2: Imagenología del tracto urinario
Este capítulo durará aproximadamente 24 minutos para leer.
Introducción
La obtención de imágenes diagnósticas es una herramienta poderosa para comprender la historia natural de muchas afecciones que afectan el tracto genitourinario (GU), tanto desde una perspectiva anatómica como funcional. Con un uso óptimo, las imágenes diagnósticas mejoran los resultados del paciente mientras minimizan el riesgo para el paciente. Existen múltiples modalidades disponibles para obtener imágenes detalladas del cuerpo humano, incluidas la ecografía (US), la resonancia magnética (MRI), la tomografía computarizada (CT), la centellografía nuclear y la fluoroscopia/radiografía. Los avances tecnológicos en estas modalidades son continuos.
En el ámbito pediátrico, las indicaciones para la obtención de imágenes diagnósticas dependen no solo de la presentación clínica, sino también de la edad (o nivel de desarrollo) del paciente. Puede requerirse más de una técnica de imagen para evaluar completamente la anatomía y fisiología del tracto genitourinario (GU). Si bien la presentación clínica es primordial y a menudo permitirá un enfoque algorítmico de la obtención de imágenes, el mejor enfoque en un adolescente puede diferir del de un bebé o un niño pequeño.
Consideraciones sobre la radiación
En los últimos veinte a treinta años, tanto los clínicos como el público en general se han mostrado más preocupados por la exposición a la radiación derivada de las imágenes médicas y sus posibles peligros, especialmente en pacientes pediátricos. El riesgo de cánceres futuros puede ser mayor en los niños por varios factores. En primer lugar, los niños pequeños son más pequeños que los adultos, lo que conlleva más energía cinética por unidad de masa para la misma cantidad de exposición. Los niños también tienen mayores tasas de proliferación celular, lo que incrementa la radiosensibilidad. Por último, los niños tienen una mayor esperanza de vida desde el momento de la exposición a la radiación, lo que permite que las mutaciones cromosómicas inducidas por la radiación se vuelvan clínicamente relevantes.1
En muchos casos, la pregunta clínica puede responderse sin el uso de radiación ionizante. No obstante, se debe tener cuidado en sopesar los riesgos y beneficios de un procedimiento. En casos de traumatismo, el estándar de oro sigue siendo una TC de abdomen y pelvis con contraste.2 Debe tranquilizar al médico solicitante saber que veinte años de avances en la tecnología de TC han permitido dosis entre diez y cien (o más) veces inferiores a las de finales del milenio.3 Por otro lado, muchos clínicos no son conscientes de que una sola radiografía abdominal (KUB) no ahorra mucha radiación en comparación con una TC moderna del abdomen.4
Si bien la RM puede parecer preferible a la TC desde el punto de vista de la radiación, la sedación para la RM sigue siendo necesaria en la mayoría de los niños menores de aproximadamente 8 años. Los riesgos de la sedación (especialmente la sedación repetida) para los niños aún no están claros. Tiempos de escaneo más rápidos, interfaces de realidad virtual y sólidos programas de vida infantil ofrecen promesas para minimizar las necesidades de sedación.5
Las preguntas que deben plantearse al elegir una investigación son:
- ¿Responderá esta investigación a la pregunta clínica?
- ¿Existe una investigación previa que responda a la pregunta clínica actual?
- ¿Hay una alternativa más segura (considere la radiación o la necesidad de sedación)?
- ¿Cuál es el nivel de desarrollo de mi paciente?
Si existe alguna duda, es aconsejable discutirlo con su radiólogo.
Contraste en la obtención de imágenes del tracto urinario
Agentes de contraste yodados
Se utiliza contraste yodado para la TC y la cistouretrografía miccional (VCUG). Al solicitar un procedimiento que implique contraste, el clínico debe conocer los riesgos potenciales. El primero es el riesgo de una reacción alérgica o similar a alérgica grave. Estos eventos adversos son muy poco probables con los agentes de contraste modernos. Se estima que la tasa de reacciones de tipo alérgico es del 0,6 %, de las cuales la mayoría son leves y autolimitadas. Para las reacciones graves, la estimación es del 0,04 %. Los antecedentes de una reacción previa de tipo alérgico al contraste yodado son el factor de riesgo más claro e importante para una reacción posterior. El riesgo en pacientes con tales antecedentes debe sopesarse cuidadosamente frente al beneficio de una tomografía computarizada posterior. La evidencia sigue siendo inconclusa en cuanto a los beneficios de la premedicación.6
The second risk that the ordering clinician should consider is the risk of contrast-induced acute kidney injury (CI-AKI), defined as a sudden deterioration in renal function that is caused by the intravascular administration of iodinated contrast. Unfortunately, the true risk of this entity is unknown even in adults. As is the case with allergic type reactions, there is one risk factor that is generally accepted, in best practice, to predispose a patient to CI-AKI; this risk factor is pre-existing renal insufficiency. No clear threshold of eGFR has been established (above which risk can be ignored). However, the American College of Radiology (ACR) recommends an eGFR cutoff of 30 mL/min/1.73 m.26
El cálculo de la TFGe en niños requiere conocer la estatura del paciente y la creatinina sérica. La TFGe aproximada se obtiene entonces mediante la ecuación de Schwartz a pie de cama:6
TFG (mL/min/1.73 m2) = 0.41 × talla / creatinina sérica
Existen fórmulas más recientes que proporcionan estimaciones más precisas de la función renal basadas en la creatinina sérica o la cistatina C, incluidas las ecuaciones CKiD-U25, que están ajustadas por edad, sexo y talla.7 Hay una calculadora disponible aquí. Es importante señalar que los pacientes con enfermedad renal crónica dependientes de diálisis pueden recibir contraste yodado sin riesgo de LRA, ya que tienen riñones sin función.
Agentes de contraste en RM
Para la RM con contraste se utiliza un quelato de gadolinio. Las reacciones alérgicas a estos agentes son aún menos probables que con el contraste yodado, con una tasa aproximada del 0,05%. El manejo de las reacciones alérgicas al gadolinio es el mismo que para los agentes de contraste yodados, y esto se tratará más adelante. Un riesgo exclusivo de los agentes de gadolinio es la fibrosis sistémica nefrogénica (FSN), una enfermedad que se asemeja a la esclerodermia y que puede conducir a contracturas articulares e insuficiencia orgánica y, en ocasiones, puede ser mortal.6 Esta entidad aún se comprende poco, pero, afortunadamente, es bastante rara. Los pacientes con lesión renal aguda, enfermedad renal crónica en estadio 4 o 5, o aquellos en diálisis pueden tener hasta un 7% de riesgo de desarrollar FSN con ciertos agentes de gadolinio (tipos I o posiblemente III). El uso de gadolinio en estos pacientes puede seguir siendo apropiado en casos poco frecuentes en los que el beneficio potencial de una RM con contraste sea muy alto. Sin embargo, en esta circunstancia es una buena práctica obtener un consentimiento informado. Siempre que sea posible, debería utilizarse en esta población un agente de gadolinio de menor riesgo (grupo II).6
Agentes de contraste ecográficos
Los agentes de contraste ecográfico consisten en una microesfera de fosfolípidos o proteínas que contiene un gas inerte ecogénico. Estos agentes mejoran transitoriamente la resolución de contraste ecográfica y están aprobados para uso intravenoso y endoluminal. Se requiere software ecográfico dedicado. Los agentes de contraste ecográfico son muy seguros, con reacciones graves que ocurren en aproximadamente el 0,01% de los casos. No se conoce toxicidad renal a las dosis aprobadas.6
Manejo de reacciones de tipo alérgico
Las recomendaciones actuales del Manual sobre Medios de Contraste del Colegio Americano de Radiología para el manejo de las reacciones alérgicas aconsejan la administración inmediata de epinefrina en presencia de lo siguiente:
- Broncoespasmo grave
- Eritema difuso con hipotensión profunda
- Edema laríngeo
- Hipotensión con taquicardia
El régimen posológico actual se resume en la tabla a continuación (Tabla 1).8
Tabla 1 Dosis de epinefrina para reacciones al contraste.
| Vía | Dosis | Notas | Repetir | Dosis máxima |
|---|---|---|---|---|
| intravenosa | 0.01 mg/kg | lentamente en una infusión IV de líquidos en curso | Cada 5-15 minutos | 1 mg |
| intramuscular | 0.01 mg/kg | Cada 5-15 minutos | 1 mg | |
| Autoinyector 15–30 kg | 0.15 mg | Use IV (arriba) si pesa menos de 15 kg | ||
| Autoinyector > 30 kg | 0.3 mg |
Criterios de adecuación del ACR
Los criterios de adecuación del Colegio Estadounidense de Radiología (ACR) son recomendaciones de consenso, disponibles gratuitamente en línea para el clínico que busca el estudio de imagen más adecuado en diversas situaciones. Las recomendaciones son elaboradas por paneles de expertos y se actualizan con frecuencia.
Tabla 2 Recomendaciones de ecografía renal según los Criterios de Adecuación del ACR.
| Indicación | Notas |
|---|---|
| Recién nacido con UTD prenatal, ecografía posnatal inicial | Idealmente con al menos 72 horas de vida |
| Recién nacido con UTD prenatal y ecografía posnatal normal | Seguimiento a los 1–6 meses según criterio del clínico |
| Recién nacido con UTD prenatal y grado SFU 1-2 o APRPD menor de 15 mm en ecografía posnatal | Considerar estudio miccional como estudio complementario |
| Recién nacido con UTD prenatal y grado SFU 3-4 o APRPD mayor de 15 mm en ecografía posnatal | También se recomiendan un estudio miccional y un estudio de medicina nuclear del sistema colector |
| Hematuria atraumática | Cuando es macroscópica o está asociada a proteinuria |
| Sospecha de urolitiasis | |
| ITU febril | En niño menor de 2 meses |
| ITU febril recurrente o mala respuesta a antibióticos |
Modalidades de imagen
Ecografía
En los niños, la ecografía (US) es un caballo de batalla. Casi siempre constituye la primera línea de investigación ante sospecha de anomalías renales (el trauma es la excepción notable). La ecografía es económica, no ionizante y está ampliamente disponible. La desventaja es que depende del operador. Especialmente cuando la interpretación es a distancia, el radiólogo depende en gran medida del ecografista.
Para las indicaciones a continuación, se recomienda la ecografía como modalidad de imagen inicial.8,9
Anatomía renal
Las dimensiones renales según la edad están bien establecidas y el crecimiento adecuado puede demostrarse fácilmente mediante ecografía. Existe un margen de error aproximado de más/menos 7 mm según los planos de imagen o el movimiento del paciente (con la TC como patrón de oro), lo que significa que la comparación de varias ecografías previas ofrecerá una indicación más fiable del crecimiento renal (o de su ausencia). Los riñones pueden ser lobulados o presentar columnas de Bertin prominentes como variantes normales.
La diferenciación corticomedular suele ser muy evidente en los neonatos. A medida que los glomérulos corticales involucionan fisiológicamente conforme el niño madura, la corteza normalmente se vuelve más hipoecoica. Así, el riñón normal en un niño mayor o adolescente se asemejará al aspecto del adulto, con corteza y médula casi isoecoicas que contrastan claramente con el seno renal hiperecogénico. La familiaridad con el aspecto normal del riñón a diferentes edades es esencial para el radiólogo pediátrico y permite el diagnóstico de anomalías sutiles.
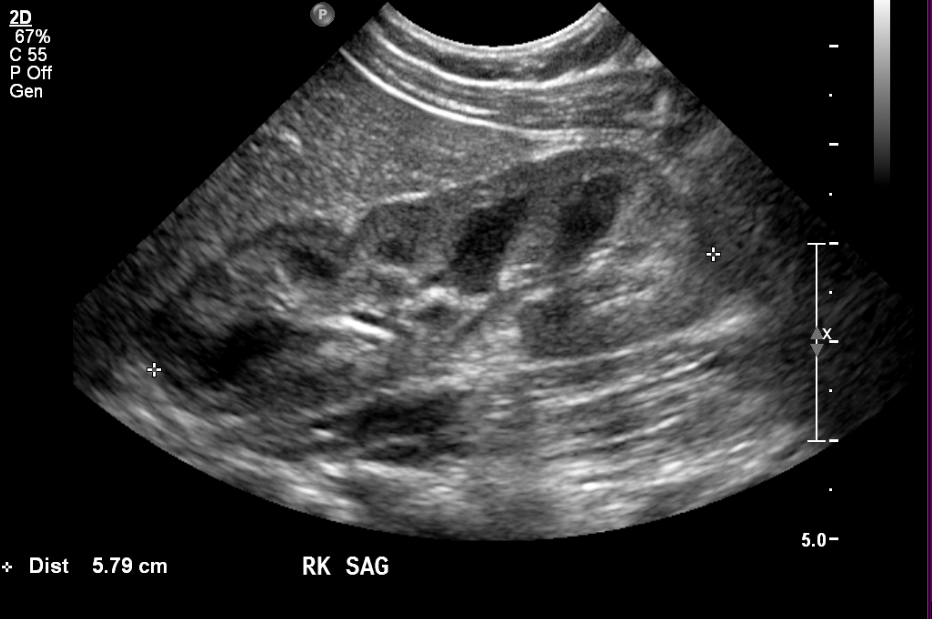
Figura 1 Riñón normal en un lactante de cuatro meses de edad.
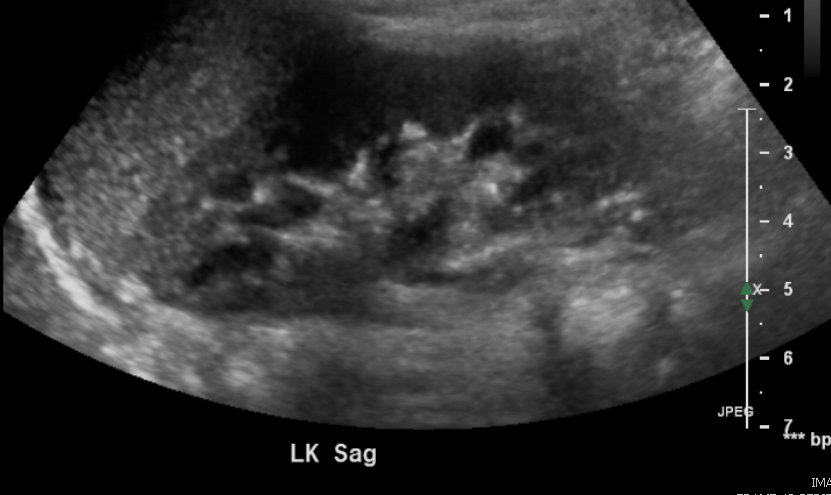
Figura 2 Riñón normal en un niño de 3 años.
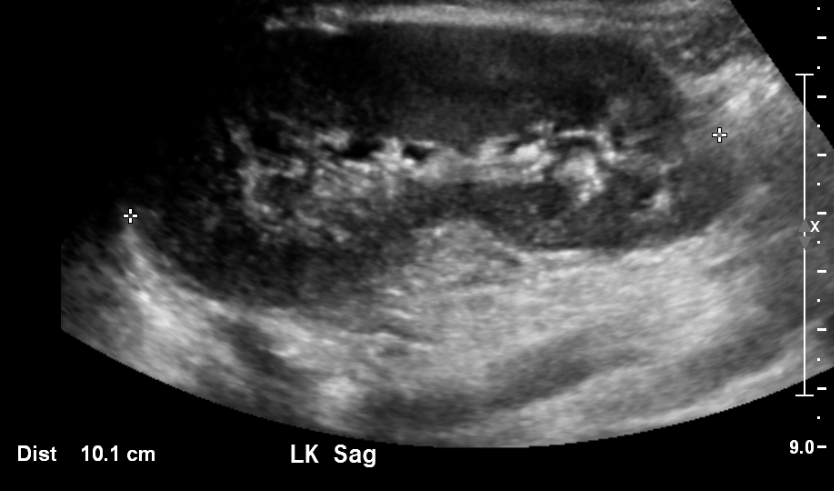
Figura 3 Riñón normal en un paciente de 11 años.
Seguimiento de la dilatación prenatal del tracto urinario
Esta es la indicación más frecuente para una ecografía renal pediátrica en el ámbito ambulatorio. La arquitectura del sistema colector dilatado suele estar bien caracterizada por la ecografía y puede aportar pistas sobre la etiología. A pesar de numerosos esfuerzos de estandarización (iniciados en 1988 y aún en curso), la terminología descriptiva ha sido lamentablemente inconsistente. Esta inconsistencia ha dificultado los esfuerzos por correlacionar los desenlaces urológicos con el grado de dilatación prenatal del sistema colector. Si bien los criterios de idoneidad de la ACR se basan en el sistema de clasificación SFU de 1988, la mayoría de los hospitales pediátricos han adoptado ahora el sistema de clasificación UTD. Este sistema de clasificación se desarrolló en 2014. UTD introdujo una terminología descriptiva unificada tanto para la ecografía prenatal como posnatal con el fin de facilitar una evaluación perinatal estandarizada. Se recomienda al clínico familiarizarse con la redacción empleada en su servicio de radiología afiliado y promover la estandarización siempre que sea posible.
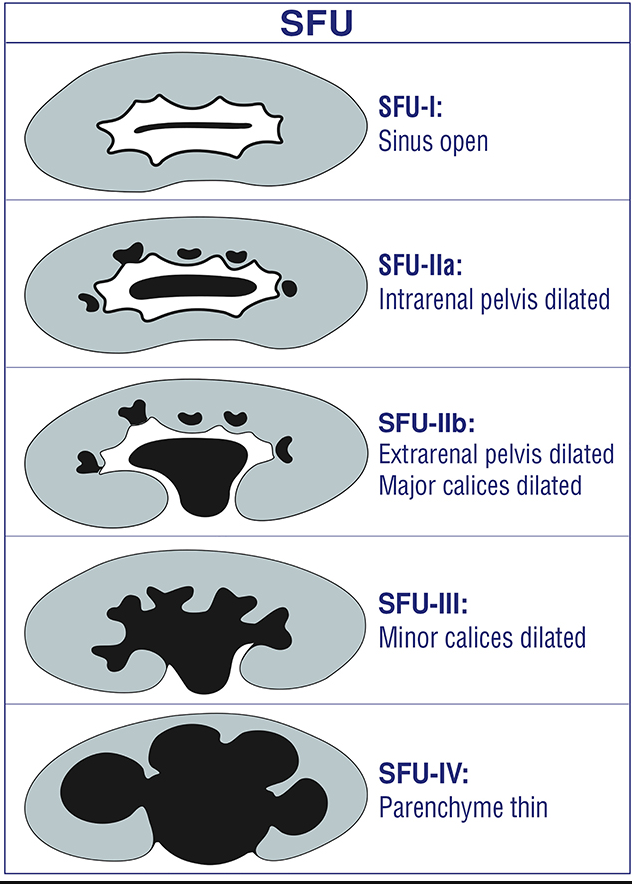
Figura 4 Clasificación SFU de la dilatación del sistema colector
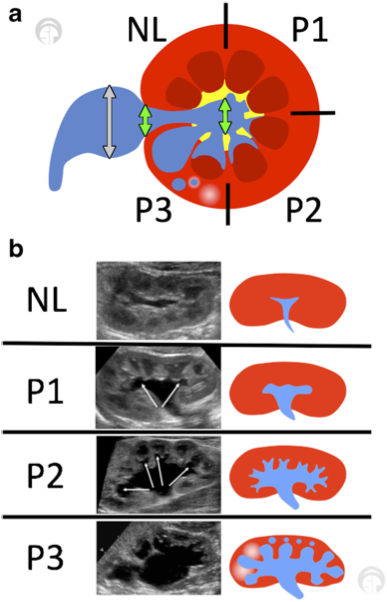
Figura 5 (a) La clasificación de la dilatación del tracto urinario (UTD) muestra una vista transversal del riñón en su porción media/interpolar. Las flechas verdes indican el diámetro anteroposterior (utilice la medición mayor). No se debe incluir la pelvis extrarrenal (flechas grises). (b) Vistas longitudinales de varios grados dentro del sistema de clasificación UTD. Adaptado de Chow et al 2017.10
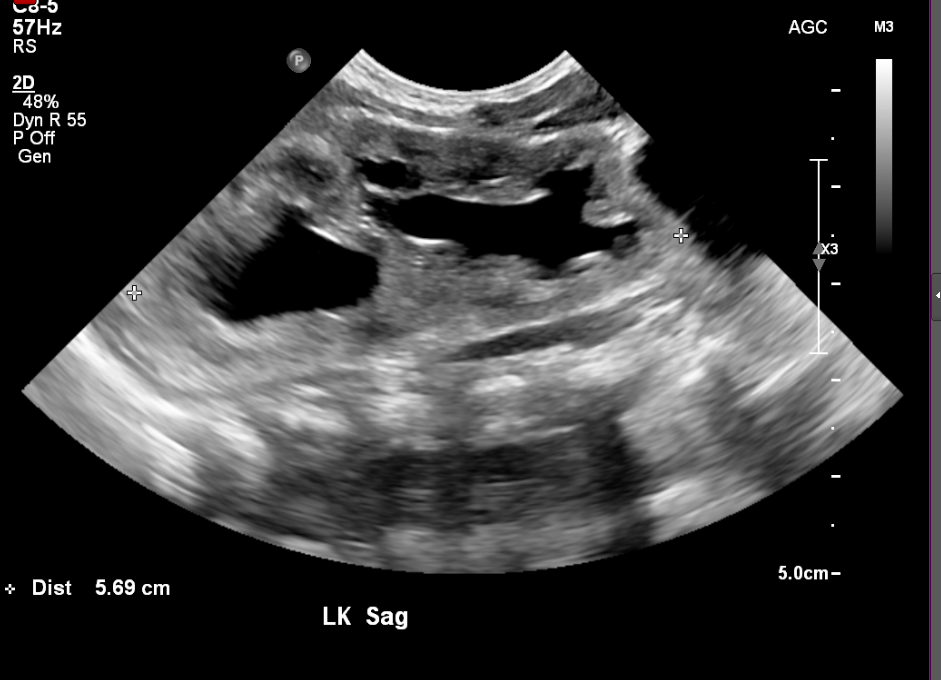
Figura 6 Sistema colector duplicado en un recién nacido
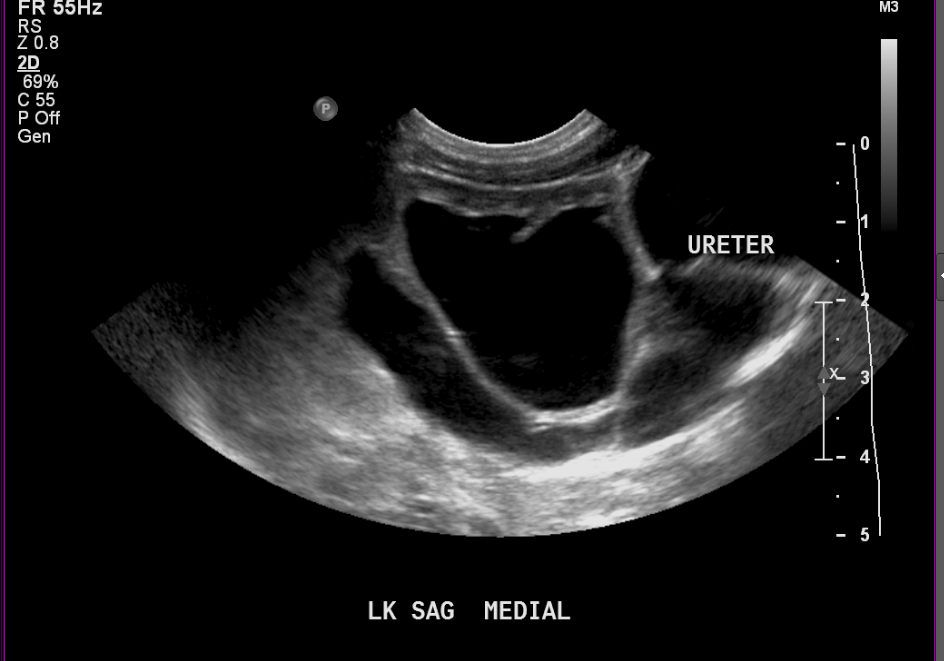
Figura 7 Mismo paciente que en (Figura 6) a las dos semanas de vida, mostrando una dilatación más grave
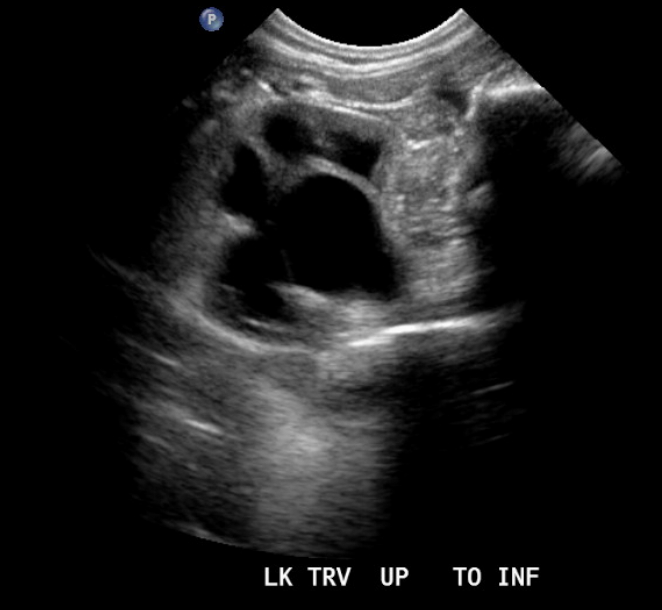
Figura 8 Obstrucción de la unión pieloureteral
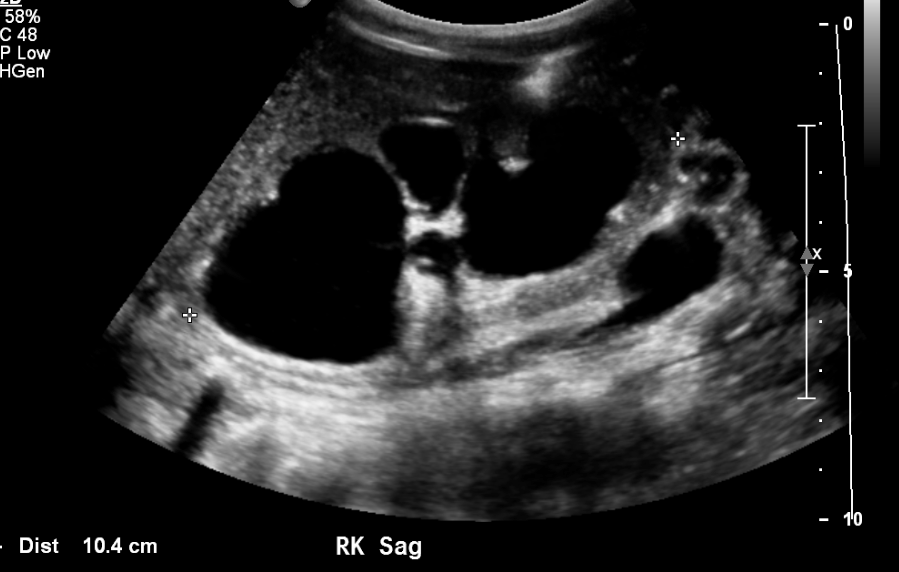
Figura 9 Caliectasia severa y adelgazamiento cortical en un paciente con válvulas uretrales posteriores
Cribado para tumor en condiciones genéticas predisponentes como Beckwith-Wiedemann o WAGR
La ecografía puede detectar masas sólidas sutiles en niños pequeños, como se ilustra en el ejemplo a continuación. La frecuencia del cribado y el rango de edad varían según la condición predisponente específica.11
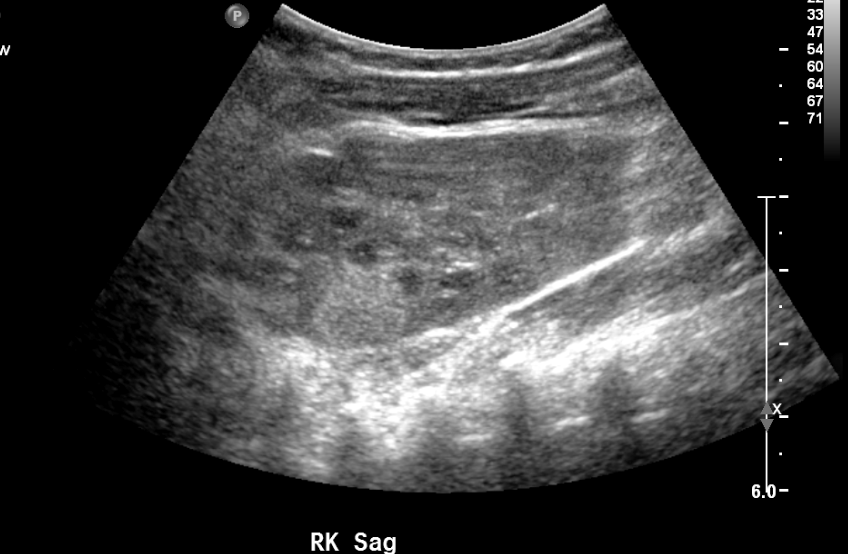
Figura 10 Masa renal hiperecogénica en la ecografía de cribado de un paciente con síndrome WAGR.
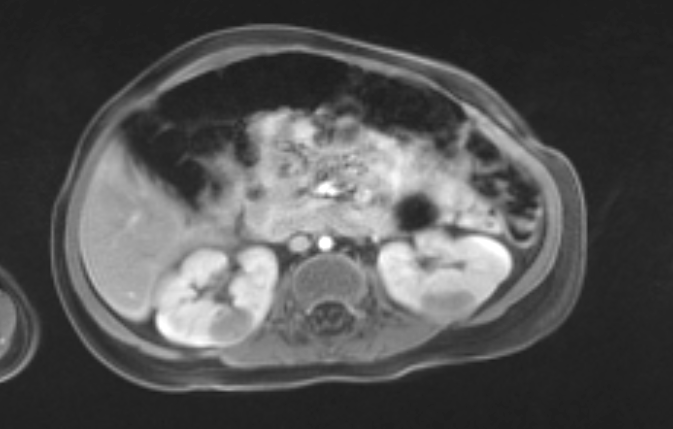
Figura 11 La RM confirmó múltiples masas (mismo paciente que la Figura 10).
Flujo sanguíneo renal
La ecografía proporciona información valiosa en casos de sospecha de estenosis de la arteria renal.
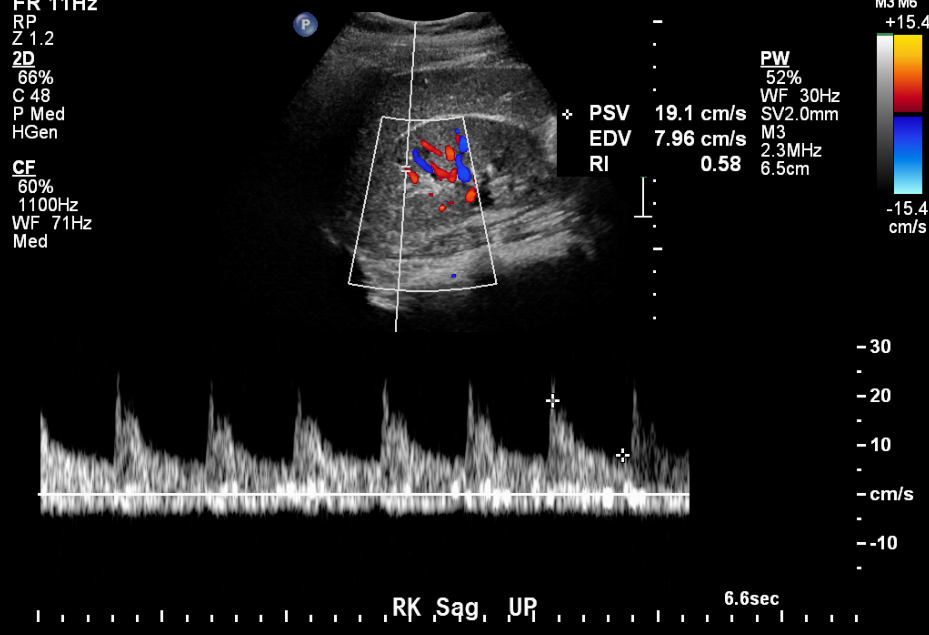
Figura 12 Trazado arterial normal en un paciente de 11 años con hipertensión.
Cálculos renales
Los cálculos obstructivos pueden ser directamente visibles por ecografía o pueden inferirse por la presencia de un sistema colector dilatado en el lado sintomático. El artefacto de centelleo puede aprovecharse para detectar cálculos.
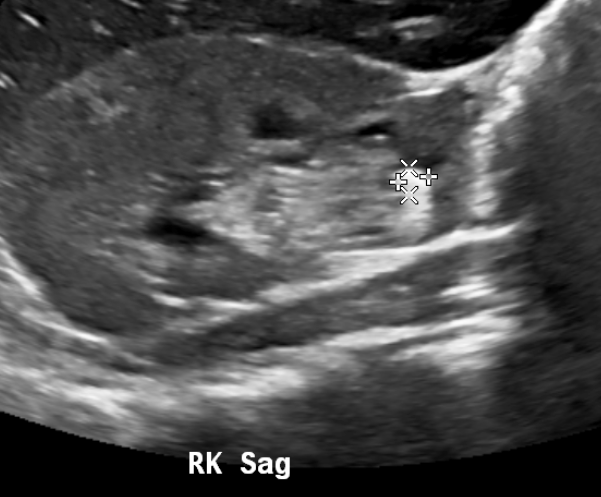
Figura 13 Cálculo en el polo inferior del riñón derecho.
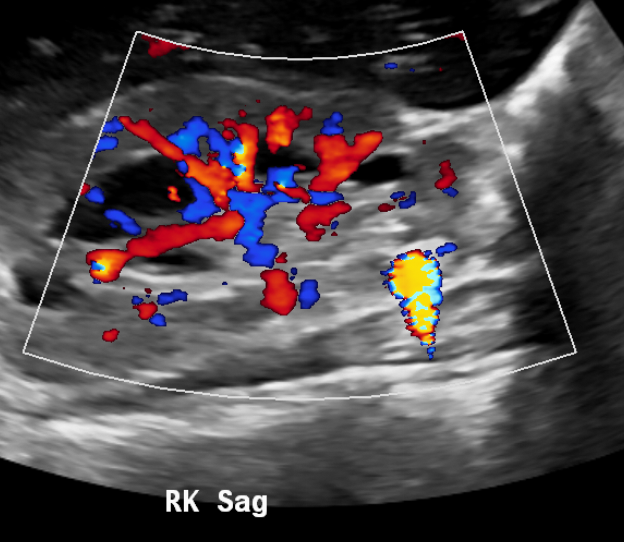
Figura 14 El artefacto de centelleo mejora la conspicuidad de los cálculos.
Anomalías vesicales
Una vejiga adecuadamente distendida puede evaluarse en busca de engrosamiento de la pared, trabeculación, detritos o cálculos. Las masas vesicales pueden hallarse de forma incidental. Las imágenes posmiccionales son útiles en pacientes con sospecha de micción disfuncional.
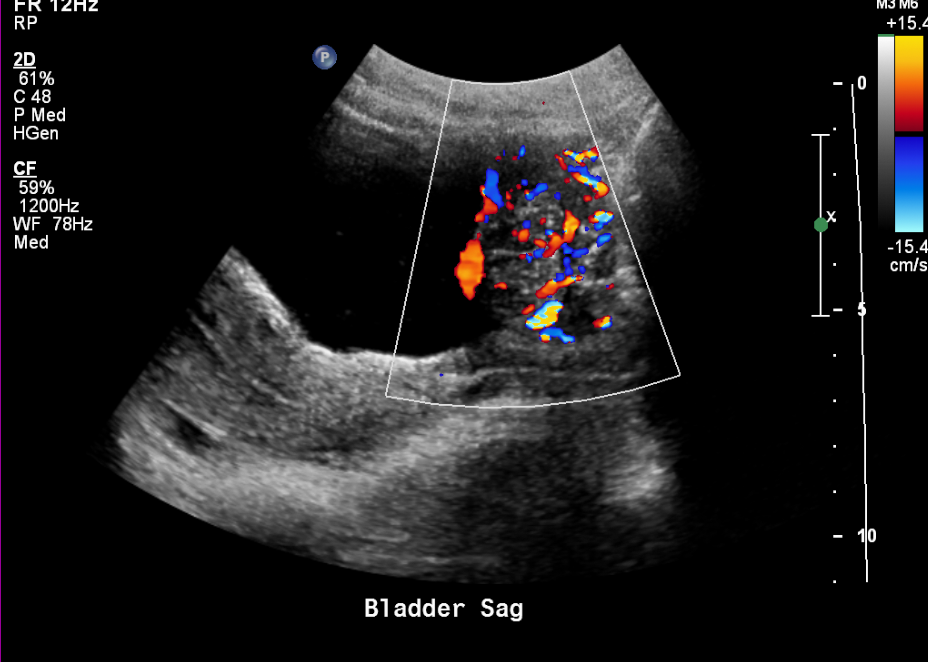
Figura 15 Masa vesical altamente vascularizada en un adolescente.
Técnica:
- Vejiga llena (en un niño con control de esfínteres).
- La vejiga se explora primero antes que el resto del abdomen para evitar el vaciamiento precoz de la vejiga.
- Vistas sagitales de los riñones; las mejores para la longitud. Idealmente, el ecografista debería revisar ecografías previas para tener una idea de la longitud renal esperada.
- Vista transversal a nivel del polo superior, interpolar e inferior; la mejor para la dilatación del sistema colector intrarrenal. Una pelvis extrarrenal vista en el plano sagital puede simular una dilatación leve del sistema colector intrarrenal.
Ecografía escrotal
La ecografía es la modalidad de elección para evaluar el escroto en el contexto de dolor o anomalía palpable.12
Técnica:
- Imágenes de los testículos en su mayor dimensión en dos planos y mediciones
- Imágenes de la cabeza del epidídimo (niños mayores) y mediciones
- Doppler color que muestre ambos testículos
- Doppler espectral que muestre flujo arterial en ambos testículos
- Vistas de los cordones espermáticos (idealmente clips de movimiento)
- Documentar hidrocele o varicocele si están presentes
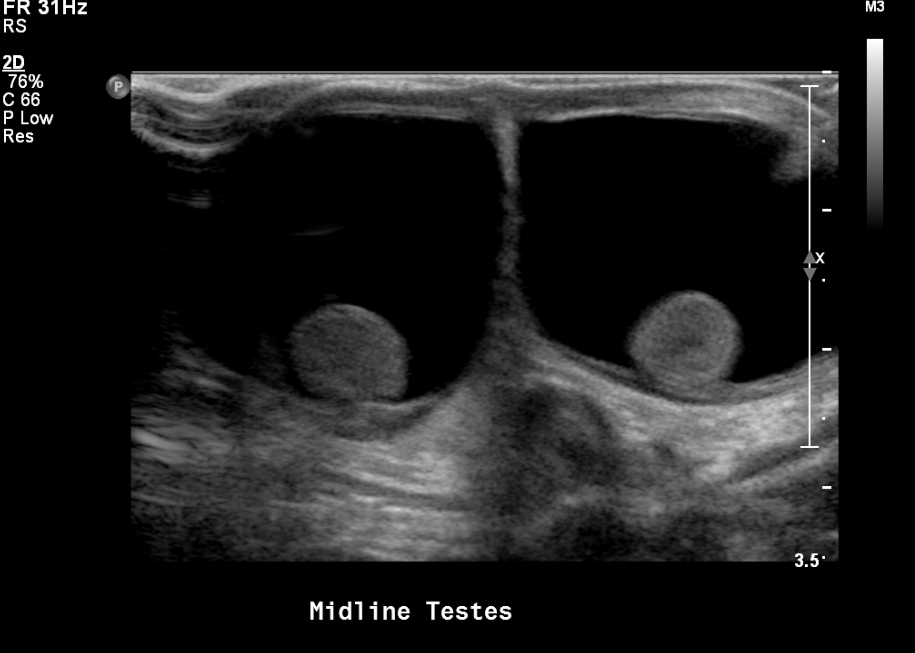
Figura 16 Hidroceles bilaterales en un neonato
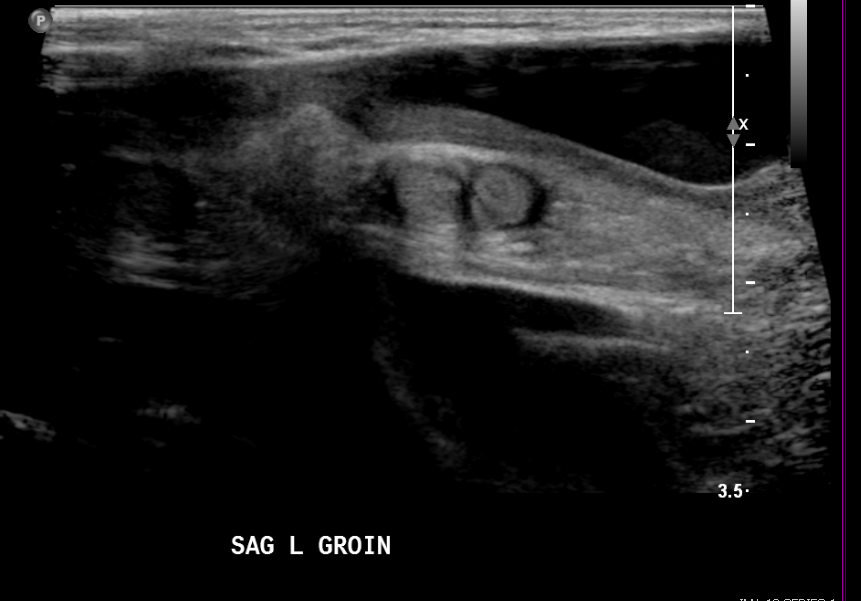
Figura 17 Hidrocele no comunicante en un neonato
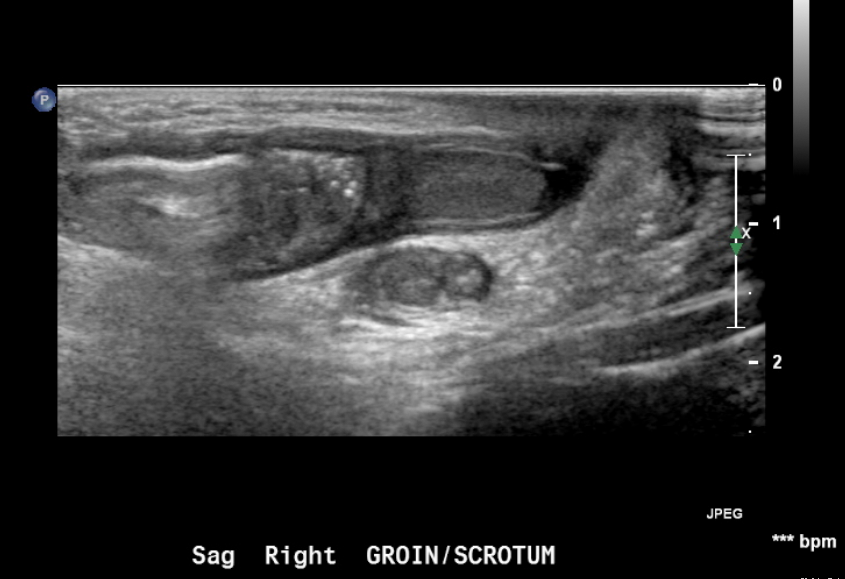
Figura 18 Hernia inguinal en un recién nacido
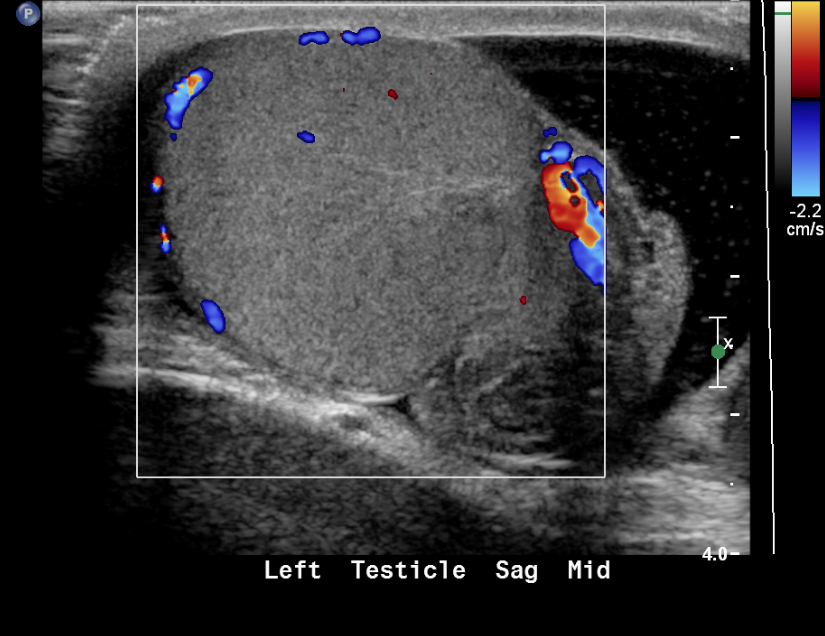
Figura 19 Testículo torsionado con ausencia de flujo en Doppler color
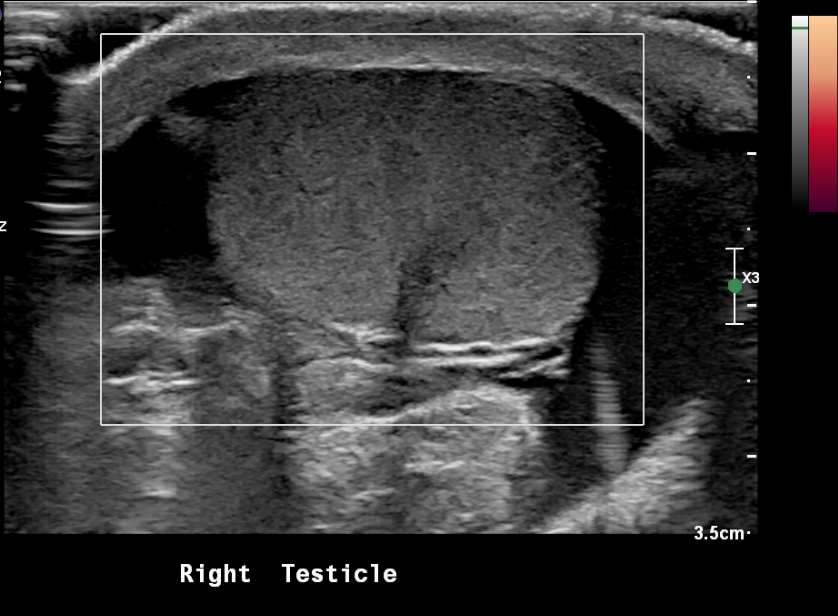
Figura 20 Testículo en torsión con ausencia de flujo sanguíneo en Doppler de potencia
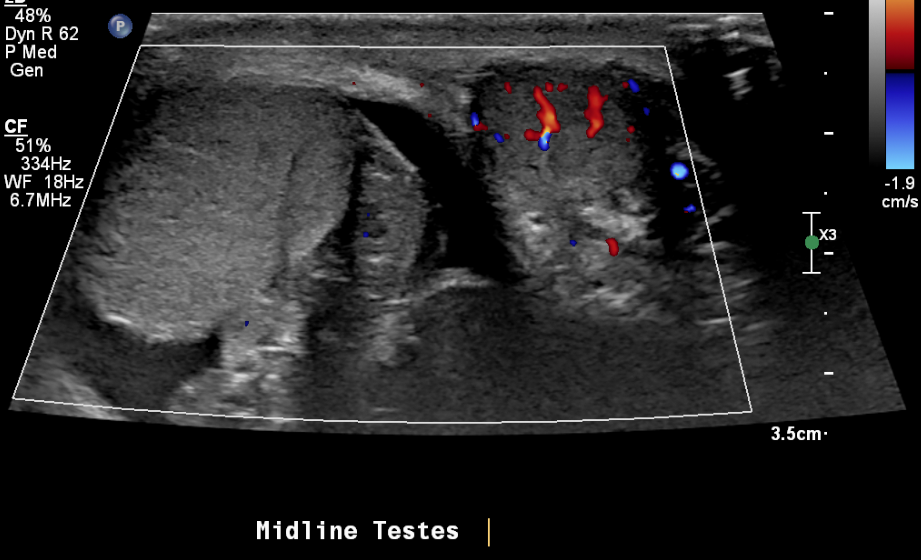
Figura 21 Testículo derecho con torsión y flujo sanguíneo normal al testículo izquierdo
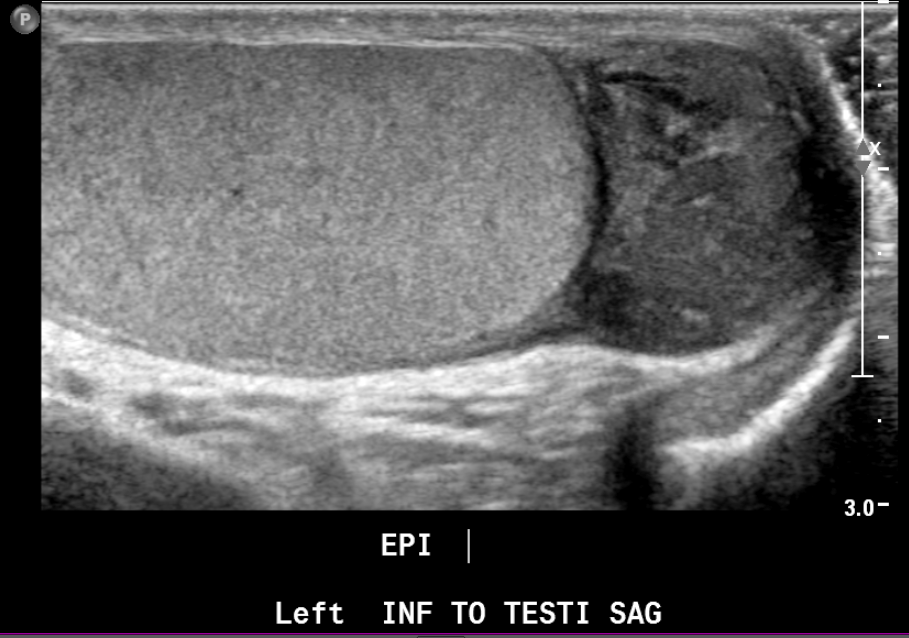
Figura 22 Rabdomiosarcoma paratesticular en un adolescente con masa palpable indolora
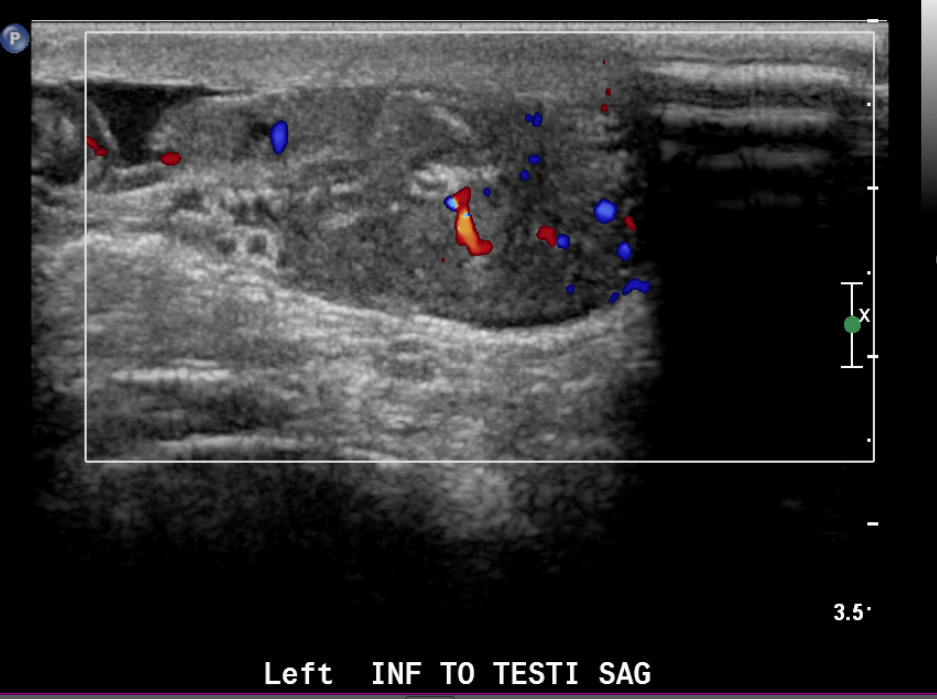
Figura 23 Imagen en color de rabdomiosarcoma paratesticular
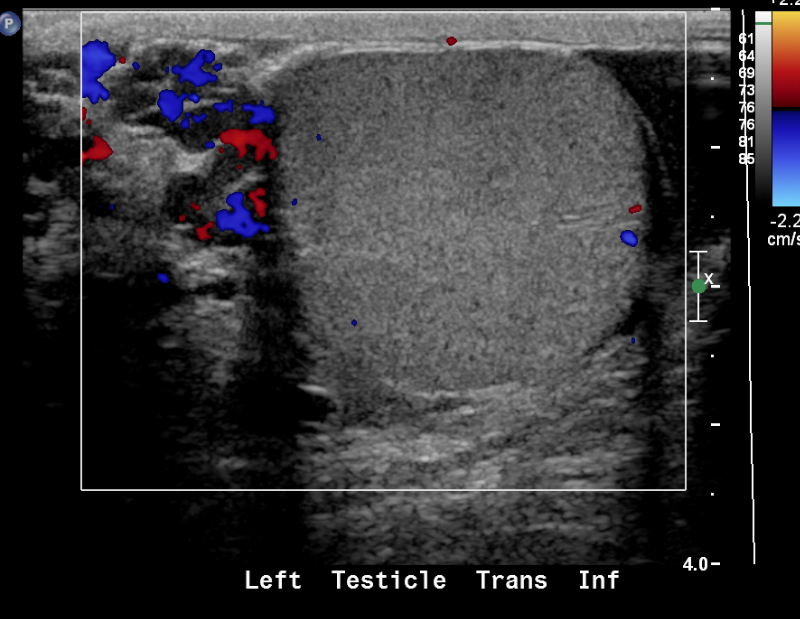
Figura 24 Varicocele
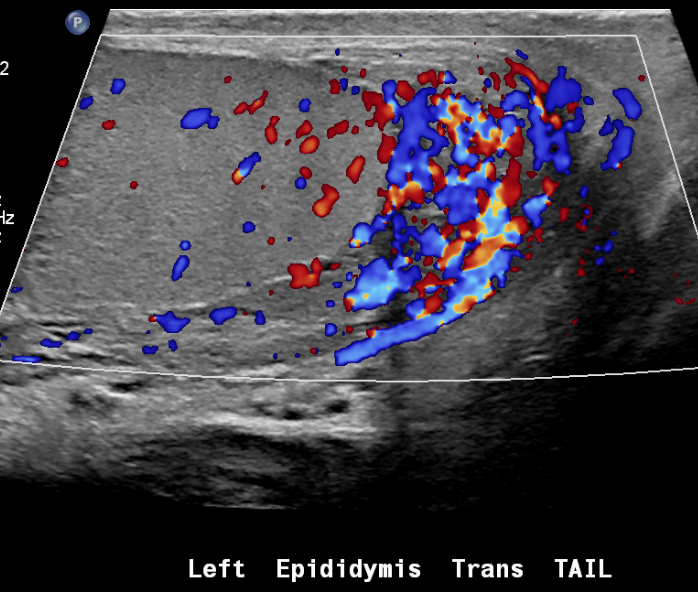
Figura 25 Epididimitis
Radiografía
Las radiografías utilizan la diferencia en la densidad radiográfica de las distintas partes del cuerpo para crear una imagen (aire, grasa, líquido, hueso y metal). Las radiografías modernas se almacenan mediante un sistema de archivado y comunicación de imágenes (PACS), que es un sistema de imágenes digitales. Las ventajas de PACS incluyen una resolución mejorada de las imágenes y la capacidad de compartir el acceso de forma instantánea y remota.
Las radiografías abdominales se realizan con frecuencia en el entorno agudo (dolor abdominal agudo) para evaluar el patrón de gas intestinal y excluir aire libre (para esto último suelen requerirse dos proyecciones).
Otros hallazgos potenciales:
- Calcificaciones de partes blandas
- Masas abdominales grandes
- Organomegalia
- Anomalías óseas
- Posicionamiento de dispositivos de soporte (p. ej., stents ureterales)
También es común que los urólogos pediátricos soliciten en el ámbito ambulatorio una única radiografía abdominal en decúbito supino en casos de estreñimiento conocido o sospechado. Por consenso de expertos, esto no está indicado en niños con grandes cantidades de heces en el examen rectal.13 Puede haber una indicación para esta práctica en casos en los que no sea posible realizar un examen rectal (como en antecedentes traumáticos). El profesional debe recordar que las radiografías abdominales pueden conllevar una carga de radiación no despreciable con el tiempo.
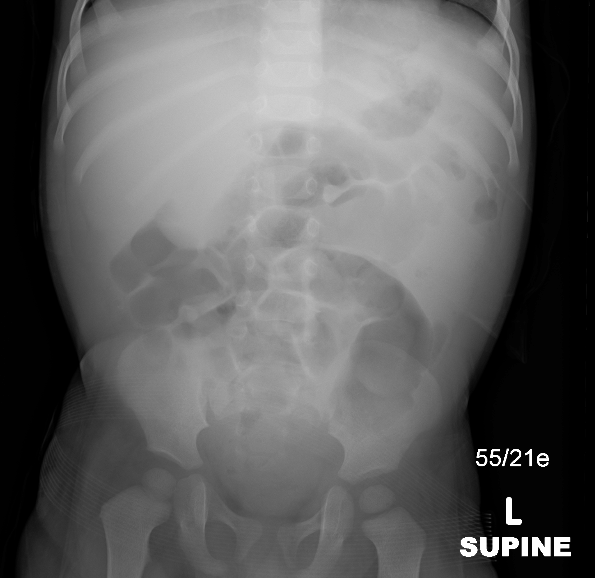
Figura 26 Radiografía abdominal que muestra efecto de masa en el cuadrante superior derecho
Cistouretrografía miccional y urosonografía miccional
La VCUG es un procedimiento radiológico especializado que se utiliza principalmente para evaluar la presencia de reflujo vesicoureteral (RVU) y para evaluar la morfología uretral. La justificación ha sido la presunta relación entre el reflujo y la cicatrización posinfecciosa. A pesar de treinta años de estudio desde el inicio de la profilaxis antibiótica rutinaria para el reflujo, persisten interrogantes. Las cistografías se realizan con menos frecuencia que en el pasado, y con frecuencia se prefiere la conducta expectante a la profilaxis antibiótica.14 Tradicionalmente, los estudios miccionales se han realizado mediante fluoroscopia. En Estados Unidos está en marcha una transición hacia el uso de ecografía con contraste (ceUS). En Europa, la ecografía con contraste se realiza desde la década de 1990 y ha estado ampliamente disponible desde principios de la década de 2000. Sin embargo, los agentes de contraste para ecografía no fueron aprobados para su uso en Estados Unidos hasta 2016. Persiste el debate sobre la sensibilidad de la ceVUS para detectar anomalías uretrales en el lactante varón. Al momento de esta redacción, los criterios de adecuación del ACR siguen recomendando la VCUG sobre la ceVUS en ese contexto, a pesar de la existencia de artículos de revisión que muestran una excelente visualización de la anatomía uretral mediante ceVUS.8,15,16
La fluoroscopia todavía se utiliza con frecuencia, incluso en la institución de este autor, a pesar de que expone al paciente a radiación ionizante. Otra desventaja de la fluoroscopia tradicional es que requiere que el paciente permanezca en decúbito supino para orinar; esto suele ser difícil para los pacientes con control de esfínteres.
Se requiere cateterismo vesical aséptico para ambos tipos de prueba, y a menudo es la parte más difícil del examen para el paciente. Se introduce una sonda de alimentación pequeña (de 5 a 10 Fr, según el tamaño y la edad del paciente) a través de la uretra hasta la vejiga. El uso de sedación en niños de aproximadamente dos a seis años es controvertido. Deben sopesarse los riesgos potenciales de la sedación frente al daño ocasionado por el estrés psicológico. El uso de una tabla octagonal o similar para inmovilizar al paciente también es controvertido. Algunos radiólogos pediátricos prefieren usar la tabla en lactantes mayores o niños pequeños. Un profesional de Child Life es un recurso valioso durante estos estudios y puede ser una presencia reconfortante tanto para el paciente como para el progenitor acompañante.
Una vez colocado el catéter y vaciada la vejiga, se instila el medio de contraste bajo observación fluoroscópica (o ecográfica). El examen ecográfico puede utilizar un manguito de presión arterial para infundir el contraste a baja presión. Para la VCUG, el contraste se instila por gravedad. Las imágenes estándar obtenidas tanto en VCUG como en ceVUS son esencialmente las mismas.
Técnica de VCUG:
- Imagen puntual fluoroscópica del abdomen (lactante) o a nivel de los riñones y a nivel de la vejiga (niño mayor)
- Imagen puntual o retención de la última imagen de la vejiga al llenado inicial (evaluación de ureterocele)
- Controles intermitentes para evaluar reflujo
- Cerca de la capacidad vesical estimada, obtener proyecciones oblicuas bilaterales con retención de la última imagen o imágenes puntuales
- En la primera micción, comprobar reflujo
- Segundo ciclo (como mínimo) en lactante o paciente con ITU febril
- Proyección lateral en la micción de la uretra en paciente varón (idealmente con el catéter retirado)
Las imágenes estándar de la urosonografía miccional serán en su mayoría similares, con la excepción de la vista de localización inicial. Las vistas panorámicas pueden ayudar a ampliar el campo de visión en la ecografía. Sin embargo, el campo de visión reducido sigue siendo una limitación de la ecografía.
Se debe tener cuidado de llenar la vejiga de manera adecuada y no sobrellenarla. Una fórmula útil de capacidad vesical (para niños mayores de 12 meses) es (edad + 2) multiplicado por 30 para la capacidad vesical prevista (en mL). Se espera que un recién nacido retenga alrededor de 50 mL. Deben obtenerse imágenes puntuales oblicuas fluoroscópicas o, al menos, retenciones de la última imagen con distensión vesical máxima para evaluar reflujo de bajo grado (que puede quedar oculto en la proyección AP). Dar golpecitos sobre la vejiga, un masaje suave sobre el abdomen o salpicar agua tibia sobre la piel pueden ser útiles para inducir la micción en niños pequeños. La sedación ocasionalmente puede dificultar la micción de los pacientes. Debe evitarse el sobrellenado (a más del doble de la capacidad prevista), ya que puede hacer más probable este problema.
- Indicaciones
- Reflujo vesicoureteral
- Estudio de la uretra durante la micción
- Anomalías vesicales
- Contraindicaciones
- Infección del tracto urinario aguda
- Antecedente de reacción grave al contraste
- Preparación del paciente
- El paciente debe orinar antes del estudio (en niños pequeños se puede vaciar la vejiga antes del llenado).
- Profilaxis antibiótica con trimetoprim 2 mg/kg/dosis una vez al día durante 3 días antes del estudio (en niños que no reciben antibióticos profilácticos)
- Nota del editor: La evidencia al respecto es limitada y la práctica no es universal.
- Complicaciones
- Reacciones relacionadas con el contraste
- Infección del tracto urinario
- Sobredistensión vesical y rotura
- Traumatismo por catéter
- Hallazgos
- Grado I – reflujo hacia un uréter no dilatado
- Grado II – reflujo hacia la pelvis renal y los cálices sin dilatación
- Grado III – dilatación leve/moderada del uréter, la pelvis renal y los cálices con mínimo embotamiento de los fórnices
- Grado IV – dilatación de la pelvis renal y los cálices con tortuosidad ureteral moderada
- Grado V – dilatación marcada del uréter, la pelvis renal y los cálices; tortuosidad ureteral; pérdida de las impresiones papilares
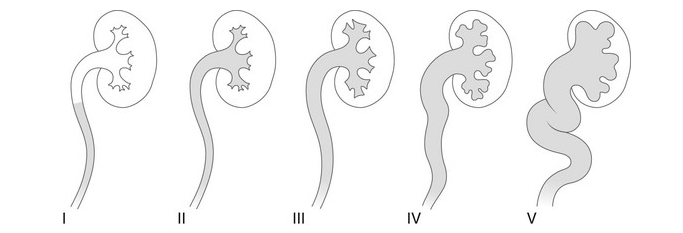
Figura 27 Clasificación radiológica del reflujo vesicoureteral
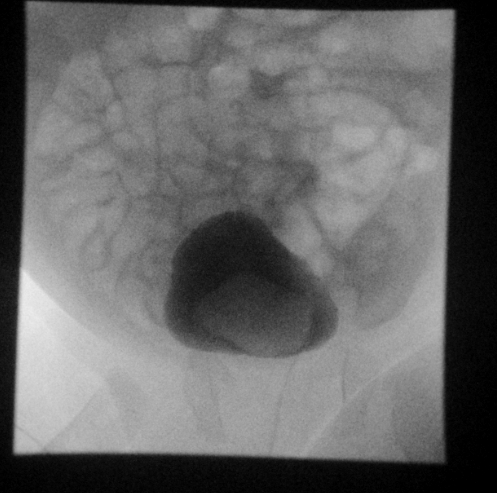
Figura 28 Ureterocele en retención de la última imagen fluoroscópica
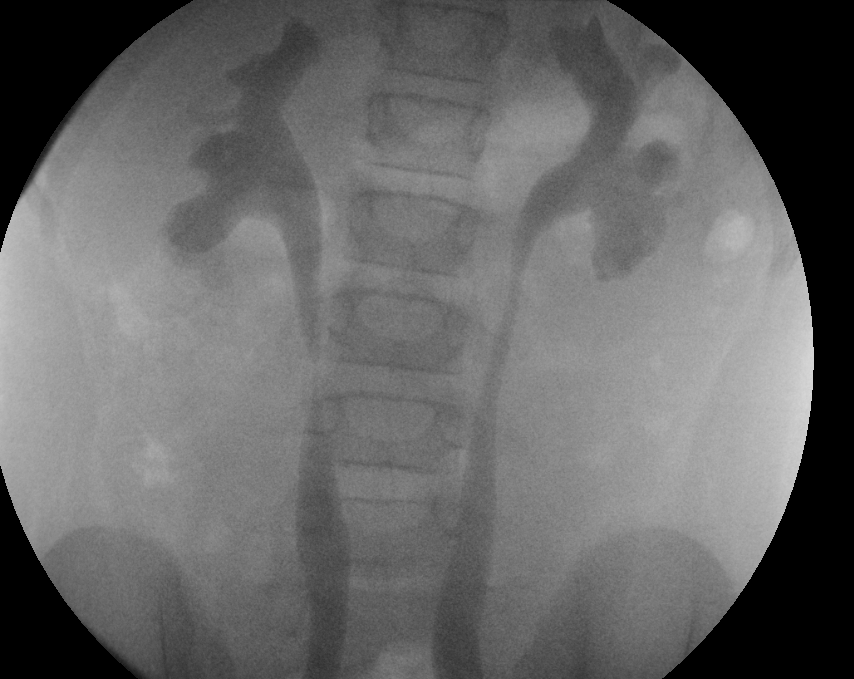
Figura 29 Reflujo de grado 3 hacia ambos riñones
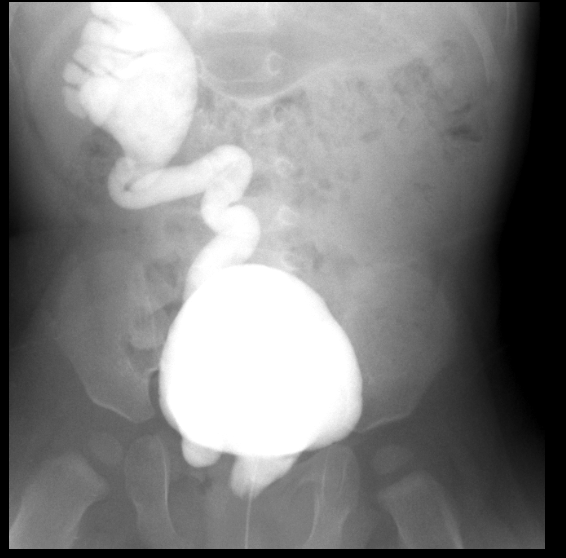
Figura 30 Reflujo de grado 5 en el lado derecho con inserción ectópica en la uretra
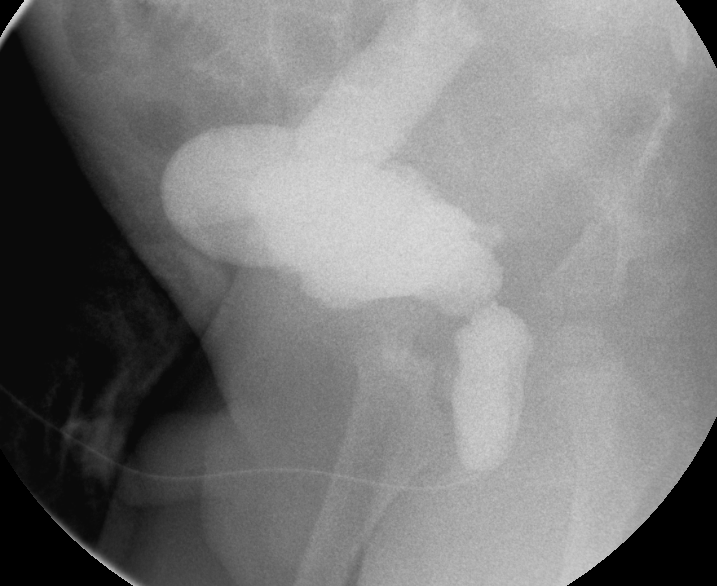
Figura 31 Uretra posterior dilatada y uréter derecho marcadamente dilatado en el contexto de válvulas uretrales posteriores
Estudios con radionúclidos
DMSA / Gammagrafía renal estática
El ácido dimercaptosuccínico (DMSA) se une a las proteínas plasmáticas y es retenido por la corteza renal durante el aclaramiento renal, lo que permite la evaluación funcional. Los datos cuantitativos sobre la función renal son información crucial que no puede obtenerse mediante ecografía. La gammagrafía con Tc99m ácido dimercaptosuccínico (DMSA) se ha considerado la prueba de elección en la evaluación del daño parenquimatoso debido a pielonefritis aguda o crónica y proporciona datos sobre la función renal diferencial. No se requiere un catéter vesical para la exploración con DMSA, ya que el radiofármaco no se excreta en la orina. La SPECT con DMSA aumenta la sensibilidad, pero se requiere sedación en niños pequeños para obtener imágenes de alta calidad.17 El DMSA no proporciona información sobre el sistema colector ni sobre la urodinámica.
Fragmento:
- Indicaciones
- Evaluar la función renal
- Estudio de infección renal
- Anomalías renales congénitas (p. ej., riñones en herradura)
- Cicatrices renales y lesiones renales
- Contraindicaciones
- Ninguna
- Técnica
- Se inyecta DMSA por vía intravenosa y se obtienen imágenes con una gammacámara 1–6 horas después.
- Información obtenida
- Función renal relativa
- Captación absoluta
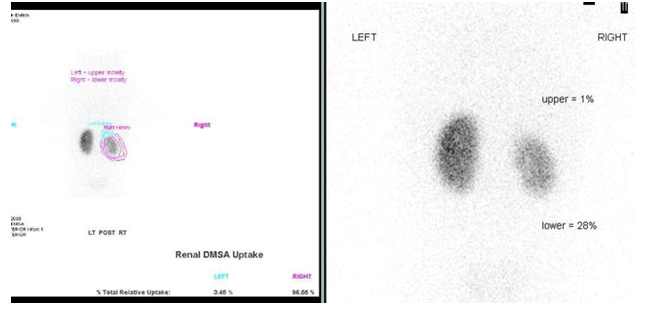
Figura 32 Gammagrafía renal con DMSA que muestra cicatrices y función reducida del polo inferior derecho de un riñón duplicado y un polo superior no funcionante
Gammagrafía renal con MAG-3 / Gammagrafía renal dinámica
Mercaptoacetiltriglicina (MAG-3) está altamente unida a proteínas plasmáticas, por lo que se elimina por secreción tubular. La gammagrafía dinámica con MAG3 aporta información sobre la función y la morfología renales. Se requiere sedación para niños pequeños (menores de aproximadamente 5 años). La parte inicial del estudio demuestra la perfusión y la función cortical renal, lo que permite distinguir entre lesiones reversibles e irreversibles. A continuación, el sistema colector y la urodinámica se caracterizan con ayuda de furosemida. Una gammagrafía con MAG-3 tranquilizadora permite al urólogo pediatra observar de forma segura a un paciente con hidronefrosis en la mayoría de los casos.18
- Indicaciones
- Sistema obstruido vs no obstruido
- Estenosis de la arteria renal
- Enfermedad por reflujo
- Traumatismo renal
- Contraindicaciones
- Ninguna
- Técnica
- Hidratar al paciente y vaciar la vejiga
- Colocación de catéter vesical
- Inyección de MAG-3
- Imagen a los 2–3 minutos con una gammacámara para la fase parenquimatosa
- Imagen a partir de los 18–20 minutos
- Furosemida 1 mg/kg, máximo 40 mg, inyección cuando la pelvis renal dilatada esté llena
- Imágenes secuenciales durante aproximadamente 20 minutos adicionales
- Regiones de interés delineadas alrededor de los riñones
- Se generan curvas de aclaramiento y se calcula la t½ (tiempo para que se elimine la mitad del radiotrazador)
- Información obtenida
- Perfusión renal
- Perfusión cortical (función renal diferencial)
- Tiempos de tránsito del parénquima y de todo el riñón
- T½ menor de 10 minutos es normal
- T½ entre 10 y 20 minutos indeterminado
- T½ mayor de 20 minutos sugiere obstrucción
- Escollos
- Deshidratación que ocasiona una respuesta diurética deficiente
- Función renal subyacente deficiente que ocasiona una respuesta diurética deficiente
- Vejiga distendida o de baja complacencia (colocar catéter)
- Reflujo de alto grado temprano (VCUG/ VUS previos)
- Sistema colector patuloso que retrasa el lavado
- Regiones de interés que incluyen el bazo o el hígado
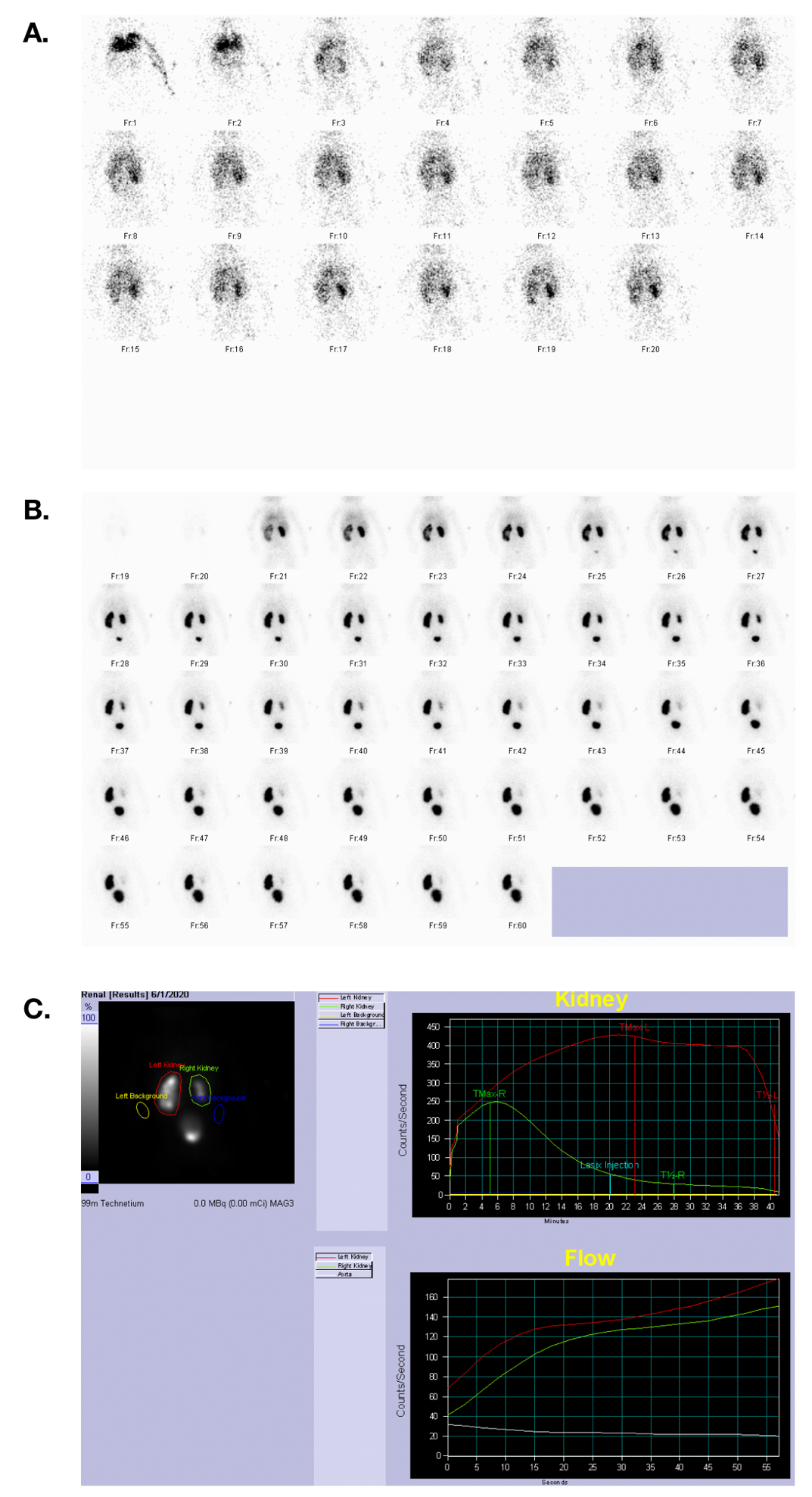
Figura 33 Gammagrafía con MAG 3 que muestra el flujo sanguíneo, la fase excretora y las curvas tiempo-actividad en un paciente con función renal simétrica y una obstrucción de la UPJ izquierda.
Tomografía computarizada
TC con contraste intravenoso es el estándar de atención en niños para la evaluación del traumatismo abdominal cerrado. Es rápida, ampliamente disponible y permite el monitoreo por parte del radiólogo en tiempo real. Idealmente, si se identifica una lesión del sistema colector renal, se puede obtener una segunda adquisición tardía a nivel de los riñones o de la vejiga (según el nivel de lesión sospechada). Con raras excepciones, este es el único uso apropiado de la TC multifásica en la población pediátrica. En Estados Unidos, la RM ha reemplazado en gran medida a la TC para la estadificación tumoral del abdomen y la pelvis pediátricos.
Una TC sin contraste puede ser ocasionalmente útil en el contexto pediátrico para el seguimiento de cálculos renales y ureterales, o para el diagnóstico cuando la ecografía no es concluyente.2 La ecografía puede no mostrar el cálculo obstructivo, especialmente si se encuentra en el uréter medio (donde las sombras del gas intestinal a menudo bloquean el haz ultrasónico). Sin embargo, la presencia de un sistema colector dilatado en el lado sintomático puede permitir al clínico inferir que hay un cálculo. En niños con cálculos conocidos o con afecciones que predisponen a cálculos, puede considerarse la TC. Pueden emplearse dosis de radiación muy bajas para evaluar la carga litiásica o para el seguimiento tras la intervención por cálculos.19 Los protocolos para hematuria (TC trifásica con fases sin contraste, con contraste y tardía) no son apropiados en niños debido a la elevada carga de radiación.2
En los Estados Unidos, la RM ha reemplazado en gran medida a la TC para la estadificación tumoral del abdomen y la pelvis pediátricos.20 Sin embargo, nuestros cirujanos pediátricos solicitan ocasionalmente una angiografía por TC (CTA) con fines de planificación. La CTA muestra las relaciones entre órganos y la anatomía vascular con menos artefactos y mayor resolución que la RM, lo que permite al cirujano planificar un abordaje más preciso en casos de tumor de Wilms, neuroblastoma u otras neoplasias malignas. Dado que la angiografía por TC requiere una inyección de contraste más rápida y conlleva una dosis de radiación más alta, estos estudios deben ser supervisados activamente por un radiólogo pediátrico.
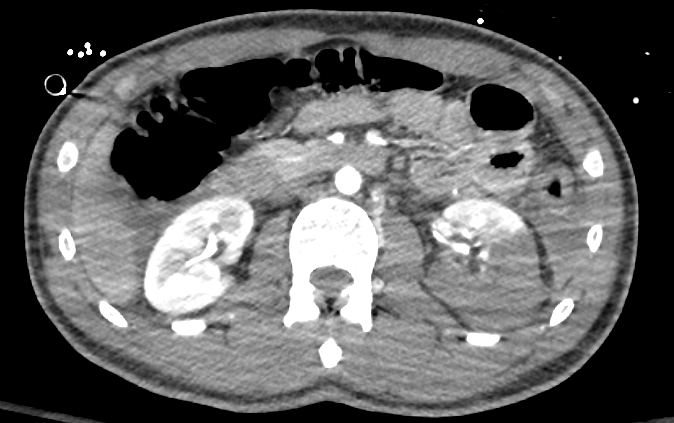
Figura 34 TC con contraste que muestra laceración renal de alto grado del riñón izquierdo.
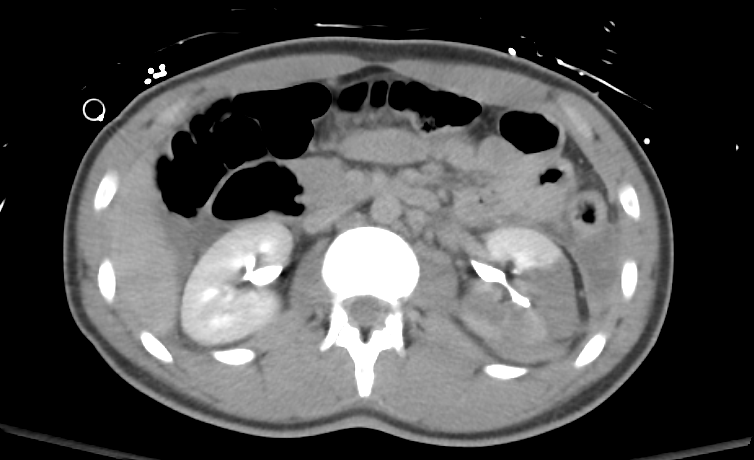
Figura 35 La imagen tardía a través del riñón descarta la disrupción del sistema colector.
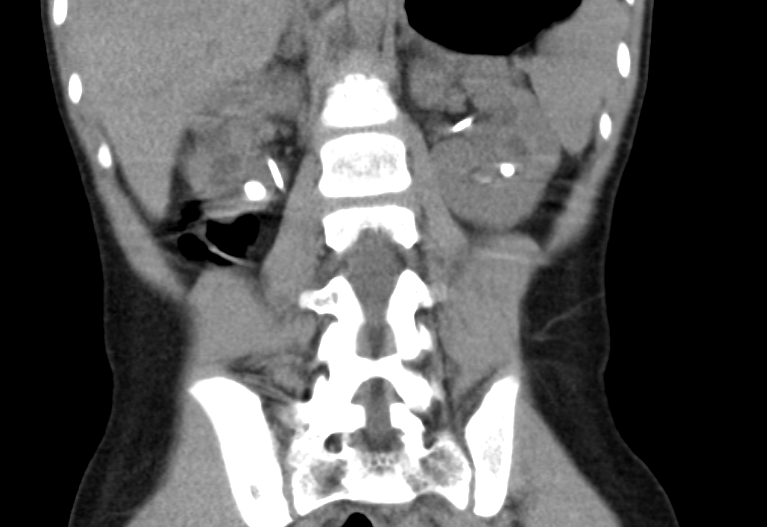
Figura 36 Estudio por TC de litiasis en paciente con cistinuria; nótese la presencia de stents bilaterales parcialmente visibles.
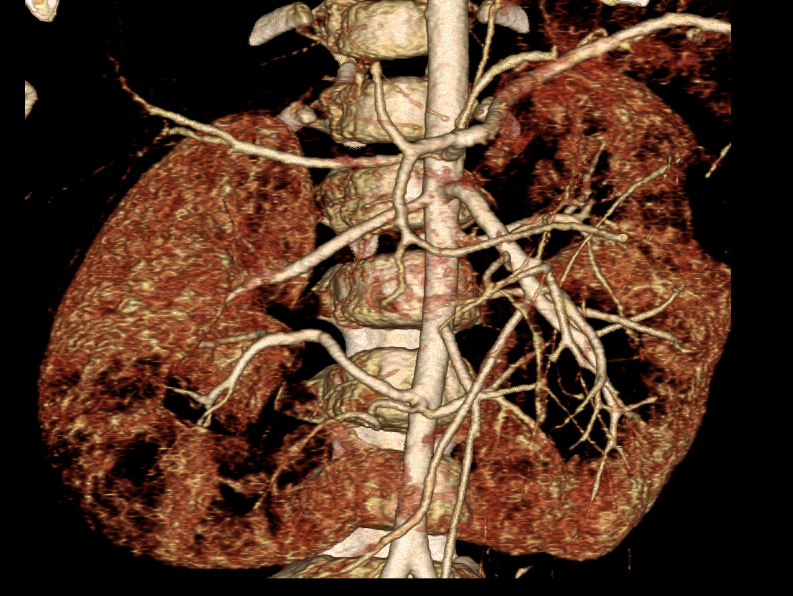
Figura 37 Angiografía por TC con reconstrucción 3D en un paciente oncológico con anatomía arterial compleja; se identifican tres arterias renales para cada hemirriñón
Resonancia magnética
La RM aprovecha las propiedades magnéticas del núcleo de hidrógeno para producir información matemática sobre los tejidos blandos del cuerpo humano, que luego puede transformarse en una imagen detallada. Es superior a cualquier otra modalidad para el contraste de tejidos blandos y no utiliza radiación ionizante. Estas ventajas la han convertido en la modalidad de elección para la estadificación de la mayoría de los cánceres en el abdomen y la pelvis. Existen numerosas secuencias de pulso de RM disponibles comercialmente. Por lo tanto, la RM se adapta a la pregunta clínica (más que la TC), lo que requiere protocolos específicos.
La resolución de la RM sigue siendo inferior a la de la TC (siendo aproximadamente un orden de magnitud inferior a la de la TC en este aspecto), y la evaluación del tejido pulmonar sigue siendo limitada en el entorno clínico. La RM también consume tiempo y es ruidosa, lo que requiere alguna forma de sedación en la mayoría de los niños menores de unos ocho años. Aunque los pacientes adultos pueden (por lo general) seguir instrucciones de apnea, esto es difícil o imposible para los niños pequeños. El software que permite la adquisición con respiración libre es esencial para la obtención de imágenes corporales por RM en niños.21 Estas secuencias suelen tardar más que una secuencia en apnea, pero aun así pueden obtenerse con bastante rapidez (en aproximadamente tres minutos). La duración de una exploración por RM puede oscilar entre treinta minutos y más de una hora, según el protocolo específico.
La evaluación del tracto urinario se denomina urografía por resonancia magnética (MRU). La MRU es capaz de proporcionar información tanto anatómica como funcional en un solo estudio.8 La MRU se correlaciona bien con la gammagrafía renal con Lasix en términos de evaluación de la función renal. Se hidrata a los pacientes con solución salina normal (aproximadamente 10 mL/kg) y se administra Lasix al inicio del estudio, a una dosis de 1 mg/kg hasta un máximo de 20 mg. En casos de sospecha de obstrucción de la unión ureteropélvica, puede ser útil obtener imágenes con el paciente en decúbito prono o sobre el lado contralateral a la obstrucción sospechada. En pacientes sedados, puede colocarse un catéter Foley.8 La MRU en pacientes sedados es un esfuerzo colaborativo que requiere una buena comunicación entre el urólogo, el equipo de radiología y el equipo de sedación o anestesia.
La angiografía por RM evita la exposición del paciente a la radiación, pero tiene menor resolución y es más susceptible a artefactos que la CTA.
En el diagnóstico fetal, la RM es una modalidad complementaria a la ecografía. Las secuencias de pulso rápidas son esenciales para la obtención de imágenes fetales. Si bien la RM fetal se solicita con mayor frecuencia para una evaluación adicional de una anomalía cerebral, también puede aportar información adicional sobre el tracto urinario fetal. Puede ser especialmente útil en el contexto de la planificación quirúrgica de anomalías urogenitales complejas.22 En general, la calidad de imagen en la RM fetal mejora en etapas más avanzadas de la gestación.
- Indicaciones de la RM en pediatría
- Estadificación de cánceres
- Sospecha de masa renal
- Cribado de pacientes en riesgo de tumor (p. ej., TS, VHL)
- Puede ser adecuada para pielonefritis complicada
- Urografía por RM
- Método
- Urografía sensible al líquido utilizando técnicas de RM fuertemente ponderadas en T2
- Urografía excretora por RM utilizando secuencias ponderadas en T1 con contraste y furosemida
- Indicaciones
- Anatomía del sistema colector
- Determinar el nivel de obstrucción en las vías renales (intrínseca y extrínseca)
- Anomalías congénitas
- Método
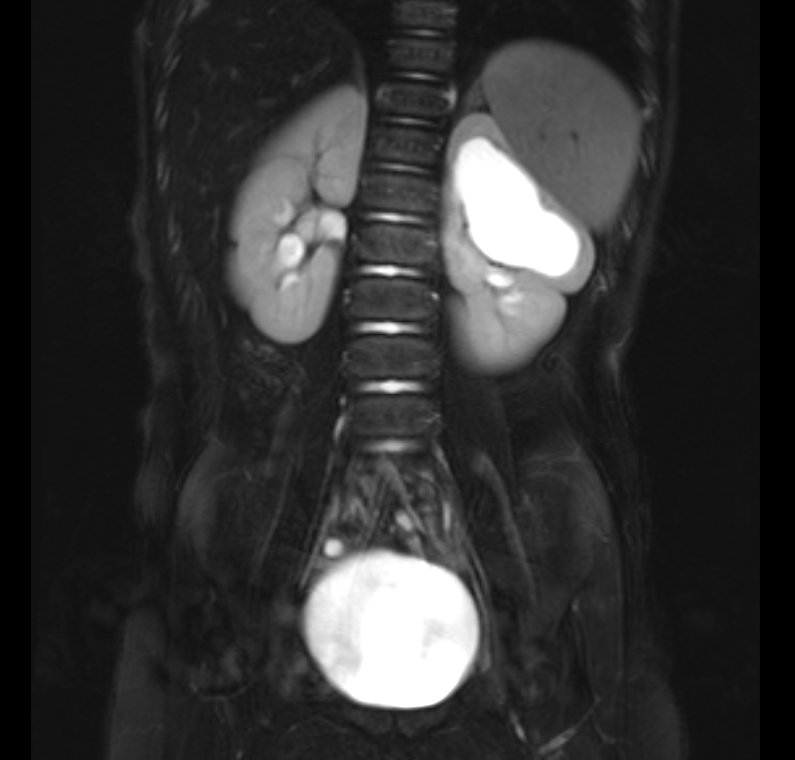
Figura 38 Imagen de RM sensible al líquido con supresión de grasa que muestra dilatación del polo superior izquierdo en un riñón duplicado.
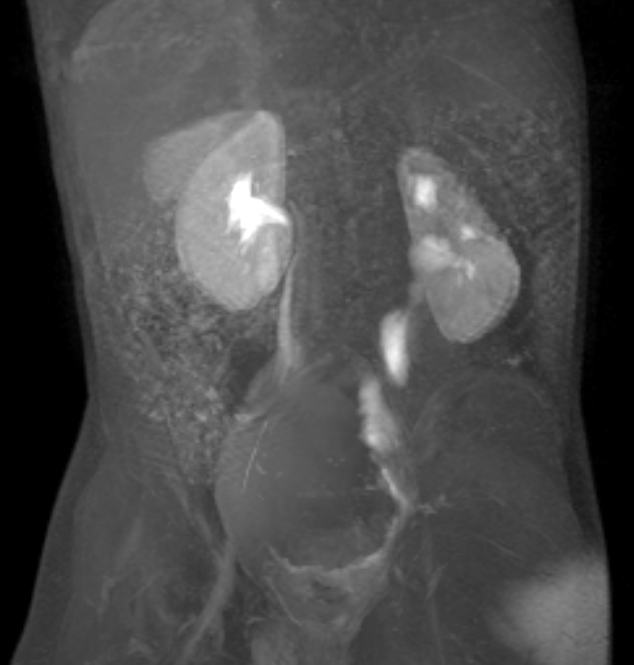
Figura 39 La imagen dinámica de RM poscontraste muestra un megauréter izquierdo dilatado pero no obstruido.
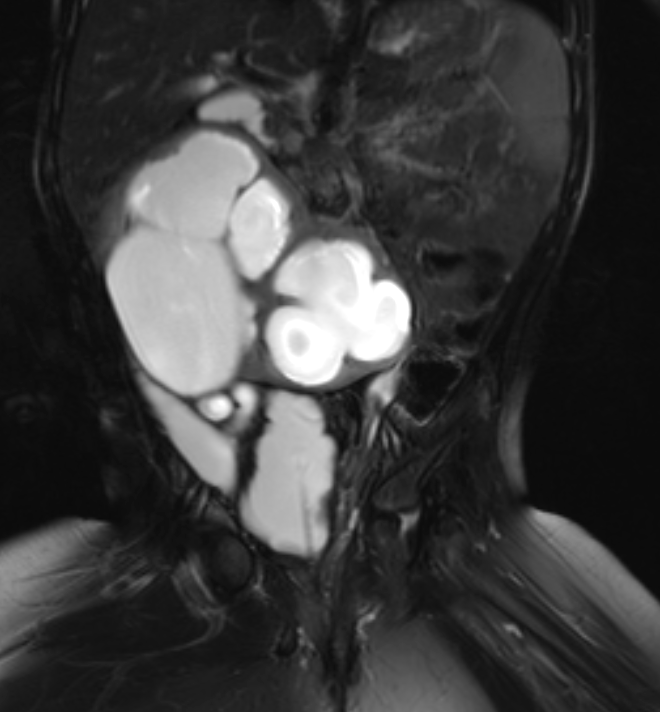
Figura 40 La imagen de RM sensible al líquido con supresión de grasa muestra un riñón en herradura hidronefrótico y una morfología de vejiga neurogénica.
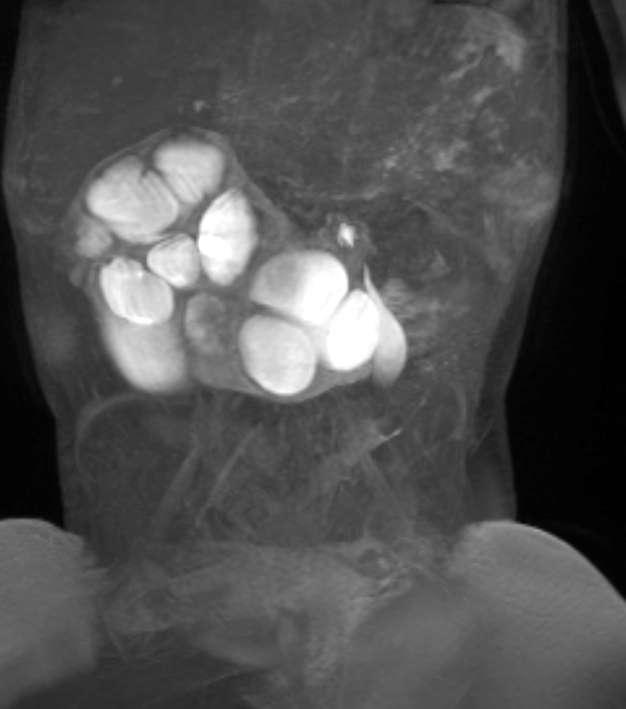
Figura 41 La serie dinámica de RM poscontraste confirma la obstrucción del riñón en herradura.
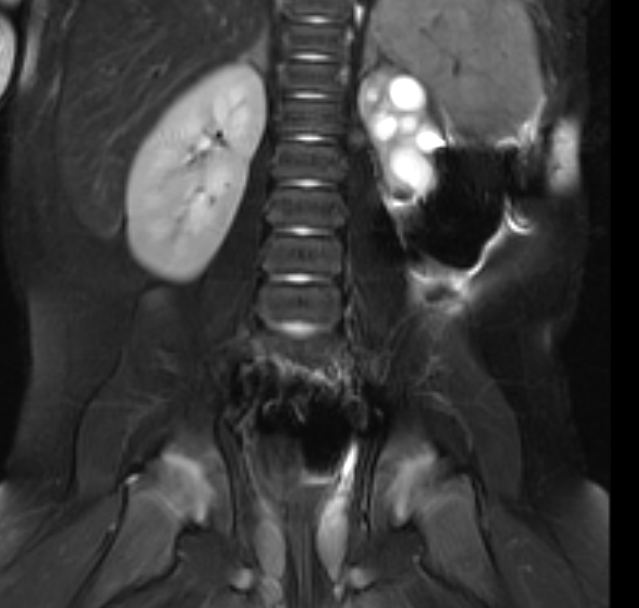
Figura 42 Imagen de RM sensible al líquido con supresión de grasa muestra un pequeño riñón izquierdo displásico.
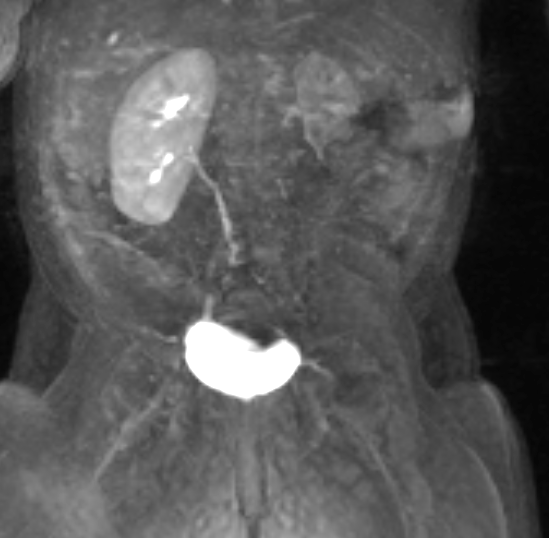
Figura 43 La serie dinámica de RM poscontraste muestra realce tardío y mínimo del riñón displásico, lo que indica función limitada.
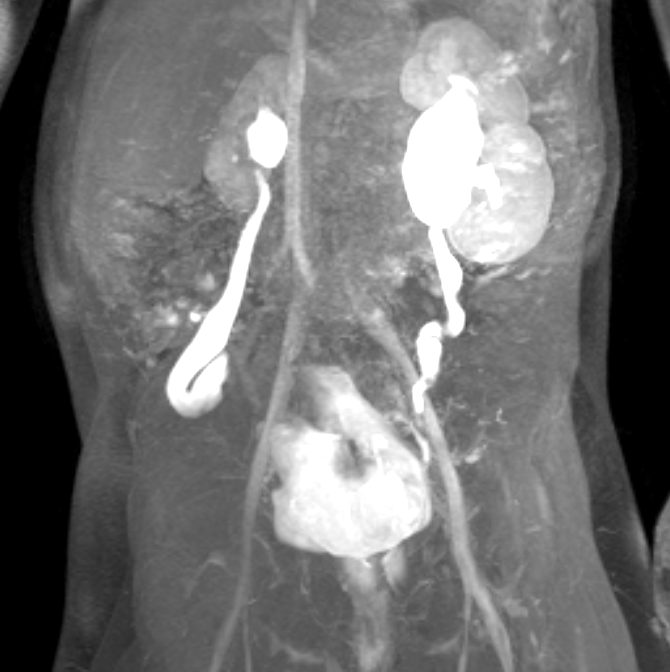
Figura 44 La serie dinámica de RM tras la administración de contraste muestra un riñón derecho pequeño en un paciente con síndrome de Prune-Belly y reflujo vesicoureteral bilateral.
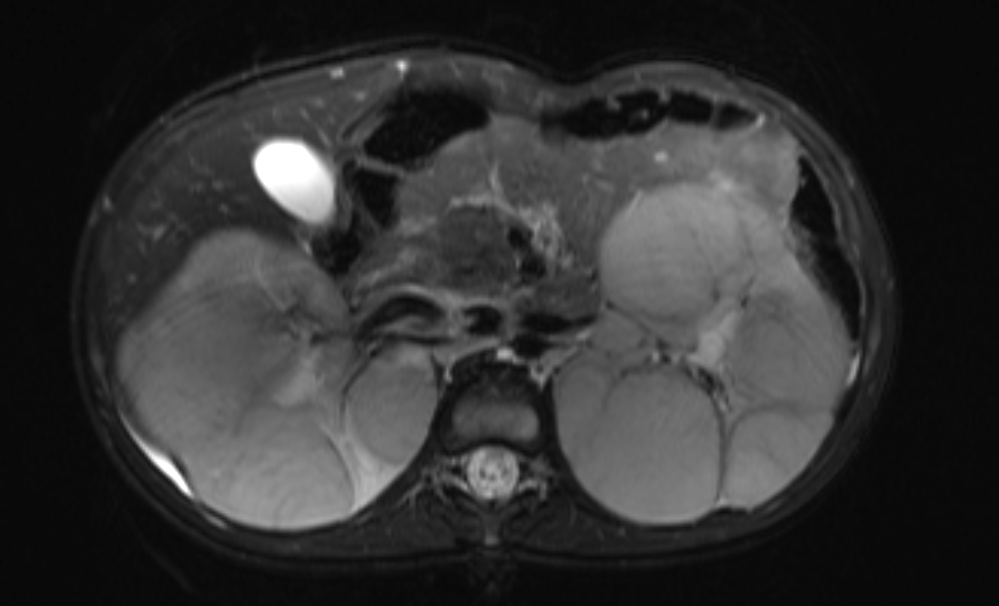
Figura 45 Imagen de RM sensible al líquido con supresión de grasa que muestra reemplazo del parénquima renal por múltiples masas en un paciente con nefroblastomatosis.
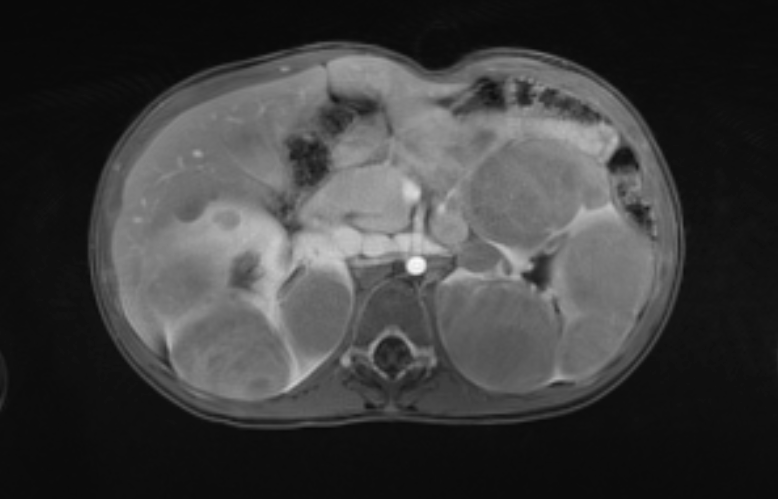
Figura 46 Las imágenes de RM poscontraste muestran las características de realce de las masas.
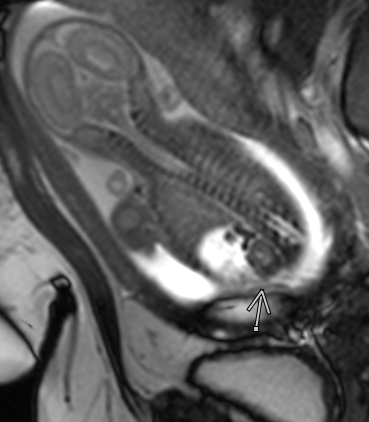
Figura 47 Vista de un riñón y glándula suprarrenal normales en un feto con ascitis
Intervención en urología pediátrica
Los procedimientos intervencionistas son fundamentales para el manejo de las afecciones urológicas pediátricas.23,24 La subespecialidad de radiología pediátrica es distinta tanto de la radiología pediátrica como de la radiología intervencionista de adultos, y los profesionales de IR pediátrica siguen siendo pocos en número, a pesar del crecimiento de los últimos quince años.25
Drenaje de las vías urinarias secundario a obstrucción/nefrostomías
- Debe reservarse para pacientes en los que no sea posible el abordaje de drenaje retrógrado
- La preparación preprocedimental incluiría la evaluación del riesgo de sangrado y de infección
- Profilaxis antibiótica
- Acceso al sistema colector renal ya sea mediante ecografía, fluoroscopia o tomografía computarizada
- Complicaciones
- Hemorragia
- Sepsis
- Lesión de órgano
- Lesión torácica (es decir, neumotórax)
Nefrolitotomía percutánea
- Reservado para pacientes que no son candidatos a la ureteroscopia o a la litotricia por ondas de choque
- Procedimiento en dos pasos
- Acceso al sistema colector por el radiólogo utilizando ecografía/fluoroscopia
- Extracción del cálculo por el urólogo
Otras intervenciones
- Colocación de stent ureteral
- Dilatación ureteral secundaria a estenosis
- Biopsia de tumores renales
- Embolización de tumores renales o hemorragia renal
Lecturas recomendadas
- Brown BP, Simoneaux SF, Dillman JR, Rigsby CK, Iyer RS, Alazraki AL, et al.. ACR Appropriateness Criteria® Antenatal Hydronephrosis–Infant. J Am Coll Radiol 2020; 17 (11): S367–s379. DOI: 10.1016/j.jacr.2020.09.017.
- Chung EM, Soderlund KA, Fagen KE. Imaging of the Pediatric Urinary System. Radiol Clin North Am 2017; 55 (2): 337–357. DOI: 10.1016/j.rcl.2016.10.010.
- Dillman JR, Rigsby CK, Iyer RS, Alazraki AL, Anupindi SA, Brown BP, et al.. ACR Appropriateness Criteria® Hematuria-Child. J Am Coll Radiol 2018; 15 (5): S91–s103. DOI: 10.1016/j.jacr.2018.03.022.
- Dillman JR, Trout AT, Smith EA. MR urography in children and adolescents: techniques and clinical applications. Abdom Radiol (NY) 2016; 41 (6): 1007–1019. DOI: 10.1007/s00261-016-0669-z.
- Duran C, Beltrán VP, González A, Gómez C, Riego Jdel. Contrast-enhanced Voiding Urosonography for Vesicoureteral Reflux Diagnosis in Children. Radiographics 2017; 37 (6): 1854–1869. DOI: 10.1148/rg.2017170024.
- Chow JS, Koning JL, Back SJ, Nguyen HT, Phelps A, Darge K. Classification of pediatric urinary tract dilation: the new language. Pediatric Radiology 2017; 47: 1109–1115.
Referencias
- Strauss KJ, Goske MJ, Kaste SC, Bulas D, Frush DP, Butler P, et al.. Image Gently: Ten Steps You Can Take to Optimize Image Quality and Lower CT Dose for Pediatric Patients. AJR Am J Roentgenol 1976; 194 (4): 868–873. DOI: 10.2214/ajr.09.4091.
- Dillman JR, Rigsby CK, Iyer RS, Alazraki AL, Anupindi SA, Brown BP, et al.. ACR Appropriateness Criteria® Hematuria-Child. J Am Coll Radiol 2018; 15 (5): S91–s103. DOI: 10.1016/j.jacr.2018.03.022.
- Goodman TR, Mustafa A, Rowe E. Pediatric CT radiation exposure: where we were, and where we are now. Pediatr Radiol 2019; 49 (4): 469–478. DOI: 10.1007/s00247-018-4281-y.
- Kuebker J, Shuman J, Hsi RS, Herrell SD, Miller NL. Radiation From Kidney-Ureter-Bladder Radiographs Is Not Trivial. Urology 2019; 125: 46–49. DOI: 10.1016/j.urology.2018.11.035.
- Dong S-Z, Zhu M, Bulas D. Techniques for minimizing sedation in pediatric MRI. J Magn Reson Imaging 2019; 50 (4): 1047–1054. DOI: 10.1002/jmri.26703.
- Drugs ACRC on, Media C. ACR Manual On Contrast Media. American College of Radiology; 2022.
- Brown BP, Simoneaux SF, Dillman JR, Rigsby CK, Iyer RS, Alazraki AL, et al.. ACR Appropriateness Criteria® Antenatal Hydronephrosis–Infant. J Am Coll Radiol 2020; 17 (11): S367–s379. DOI: 10.1016/j.jacr.2020.09.017.
- Karmazyn BK, Alazraki AL, Anupindi SA, Dempsey ME, Dillman JR, Dorfman SR, et al.. ACR Appropriateness Criteria® Urinary Tract Infection–Child. J Am Coll Radiol 2018; 14 (5): S362–s371. DOI: 10.1016/j.jacr.2017.02.028.
- Kalish JM, Doros L, Helman LJ, Hennekam RC, Kuiper RP, Maas SM, et al.. Surveillance Recommendations for Children with Overgrowth Syndromes and Predisposition to Wilms Tumors and Hepatoblastoma. Clin Cancer Res 2017; 23 (13): e115–e122. DOI: 10.1158/1078-0432.ccr-17-0710.
- Wang CL, Aryal B, Oto A, Allen BC, Akin O, Alexander LF, et al.. ACR Appropriateness Criteria® Acute Onset of Scrotal Pain-Without Trauma, Without Antecedent Mass. J Am Coll Radiol 2019; 16 (5): S38–s43. DOI: 10.1016/j.jacr.2019.02.016.
- Rerksuppaphol S, Barnes G. Guidelines for Evaluation and Treatment of Gastroesophageal Reflux In Infants and Children: Recommendations of the North American Society for Pediatric Gastroenterology and Nutrition. J Pediatr Gastroenterol Nutr 2006; 35 (4): 583. DOI: 10.1097/00005176-200210000-00024.
- Chung EM, Soderlund KA, Fagen KE. Imaging of the Pediatric Urinary System. Radiol Clin North Am 2017; 55 (2): 337–357. DOI: 10.1016/j.rcl.2016.10.010.
- Barnewolt CE, Acharya PT, Aguirre Pascual E, Back SJ, Beltrán Salazar VP, Chan PKJ, et al.. Contrast-enhanced voiding urosonography part 2: urethral imaging. Pediatr Radiol 2021; 51 (12): 2368–2386. DOI: 10.1007/s00247-021-05116-6.
- Duran C, Beltrán VP, González A, Gómez C, Riego Jdel. Contrast-enhanced Voiding Urosonography for Vesicoureteral Reflux Diagnosis in Children. Radiographics 2017; 37 (6): 1854–1869. DOI: 10.1148/rg.2017170024.
- Sheehy N, Tetrault TA, Zurakowski D, Vija AH, Fahey FH, Treves ST. Pediatric 99mTc-DMSA SPECT Performed by Using Iterative Reconstruction with Isotropic Resolution Recovery: Improved Image Quality and Reduced Radiopharmaceutical Activity. Radiology 2009; 251 (2): 511–516. DOI: 10.1148/radiol.2512081440.
- Wood LN, Souders CE, Freedman AL. Is a Reassuring MAG-3 Diuretic Renal Scan Really Reassuring? Curr Urol 2014; 8 (4): 178–182. DOI: 10.1159/000365713.
- Rodger F, Roditi G, Aboumarzouk OM. Diagnostic Accuracy of Low and Ultra-Low Dose CT for Identification of Urinary Tract Stones: A Systematic Review. Urol Int 2018; 100 (4): 375–385. DOI: 10.1159/000488062.
- Voss SD. Staging and following common pediatric malignancies: MRI versus CT versus functional imaging. Pediatr Radiol 2018; 48 (9): 1324–1336. DOI: 10.1007/s00247-018-4162-4.
- Darge K, Anupindi SA, Jaramillo D. MR Imaging of the Abdomen and Pelvis in Infants, Children, and Adolescents. Radiology 2011; 261 (1): 12–29. DOI: 10.1148/radiol.11101922.
- Calvo-Garcia MA, Kline-Fath BM, Levitt MA, Lim F-Y, Linam LE, Patel MN, et al.. Fetal MRI clues to diagnose cloacal malformations. Pediatr Radiol 2011; 41 (9): 1117–1128. DOI: 10.1007/s00247-011-2020-8.
- Hwang JY, Shin JH, Lee YJ, Yoon HM, Cho YA, Kim KS. Percutaneous nephrostomy placement in infants and young children. Diagn Interv Imaging 2018; 99 (3): 157–162. DOI: 10.1016/j.diii.2017.07.002.
- Sweed Y, Singer-Jordan J, Papura S, Loberant N, Yulevich A. Use of angiographic embolization in pediatric abdominal trauma-induced solid organ injuries. Ulus Travma Acil Cerrahi Derg 2016; 8 (11): 65–68. DOI: 10.5505/tjtes.2018.00056.
- Kaufman CS, James CA, Harned RK, Connolly BL, Roebuck DJ, Cahill AM, et al.. Pediatric interventional radiology workforce survey: 10-year follow-up. Pediatr Radiol 2017; 47 (6): 651–656. DOI: 10.1007/s00247-017-3796-y.
- Pierce CB, Muñoz A, Ng DK, Warady BA, Furth SL, Schwartz GJ. Age- and sex-dependent clinical equations to estimate glomerular filtration rates in children and young adults with chronic kidney disease. Kidney Int 2021; 99 (4): 948–956. DOI: 10.1016/j.kint.2020.10.047.
- Dillman JR, Trout AT, Smith EA. MR urography in children and adolescents: techniques and clinical applications. Abdom Radiol (NY) 2016; 41 (6): 1007–1019. DOI: 10.1007/s00261-016-0669-z.
- Chow JS, Koning JL, Back SJ, Nguyen HT, Phelps A, Darge K. Classification of pediatric urinary tract dilation: the new language. Pediatric Radiology 2017; 47: 1109–1115.
Última actualización: 2025-09-21 13:35