, Saran RK, Saxena G. Congenital bladder diverticulum presenting as bladder outlet obstruction in infants and children. J Pediatr Urol 2012; 8 (4): 348–353. DOI: 10.1016/j.jpurol.2011.07.001.
- Alexander RE, Kum JB, Idrees M. Bladder Diverticulum: Clinicopathologic Spectrum in Pediatric Patients. Pediatr Dev Pathol 2012; 15 (4): 281–285. DOI: 10.2350/12-02-1154-oa.1.
- Evangelidis A, Castle EP, Ostlie DJ, Snyder CL, Gatti JM, Murphy JP. Surgical management of primary bladder diverticula in children. J Pediatr Surg 2005; 40 (4): 701–703. DOI: 10.1016/j.jpedsurg.2005.01.003.
- Marte A, Cavaiuolo S, Esposito M, Pintozzi L. Vesicoscopic Treatment of Symptomatic Congenital Bladder Diverticula in Children: A 7-Year Experience. Eur J Pediatr Surg 2015; 26 (03): 240–244. DOI: 10.1055/s-0035-1551564.
- Cruz ML da, Macedo A, Garrone G, Ottoni SL, Oliveira DE, Rosário Souza GRM do. Primary congenital bladder diverticula: Where does the ureter drain? Afr J Paediatr Surg 2015; 12 (4): 280. DOI: 10.4103/0189-6725.172574.
- Livne PM, Gonzales ET. Congenital bladder diverticula causing ureteral obstruction. Urology 1985; 25 (3): 273–276. DOI: 10.1016/0090-4295(85)90327-9.
- Stein RJ, Matoka DJ, Noh PH, Docimo SG. Spontaneous perforation of congenital bladder diverticulum. Urology 2005; 66 (4): 881.e5–881.e6. DOI: 10.1016/j.urology.2005.04.004.
- Britto MM, Yao HHI, Campbell N. Delayed diagnosis of spontaneous rupture of a congenital bladder diverticulum as a rare cause of an acute abdomen. ANZ J Surg 2019; 89 (9): 385–387. DOI: 10.1111/ans.14559.
- Jorion JL, Michel M. Spontaneous rupture of bladder diverticula in a girl with Ehlers-Danlos syndrome. J Pediatr Surg 1999; 34 (3): 483–484. DOI: 10.1016/s0022-3468(99)90506-7.
- Temiz A, Akcora B, Atik E. An atypical bladder diverticulum presented with recurrent peritonitis: case report. Ulus Travma Acil Cerrahi Derg 2011; 17 (4): 365–367. DOI: 10.5505/tjtes.2011.81542.
- Christman MS, Casale P. Robot-Assisted Bladder Diverticulectomy in the Pediatric Population. J Endourol 2012; 26 (10): 1296–1300. DOI: 10.1089/end.2012.0051.
- Nerli RB, Ghagane SC, Musale A, Deole S, Hiremath MB, Dixit NS, et al.. Congenital bladder diverticulum in a child: Surgical steps of extra-vesical excision. Urol Case Rep 2019; 22: 42–43. DOI: 10.1016/j.eucr.2018.10.009.
- Cerwinka WH, Scherz HC, Kirsch AJ. Endoscopic Treatment of Vesicoureteral Reflux with Dextranomer/Hyaluronic Acid in Children. Adv Urol 2008; 2008: 1–7. DOI: 10.1155/2008/513854.
- Aydogdu O, Burgu B, Soygur T. Predictors of Surgical Outcome in Children With Vesicoureteral Reflux Associated With Paraureteral Diverticula. Urology 2010; 76 (1): 209–214. DOI: 10.1016/j.urology.2009.11.052.
Ultima atualização: 2025-09-21 13:35