19: Vejiga neurógena
Este capítulo durará aproximadamente 14 minutos para leer.
Introducción
El disrafismo espinal congénito representa la gran mayoría de los casos de vejiga e intestino neurogénicos pediátricos.1 Otras causas de disfunción neurogénica que comprometen la médula espinal incluyen la agenesia sacra, la médula anclada, las malformaciones cloacales y las lesiones de la médula espinal. Los niños con anomalías del sistema nervioso central, como la diplejía espástica (parálisis cerebral), también pueden presentar vejiga neurogénica.
Una dinámica vesical deficiente puede conducir a enfermedad renal crónica y a una multitud de posibles secuelas.2 El manejo de la vejiga neurógena incluye lograr un almacenamiento urinario a baja presión y proporcionar continencia urinaria, preservando la función del tracto urinario superior. El tratamiento óptimo requiere un equipo de atención multidisciplinario para prevenir y manejar las posibles secuelas que pueden afectar la función, la calidad de vida y la supervivencia. Dado que la espina bífida representa la mayoría de los casos de vejiga neurógena, será el foco del presente capítulo.
Embriología
Los defectos del tubo neural (DTN) son malformaciones del cerebro y la médula espinal resultantes de la falta de cierre del tubo neural durante la tercera y cuarta semanas del desarrollo intrauterino.3 La espina bífida ocurre cuando la médula espinal queda expuesta y/o protruye hacia la superficie junto con las meninges a través de un defecto de la pared vertebral. El desarrollo del canal raquídeo comienza el día 18 de la gestación y se completa hacia el día 35, cerrándose en dirección caudal desde el extremo cefálico del cuerpo. El disrafismo espinal es atribuible a un desarrollo anormal del mesodermo craneal.3 La falta de crecimiento del mesodermo sobre la médula espinal en desarrollo da lugar a una lesión abierta, que se observa con mayor frecuencia en el área lumbosacra (Tabla 1). La médula espinal expuesta y sus raíces nerviosas, que pueden protruir hacia el saco de meningocele, y la tensión sobre la médula espinal a medida que se desplaza cranealmente por el canal con la elongación del feto (de L2, L3 en la gestación media a tardía, a L1 al nacer), contribuyen al cuadro variable de lesión neurológica del tracto urinario inferior y de las extremidades inferiores.4 La hidrocefalia y la malformación de Arnold-Chiari tipo II (una combinación de mielomeningocele y herniación de las amígdalas cerebelosas) se asocian frecuentemente con la espina bífida.5
Tabla 1 Nivel espinal del mielomeningocele.
| Ubicación | Incidencia |
|---|---|
| Cervical-Torácica alta | 2% |
| Torácica baja | 5% |
| Lumbar | 26% |
| Lumbosacra | 47% |
| Sacra | 20% |
Epidemiología
La espina bífida es el defecto del tubo neural más común, que ocurre en aproximadamente 1 de cada 3,000 nacidos vivos en los Estados Unidos y 1 por 1,000 nacidos vivos a nivel mundial.6,7 Aunque la incidencia de espina bífida disminuyó significativamente tras la introducción de la fortificación obligatoria de productos de granos enriquecidos con ácido fólico en los Estados Unidos, la carga poblacional de la espina bífida persiste tanto en la prevalencia al nacimiento como en los resultados a largo plazo dispares.8,9,10 Se ha demostrado que las diferencias en la raza/etnia materna afectan la prevalencia de espina bífida, con las madres hispanas presentando una prevalencia mayor que las mujeres caucásicas y negras.11 El predominio por sexo difiere según el país; en los Estados Unidos, se considera que la espina bífida es más prevalente en niñas que en niños.12
Patogénesis
La espina bífida suele ser visible al nacer como tejido neural expuesto con o sin un saco que sobresale en el sitio de la lesión. El mielomeningocele se produce cuando la médula espinal protruye desde el canal espinal hacia un saco lleno de líquido resultante del cierre incompleto del tubo neural primario (Figura 1) La extensión y la gravedad de los déficits neurológicos dependen de la localización de la lesión a lo largo del neuroeje - según el nivel, la interrupción de la médula espinal en el sitio del defecto causa parálisis de las piernas, incontinencia urinaria y fecal, anestesia cutánea y anomalías de las caderas, las rodillas y los pies.13,14 Existe un impacto variable sobre la inervación somática, parasimpática y simpática del detrusor y de los músculos esfintéricos, que afecta la capacidad de la vejiga para almacenar y vaciar orina - el mielomeningocele casi siempre se asocia con vejiga neurógena. La mala dinámica vesical puede luego provocar enfermedad renal crónica y una multitud de posibles secuelas.2
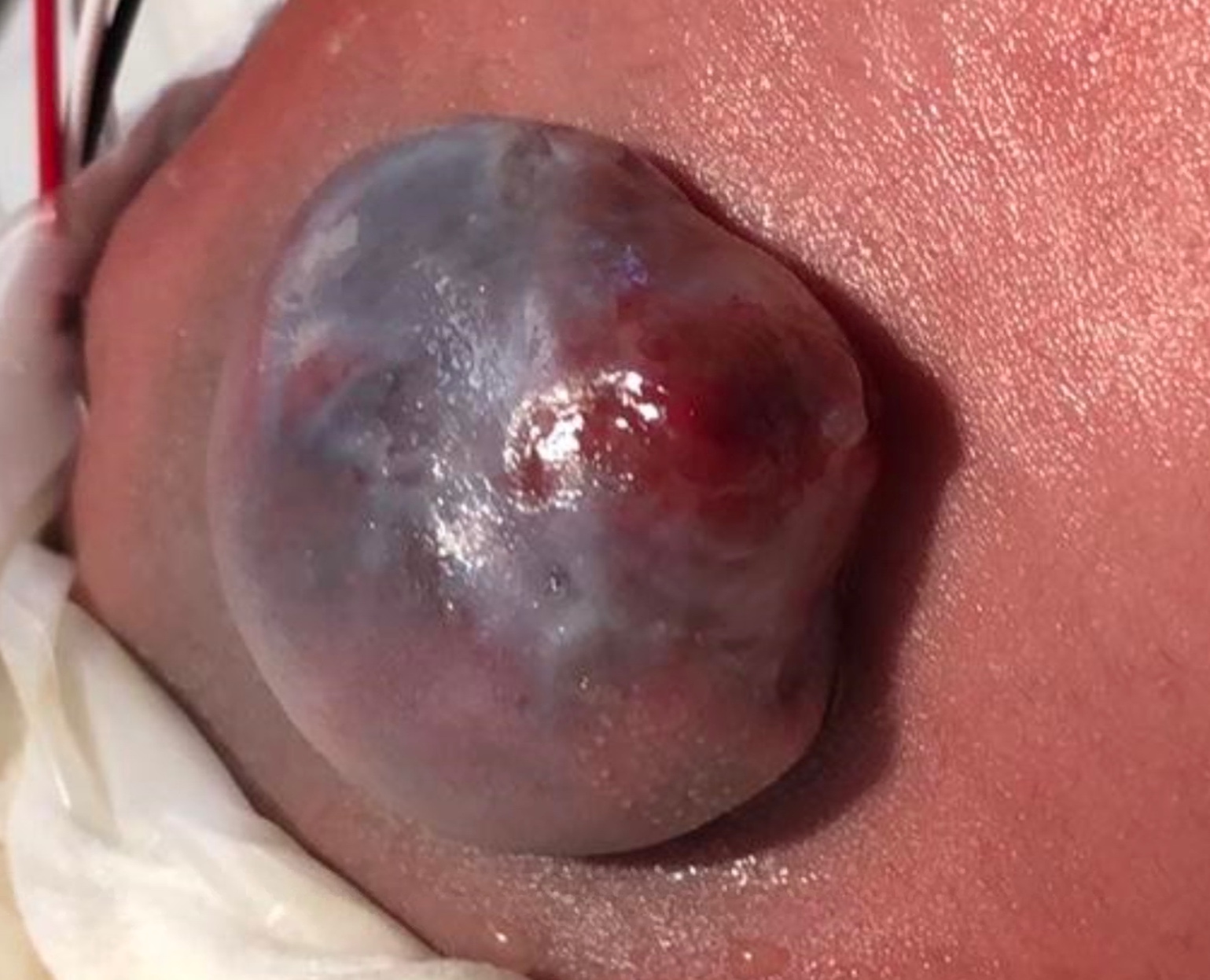
Figura 1 Mielomeningocele abierto.
El mielomeningocele suele asociarse con una malformación de Chiari tipo II del encéfalo posterior, ventriculomegalia e hidrocefalia.15 La malformación de Chiari tipo II es el descenso del vermis cerebeloso dentro del canal vertebral cervical.5 Esto da lugar a la elongación del tronco encefálico y la obliteración del cuarto ventrículo, lo que conduce a la obstrucción de la circulación del líquido cefalorraquídeo y al desarrollo de hidrocefalia en el 90% de los pacientes.5,13 El tratamiento de la hidrocefalia se requiere en aproximadamente el 82% de los casos e implica el drenaje del líquido cefalorraquídeo hacia la cavidad peritoneal u otra cavidad corporal mediante una derivación.16
Evaluación y diagnóstico
Si bien la atención óptima de la disfunción vesical relacionada con la espina bífida sigue sin conocerse, ha surgido una tendencia reciente hacia un manejo proactivo en lugar de reactivo de los niños con vejiga neurógena.17 La observación implica un seguimiento periódico mediante pruebas no invasivas, incluida la ecografía renovesical, para evaluar la dilatación/deterioro del tracto urinario superior. Sin embargo, muchos urólogos pediátricos abogan ahora por una evaluación completa del tracto urinario inferior en la lactancia con inicio de tratamiento profiláctico ya sea al nacer o si hay signos de obstrucción del tracto de salida vesical y/o presión vesical elevada durante el llenado o la micción en la urodinamia inicial.18,19,20
En 2012, los Centros para el Control y la Prevención de Enfermedades (CDC) convocaron un grupo de trabajo de urólogos pediátricos, nefrólogos, epidemiólogos clínicos, metodólogos, defensores de la comunidad y personal de los CDC para desarrollar un protocolo estandarizado que optimizara la atención urológica de los niños con espina bífida desde el período neonatal hasta los 5 años (Figura 2)21 Se obtiene una ecografía renovesical para determinar la dilatación basal del tracto superior.17 Se realizan estudios videourodinámicos antes de los 3 meses de edad – las imágenes fluoroscópicas demuestran reflujo vesicoureteral si está presente, así como contorno vesical irregular/trabeculación (Figura 3)]. La cuestión predominante al nacimiento es si existe disinergia detrusor-esfínter y si el bebé puede vaciar a bajas presiones. La presión de llenado del detrusor elevada, la disinergia detrusor-esfínter o presiones de punto de fuga elevadas (>40 cm H20) pueden resultar en daño del tracto superior y justifican una intervención temprana con cateterismo intermitente limpio (CIC) y anticolinérgicos.22,23,24,25 Con un enfoque proactivo, a los lactantes se les pauta cateterismo intermitente limpio (CIC) cada 6 horas – la frecuencia de los cateterismos puede disminuirse si los volúmenes son de forma consistente ≤30 mL; un componente clave es que la familia aprenda a realizar los cateterismos antes del alta.21,26
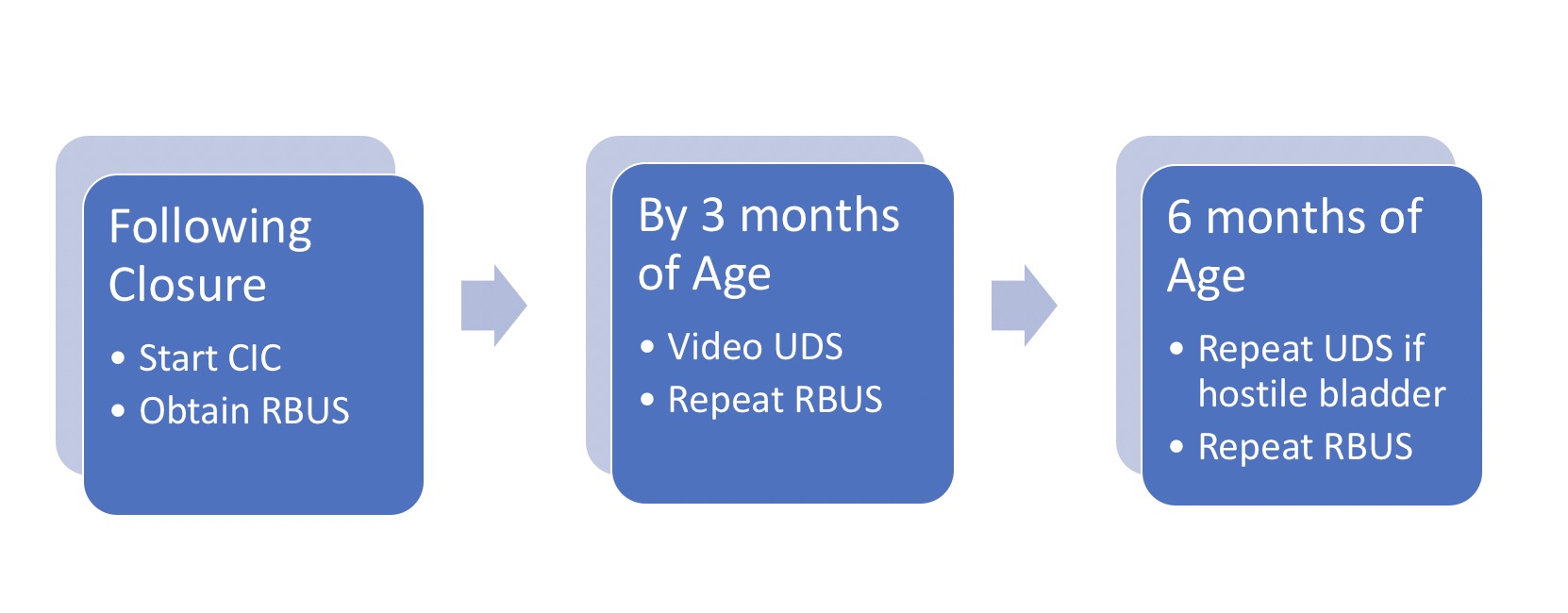
Figura 2 Protocolo para el manejo inicial del lactante con espina bífida. Tenga en cuenta que a todos los recién nacidos se les inicia CIC cada 6 horas según el protocolo; el intervalo de cateterismo puede ampliarse y luego suspenderse si los residuales son de manera consistente ≤30 mL y la hidronefrosis en la RBUS ≤ grado 2. *Adaptado de.21
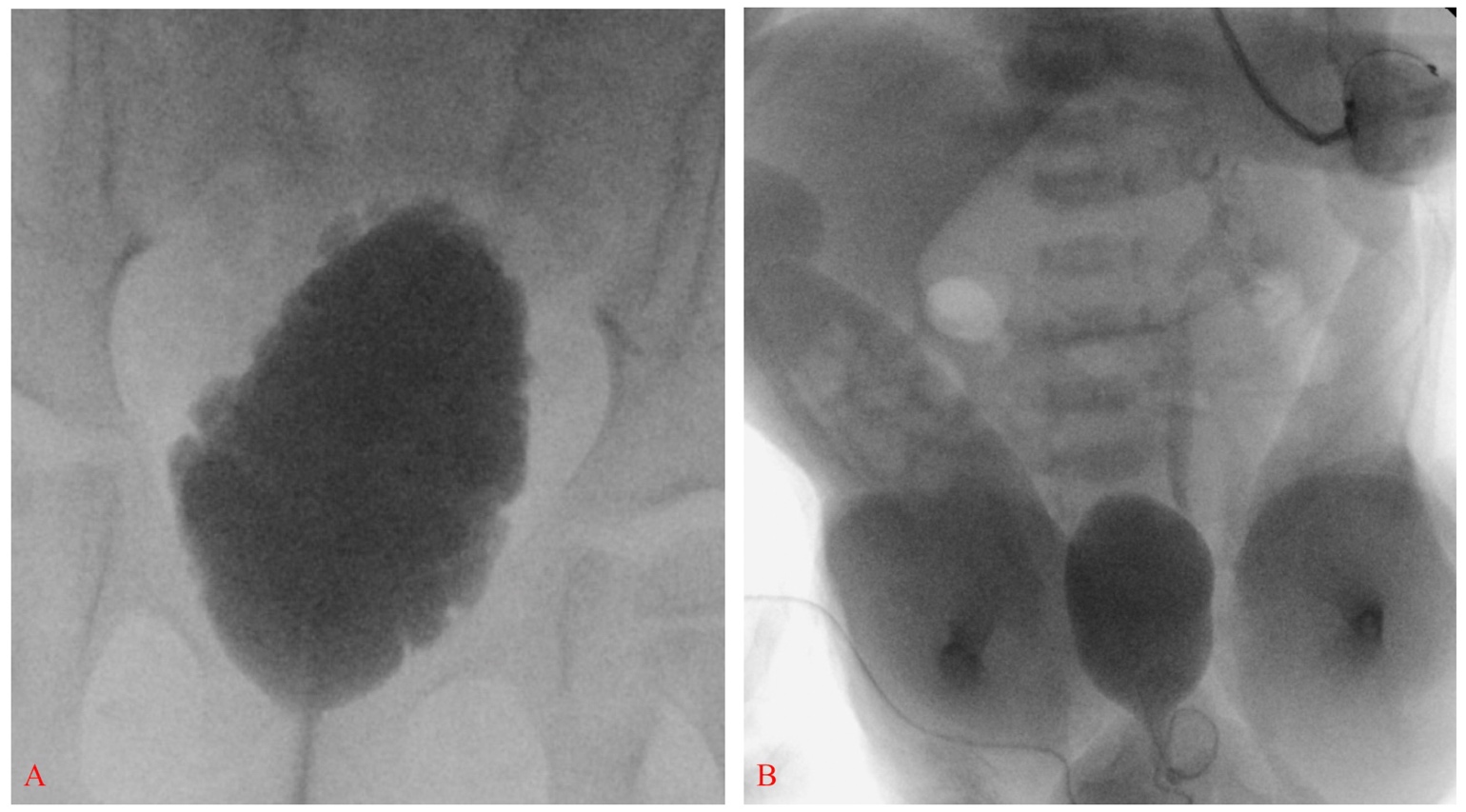
Figura 3 Vejiga alargada con contorno irregular y trabeculación compatible con vejiga neurogénica (A). Vejiga de paredes lisas con reflujo vesicoureteral izquierdo grado III en un recién nacido con espina bífida; obsérvese también pie equinovaro bilateral y flexión severa de cadera (B).
Opciones de tratamiento y sus resultados
Los objetivos del manejo de la vejiga neurogénica pediátrica incluyen lograr un almacenamiento vesical a baja presión y proporcionar continencia urinaria, al tiempo que se preserva la función del tracto urinario superior. La incidencia de deterioro del tracto urinario es significativa, reportada en más del 50%, cuando a los niños con potencial de deterioro se les da seguimiento con manejo expectante en lugar de terapia preventiva.20,23,24 La gravedad de la disfunción vesical varía ampliamente y cambia con el tiempo, la evaluación urodinámica temprana es clave para identificar a los niños con mayor riesgo – se recomienda un manejo urológico más agresivo para aquellos individuos con parámetros hostiles.27,28,29
La implementación temprana del CIC y de la terapia anticolinérgica ha demostrado ser ventajosa con el tiempo. El cateterismo intermitente suele iniciarse cada 6 horas, aumentando la frecuencia a cada 4 horas durante la vigilia y añadiendo oxibutinina 0,2 mg/kg/8 horas en aquellos con hallazgos desfavorables en UDS. Los anticolinérgicos son la base del tratamiento para la hiperactividad del detrusor y para reducir las presiones de almacenamiento intravesical en niños con baja distensibilidad del detrusor, lo cual los coloca en riesgo de compromiso renal.30,31 Además de la oxibutinina, existen varios fármacos anticolinérgicos selectivos más nuevos (como solifenacina y tolterodina) diseñados para disminuir los efectos secundarios, ya sea dirigiéndose a subtipos específicos de receptores muscarínicos o modificando los compuestos estructurales para que sean menos propensos a atravesar barreras orgánicas.32 Mirabegrón, un agonista de los receptores adrenérgicos β3, es una opción terapéutica alternativa a los antimuscarínicos, y ha demostrado ser eficaz y bien tolerado en niños y adolescentes con disfunción vesical neurógena.33 Con un drenaje adecuado y el manejo de la hiperactividad del detrusor, la vejiga suele conservar su distensibilidad, expandiéndose a medida que el niño crece y manteniendo un grosor de pared apropiado. La hidronefrosis y el reflujo vesicoureteral se desarrollan en menos del 10% y la continencia social se logra con facilidad en la mayoría de los pacientes sin intervenciones adicionales.20,34,35
La toxina botulínica tipo A (BTX-A) inhibe la liberación del neurotransmisor acetilcolina en la unión neuromuscular y se considera una alternativa para mejorar la continencia y los parámetros urodinámicos de la disfunción vesical neurógena en niños.31 Los estudios han demostrado una mejoría significativa en los parámetros clínicos y urodinámicos, como lo evidencia la continencia completa en aproximadamente del 65% al 87% de los niños y una reducción de la presión detrusora máxima y un aumento de la compliance del detrusor en la mayoría de los tratados.36
La ampliación vesical utilizando intestino delgado o colon representa el método definitivo para crear un órgano de gran capacidad y baja presión para el almacenamiento de orina en un número considerable de niños que no han respondido al tratamiento médico, pero su utilidad puede verse limitada por la morbilidad tanto a corto como a largo plazo.37,38,39 Se realizan procedimientos concomitantes sobre el cuello vesical según esté indicado. La intervención quirúrgica debe individualizarse en función de los hallazgos urodinámicos, antecedentes médicos, edad, destreza manual, así como factores sociales. El clínico debe tener una expectativa razonable de que la familia está preparada para asumir regímenes de cateterización e irrigación posoperatorios. Al menos un tercio de los niños sometidos a ampliación vesical presentarán alguna complicación, incluyendo perforación vesical, formación de cálculos y necesidad de procedimientos adicionales como la revisión del estoma.40,41,42 Las alteraciones metabólicas son una consecuencia conocida de incorporar intestino al tracto urinario, y es necesario monitorizar a los pacientes por desequilibrios metabólicos, incluida la acidosis crónica.43 La malignidad, aunque infrecuente, es una complicación tardía grave de la ampliación y subraya la importancia del seguimiento de por vida en estos pacientes.44,45,46 Cabe señalar que la necesidad de cistoplastia de aumento para mantener una capacidad de almacenamiento adecuada se reduce notablemente de casi el 60% al 16% cuando se compara con los niños seguidos de forma expectante.47
Seguimiento recomendado
- La ecografía renovesical debe realizarse al nacer, cada 3 meses durante el primer año de vida y luego anualmente junto con creatinina sérica, examen físico y evaluación de la presión arterial.21
- Urodinamia basal a los 3 meses; estudio anual a las edades de 1, 2 y 3 años y luego cada dos años durante la infancia (los UDS se realizan con mayor frecuencia si hay parámetros hostiles, para evaluar la respuesta al tratamiento o ante nueva hidroureteronefrosis/incontinencia/infección urinaria recurrente).21,48
- A medida que la población con espina bífida envejece, se debe concienciar a los pacientes, cuidadores y médicos de atención primaria sobre el riesgo basal de cáncer asociado a la vejiga neurogénica y la necesidad de una vigilancia estrecha incluso en ausencia de procedimientos reconstructivos y derivativos que aumentan el riesgo neoplásico en el tracto urinario.46
Conclusiones
La vejiga neurogénica abarca un amplio espectro de enfermedades según el grado de afectación del tracto urinario inferior y la interacción entre la capacidad de almacenamiento vesical y la función del esfínter. La base del manejo actual de la vejiga neurogénica es no quirúrgica, con anticolinérgicos y cateterismo intermitente limpio. Se ha demostrado que el tratamiento proactivo de los niños con espina bífida es eficaz para reducir la necesidad de intervención quirúrgica y el desarrollo de enfermedad renal terminal al minimizar los efectos del reflujo de alta presión sobre el tracto urinario superior. Cuando está indicado, el manejo quirúrgico se adapta a cada paciente, con base en una cuidadosa consideración de los hallazgos urodinámicos, la historia clínica, la edad y la presencia de otra discapacidad.
Puntos clave
- El manejo proactivo reduce las secuelas a largo plazo de la vejiga neurógena
- La presión de llenado del detrusor elevada, la disinergia detrusor-esfínter o las presiones de punto de fuga elevadas (>40 cm H20) pueden causar daño del tracto urinario superior
- La farmacoterapia y el CIC son las piedras angulares del manejo
Lecturas sugeridas
- Bauer SB. Neurogenic bladder: etiology and assessment. Pediatr Nephrol 2008; 23 (4): 541–551. DOI: 10.1007/s00467-008-0764-7.
- Routh JC, Cheng EY, Austin JC, Baum MA, Gargollo PC, Grady RW, et al.. Design and Methodological Considerations of the Centers for Disease Control and Prevention Urologic and Renal Protocol for the Newborn and Young Child with Spina Bifida. J Urol 2016; 196 (6): 1728–1734. DOI: 10.1016/j.juro.2016.07.081.
- Rawashdeh YF, Austin P, Siggaard C, Bauer SB, Franco I, Jong TP de, et al.. International children’s continence society’s recommendations for therapeutic intervention in congenital neuropathic bladder and bowel dysfunction in children. Neurourol Urodyn 2012; 31 (5): 615–620. DOI: 10.1002/nau.22248.
Referencias
- Bauer SB. Neurogenic bladder: etiology and assessment. Pediatr Nephrol 2008; 23 (4): 541–551. DOI: 10.1007/s00467-008-0764-7.
- OAKESHOTT PIPPA, HUNT GILLIANM, POULTON ALISON, REID FIONA. Expectation of life and unexpected death in open spina bifida: a 40-year complete, non-selective, longitudinal cohort study. Dev Med Child Neurol 2010; 52 (8): 749–753. DOI: 10.1111/j.1469-8749.2009.03543.x.
- Hadžagić-Ćatibušić F, Maksić H, Užičanin S, Heljić S, Zubčević S, Merhemić Z, et al.. Congenital Malformations of the Central Nervous System: Clinical Approach. Bosn J Basic Med Sci 2008; 8 (4): 356–360. DOI: 10.17305/bjbms.2008.2897.
- Blaivas JG, Labib KL, Bauer SB, Retik AB. Changing Concepts in the Urodynamic Evaluation of Children. J Urol 1977; 117 (6): 778–781. DOI: 10.1016/s0022-5347(17)58623-1.
- Northrup H, Volcik KA. Spina bifida and other neural tube defects. Curr Probl Pediatr 2000; 30 (10): 317–332. DOI: 10.1067/mpp.2000.112052.
- Parker SE, Mai CT, Canfield MA, Rickard R, Wang Y, Meyer RE, et al.. Updated national birth prevalence estimates for selected birth defects in the United States, 2004-2006. Birth Defects Res A Clin Mol Teratol 2010; 88 (12): 1008–1016. DOI: 10.1002/bdra.20735.
- Ryznychuk MO, Kryvchanska MI, Lastivka IV. Teaching children with spina bifida. The Child with Spina Bifida 2018; 71 (339-344): 202–236. DOI: 10.4324/9781315656861-9.
- Honein MA, Paulozzi LJ, Mathews TJ. Impact of Folic Acid Fortification of the US Food Supply on the Occurrence of Neural Tube Defects–Correction. Jama 2001; 286 (18): 2236. DOI: 10.1001/jama.286.18.2236.
- Williams LJ, Rasmussen SA, Flores A, Kirby RS, Edmonds LD. Decline in the Prevalence of Spina Bifida and Anencephaly by Race/Ethnicity: 1995–2002. Pediatrics 2005; 116 (3): 580–586. DOI: 10.1542/peds.2005-0592.
- Bestwick JP, Huttly WJ, Morris JK, Wald NJ. Prevention of Neural Tube Defects: A Cross-Sectional Study of the Uptake of Folic Acid Supplementation in Nearly Half a Million Women. PLoS One 2014; 9 (2): e89354. DOI: 10.1371/journal.pone.0089354.
- Agopian AJ, Canfield MA, Olney RS, Lupo PJ, Ramadhani T, Mitchell LE, et al.. Spina bifida subtypes and sub-phenotypes by maternal race/ethnicity in the National Birth Defects Prevention Study. Am J Med Genet A 2012; 158a (1): 109–115. DOI: 10.1002/ajmg.a.34383.
- Mitchell LE, Adzick NS, Melchionne J, Pasquariello PS, Sutton LN, Whitehead AS. Spina bifida. Lancet 2004; 364 (9448): 1885–1895. DOI: 10.1016/s0140-6736(04)17445-x.
- Jobe AH. Fetal Surgery for Myelomeningocele. N Engl J Med 2002; 347 (4): 230–231. DOI: 10.1056/nejmp020073.
- Mohd-Zin SW, Marwan AI, Abou Chaar MK, Ahmad-Annuar A, Abdul-Aziz NM. Spina Bifida: Pathogenesis, Mechanisms, and Genes in Mice and Humans. Scientifica (Cairo) 2017; 2017: 1–29. DOI: 10.1155/2017/5364827.
- Stevenson KL. Chiari Type II malformation: past, present, and future. Neurosurg Focus 2004; 16 (2): 1–7. DOI: 10.3171/foc.2004.16.2.6.
- Adzick NS, Thom EA, Spong CY. A Randomized Trial of Prenatal versus Postnatal Repair of Myelomeningocele. Yearbook of Pediatrics 2011; 2012 (11): 404–406. DOI: 10.1016/j.yped.2011.06.029.
- Wang H-HS, Lloyd JC, Wiener JS, Routh JC. Nationwide Trends and Variations in Urological Surgical Interventions and Renal Outcome in Patients with Spina Bifida. J Urol 2016; 195 (4 Part 2): 1189–1195. DOI: 10.1016/j.juro.2015.11.033.
- KURZROCK ERICA, POLSE SANFORD. Renal Deterioration In Myelodysplastic Children: Urodynamic Evaluation And Clinical Correlates. J Urol 1998; 159 (5): 1657–1661. DOI: 10.1097/00005392-199805000-00084.
- HOPPS CARINV, KROPP KENNETHA. Preservation of Renal Function in Children With Myelomeningocele Managed With Basic Newborn Evaluation And Close Followup. J Urol 2003; 169: 305–308. DOI: 10.1097/00005392-200301000-00092.
- Wu H-Y, Baskin L, Kogan BA. Neurogenic Bladder Dysfunction Due to Myelomeningocele: Neonatal Versus Childhood Treatment. J Urol 1997; 157 (6): 2295–2297. DOI: 10.1016/s0022-5347(01)64766-9.
- Routh JC, Cheng EY, Austin JC, Baum MA, Gargollo PC, Grady RW, et al.. Design and Methodological Considerations of the Centers for Disease Control and Prevention Urologic and Renal Protocol for the Newborn and Young Child with Spina Bifida. J Urol 2016; 196 (6): 1728–1734. DOI: 10.1016/j.juro.2016.07.081.
- Mcguire EJ, Wang C-C, Usitalo H, Savastano J. Modified Pubovaginal Sling in Girls with Myelodysplasia. J Urol 1986; 135 (1): 94–96. DOI: 10.1016/s0022-5347(17)45528-5.
- Bauer SB, Hallet M, Khoshbin S. Predictive value of urodynamic evaluation in newborns with myelodysplasia. Jama 1984; 252 (5): 650–652. DOI: 10.1001/jama.252.5.650.
- Ami Sidi A, Dykstra DD, Gonzalez R. The Value of Urodynamic Testing in the Management of Neonates with Myelodysplasia: A Prospective Study. J Urol 1986; 135 (1): 90–93. DOI: 10.1016/s0022-5347(17)45527-3.
- Perez LM, Khoury J, Webster GD. The Value of Urodynamic Studies in Infants Less than 1 Year Old with Congenital Spinal Dysraphism. J Urol 1992; 148 (2 Part 2): 584–587. DOI: 10.1016/s0022-5347(17)36660-0.
- BASKIN LS, KOGAN BA, BENARD F. Treatment of Infants with Neurogenic Bladder Dysfunction using Anticholinergic Drugs and Intermittent Catheterisation. Br J Urol 1990; 66 (5): 532–534. DOI: 10.1111/j.1464-410x.1990.tb15004.x.
- Joseph DB, Bauer SB, Colodny AH, Mandell J, Retik AB. Clean, Intermittent Catheterization of Infants With Neurogenic Bladder. Pediatrics 1989; 84 (1): 78–82. DOI: 10.1542/peds.84.1.78.
- Tanaka ST, Yerkes EB, Routh JC, Tu DD, Austin JC, Wiener JS, et al.. Urodynamic characteristics of neurogenic bladder in newborns with myelomeningocele and refinement of the definition of bladder hostility: Findings from the UMPIRE multi-center study. J Pediatr Urol 2021; 17 (5): 726–732. DOI: 10.1016/j.jpurol.2021.04.019.
- Snow-Lisy DC, Yerkes EB, Cheng EY. Update on Urological Management of Spina Bifida from Prenatal Diagnosis to Adulthood. J Urol 2015; 194 (2): 288–296. DOI: 10.1016/j.juro.2015.03.107.
- Ghoniem GM, Roach MB, Lewis VH, Harmon EP. The Value of Leak Pressure and Bladder Compliance in the Urodynamic Evaluation of Meningomyelocele Patients. J Urol 1990; 144 (6): 1440–1442. DOI: 10.1016/s0022-5347(17)39761-6.
- Galloway NTM, Mekras JA, Helms M, Webster GD. An Objective Score to Predict Upper Tract Deterioration in Myelodysplasia. J Urol 1991; 145 (3): 535–537. DOI: 10.1016/s0022-5347(17)38389-1.
- Andersson K-E, Chapple CR, Cardozo L, Cruz F, Hashim H, Michel MC, et al.. Pharmacological treatment of overactive bladder: report from the International Consultation on Incontinence. Curr Opin Urol 2009; 19 (4): 380–394. DOI: 10.1097/mou.0b013e32832ce8a4.
- Rawashdeh YF, Austin P, Siggaard C, Bauer SB, Franco I, Jong TP de, et al.. International children’s continence society’s recommendations for therapeutic intervention in congenital neuropathic bladder and bowel dysfunction in children. Neurourol Urodyn 2012; 31 (5): 615–620. DOI: 10.1002/nau.22248.
- Franco I, Hoebeke P. Baka-Ostrowska M, et al. Long-term efficacy and safety of solifenacin in pediatric patients aged 6 months to 18 years with neurogenic detrusor overactivity: results from two phase 3 prospective open-label studies. J Pediatr Urol 2019. 6 (2): 80 1–180 8. DOI: 10.1016/j.jpurol.2019.12.012.
- REINBERG Y, CROCKER J, WOLPERT J, VANDERSTEEN D. Therapeutic Efficacy Of Extended Release Oxybutynin Chloride, And Immediate Release And Long Acting Tolterodine Tartrate In Children With Diurnal Urinary Incontinence. J Urol 2003; 169 (317-319): 317–319. DOI: 10.1097/00005392-200301000-00095.
- Baka-Ostrowska M, Bolong DT, Persu C, Tøndel C, Steup A, Lademacher C, et al.. Efficacy and safety of mirabegron in children and adolescents with neurogenic detrusor overactivity: An open-label, phase 3, dose-titration study. Neurourol Urodyn 2021; 40 (6): 1490–1499. DOI: 10.1002/nau.24657.
- Edelstein RA, Bauer SB, Kelly MD. The long-term urologic response of neonates with myelodysplasia treated proactively with intermittent catheterization and anticholinergic therapy. J Pediatr Surg 1995; 31 (3): 455–456. DOI: 10.1016/s0022-3468(96)90810-6.
- Karsenty G, Denys P, Amarenco G, De Seze M, Gamé X, Haab F, et al.. Botulinum Toxin A (Botox®) Intradetrusor Injections in Adults with Neurogenic Detrusor Overactivity/Neurogenic Overactive Bladder: A Systematic Literature Review. Eur Urol 2009; 53 (2): 275–287. DOI: 10.1016/j.eururo.2007.10.013.
- Lemelle JL, Guillemin F, Aubert D, Guys JM, Lottmann H, Lortat-Jacob S, et al.. A Multicenter Evaluation of Urinary Incontinence Management and Outcome in Spina Bifida. J Urol 2006; 175 (1): 208???212. DOI: 10.1097/00005392-200601000-00056.
- Metcalfe PD, Rink RC. Bladder augmentation: Complications in the pediatric population. Curr Urol Rep 2007; 8 (2): 152–156. DOI: 10.1007/s11934-007-0065-x.
- Walker RD. The management of the failed bladder neck procedure in patients with spina bifida. BJU Int 2016; 92: 35–37. DOI: 10.1046/j.1464-410x.92.s1.13.x.
- Scales CD, Wiener JS. Evaluating Outcomes of Enterocystoplasty in Patients With Spina Bifida: A Review of the Literature. J Urol 2008; 180 (6): 2323–2329. DOI: 10.1016/j.juro.2008.08.050.
- Kurzrock EA. Pediatric enterocystoplasty: long-term complications and controversies. World J Urol 2009; 27 (1): 69–73. DOI: 10.1007/s00345-008-0335-3.
- Merriman LS, Arlen AM, Kirsch AJ, Leong T, Smith EA. Does augmentation cystoplasty with continent reconstruction at a young age increase the risk of complications or secondary surgeries? J Pediatr Urol 2015; 11 (1): 41.e1–41.e5. DOI: 10.1016/j.jpurol.2014.08.016.
- Hensle TW, Gilbert SM. A review of metabolic consequences and long-term complications of enterocystoplasty in children. Curr Urol Rep 2007; 8 (2): 157–162. DOI: 10.1007/s11934-007-0066-9.
- Husmann DA. Malignancy after gastrointestinal augmentation in childhood. Ther Adv Urol 2009; 1 (1): 5–11. DOI: 10.1177/1756287209104163.
- Austin JC. Long-term risks of bladder augmentation in pediatric patients. Curr Opin Urol 2008; 18 (4): 408–412. DOI: 10.1097/mou.0b013e328300587c.
- Arlen AM, Dudley AG, Kieran K. Association of spina bifida with cancer. Transl Androl Urol 2020; 9 (5): 2358–2369. DOI: 10.21037/tau-19-771.
- Kaefer M, Pabby A, Kelly M. Improved Bladder Function After Prophylactic Treatment Of The High Risk Neurogenic Bladder In Newborns With Myelomeningocele. J Urol 1999; 162: 1071. DOI: 10.1097/00005392-199909000-00032.
- Bauer SB, Nijman RJM, Drzewiecki BA, Sillen U, Hoebeke P. International Children’s Continence Society standardization report on urodynamic studies of the lower urinary tract in children. Neurourol Urodyn 2015; 34 (7): 640–647. DOI: 10.1002/nau.22783.
Última actualización: 2025-09-21 13:35