15: Anomalías de duplicación, migración y fusión
Este capítulo durará aproximadamente 28 minutos para leer.
Introducción
Las anomalías de duplicación, migración y fusión del tracto urinario son frecuentes y conducen a diversas alteraciones estructurales y funcionales. Las secuelas anatómicas de la duplicación ureteral incluyen uréter ectópico, ureterocele y reflujo vesicoureteral. Estas pueden manifestarse clínicamente como obstrucción del tracto urinario, infección, incontinencia y displasia renal. Las anomalías frecuentes de migración y fusión incluyen ectopia renal y riñón en herradura, que en ocasiones se asocian con patología que incluye displasia renal, reflujo vesicoureteral, obstrucción y litiasis. La gravedad varía ampliamente, y el tratamiento va desde la observación hasta reconstrucciones quirúrgicas mayores. El tratamiento, cuando está indicado, es principalmente quirúrgico y está dirigido a preservar la función renal, minimizar la morbilidad por infección del tracto urinario, corregir la incontinencia urinaria y aliviar los síntomas asociados con la obstrucción del tracto urinario.
Embriología
El conocimiento de la embriología normal del desarrollo renal es fundamental para comprender las anomalías de duplicación, migración y fusión. Dos estructuras embriológicas dan origen al riñón y a los uréteres humanos maduros; la yema ureteral, que se desarrolla en el sistema colector y los uréteres, y el mesénquima metanéfrico, que se desarrolla en las nefronas y el parénquima renal. La yema ureteral se origina como una evaginación epitelial del conducto mesonéfrico (de Wolff) distal. Normalmente, existe una yema ureteral única en cada lado que da lugar a uréteres únicos bilaterales. La yema ureteral crece hacia el mesénquima metanéfrico y un proceso de inducción recíproca provoca la nefrogénesis y la ramificación ureteral. La unidad renal con su sistema colector ramificado (metanefros) migra en sentido craneal y rota 90 grados, de modo que el hilio renal queda orientado medialmente. El uréter crece en sentido caudal para fusionarse con el seno urogenital, que se convierte en la vejiga. Una vez incorporado al seno urogenital, el orificio ureteral migra en sentido craneolateral desde el cuello vesical hasta la posición ortotópica en la cresta trigonal.1
Se cree que las anomalías de duplicación derivan de yemas ureterales supernumerarias o de una ramificación precoz. Las anomalías de migración resultan de un ascenso o una rotación anómalos. Si los riñones entran en contacto entre sí, pueden fusionarse, dando lugar a un riñón en herradura o a una ectopia renal cruzada fusionada.2
Epidemiología y fisiopatología
Tabla 1 Patologías clínicas comunes en la duplicación renal.
| Anomalía | Problemas potenciales |
|---|---|
| Duplicación incompleta | Obstrucción de la unión pieloureteral del polo inferior Reflujo ureteroureteral (teórico) |
| Uréter ectópico | Obstrucción del polo superior Incontinencia urinaria Pielonefritis Epididimitis |
| Ureterocele | Obstrucción del polo superior Obstrucción del polo inferior Obstrucción de la salida vesical Micción disfuncional Incontinencia urinaria por disrupción del esfínter Pielonefritis |
| Reflujo vesicoureteral | Nefropatía por reflujo Pielonefritis |
Duplicación
Las anomalías de duplicación renal constituyen la anomalía congénita del tracto urinario más frecuente, con una incidencia del 0,8–5% observada en estudios de autopsias.3,4 Un riñón dúplex se refiere a un riñón con dos hemirriñones, superior e inferior, cada uno con un sistema de drenaje pielocalicial independiente. La duplicación bilateral ocurre en el 16–40% de los casos, y la duplicación se detecta con mayor frecuencia en mujeres.3,5 Las anomalías de duplicación pueden ser hereditarias, con una incidencia del 12–30% en familias afectadas.6,7
La duplicación puede ser incompleta (parcial) o completa. En la duplicación incompleta, los dos sistemas colectores pueden unirse en cualquier punto antes de entrar en la vejiga. Esto ocurre cuando una única yema ureteral se ramifica antes de alcanzar el mesénquima metanéfrico.3 La mayoría de las duplicaciones ureterales son incompletas y clínicamente insignificantes. La duplicación parcial abarca desde una pelvis renal bífida hasta una duplicación de tipo Y, con los uréteres uniéndose en cualquier punto antes de su inserción en la vejiga. Un problema que se encuentra con la duplicación incompleta es la obstrucción de la unión pieloureteral (UPJ), que se presenta casi exclusivamente en el polo inferior.8,9 La arquitectura intrarrenal en los sistemas duplicados puede explicar la predilección de las obstrucciones de la UPJ por el polo inferior. Por lo general, el uréter del polo superior drena un único infundíbulo sin una pelvis verdadera. El polo inferior, que comprende dos tercios del parénquima, suele contener al menos dos cálices mayores y una pelvis renal verdadera.9
La patología clínicamente significativa surge con mayor frecuencia de la duplicación completa. En la duplicación completa, uréteres independientes drenan los hemirriñones superior e inferior y desembocan por separado en el tracto genitourinario. Esto ocurre cuando dos yemas ureterales brotan del conducto mesonéfrico. Cada yema o rama induce la maduración de una unidad renal suprayacente. Distalmente, el uréter inferior se incorporará primero al seno urogenital y migrará más craneolateralmente. El uréter superior se incorpora más tarde o no se incorpora en absoluto y, por lo tanto, queda desplazado medial y caudalmente en la vejiga o, si nunca alcanza el seno urogenital, se localiza ectópicamente a lo largo de los derivados del conducto mesonéfrico. El orificio ureteral craneolateral se asocia de manera confiable con el hemirriñón inferior y el orificio ureteral caudomedial con el hemirriñón superior, una relación embriológica conocida como la ley de Weigert-Meyer.10,11 En raras ocasiones, yemas ureterales adicionales o una ramificación temprana pueden dar lugar a una triplicación ureteral (Figura 1).
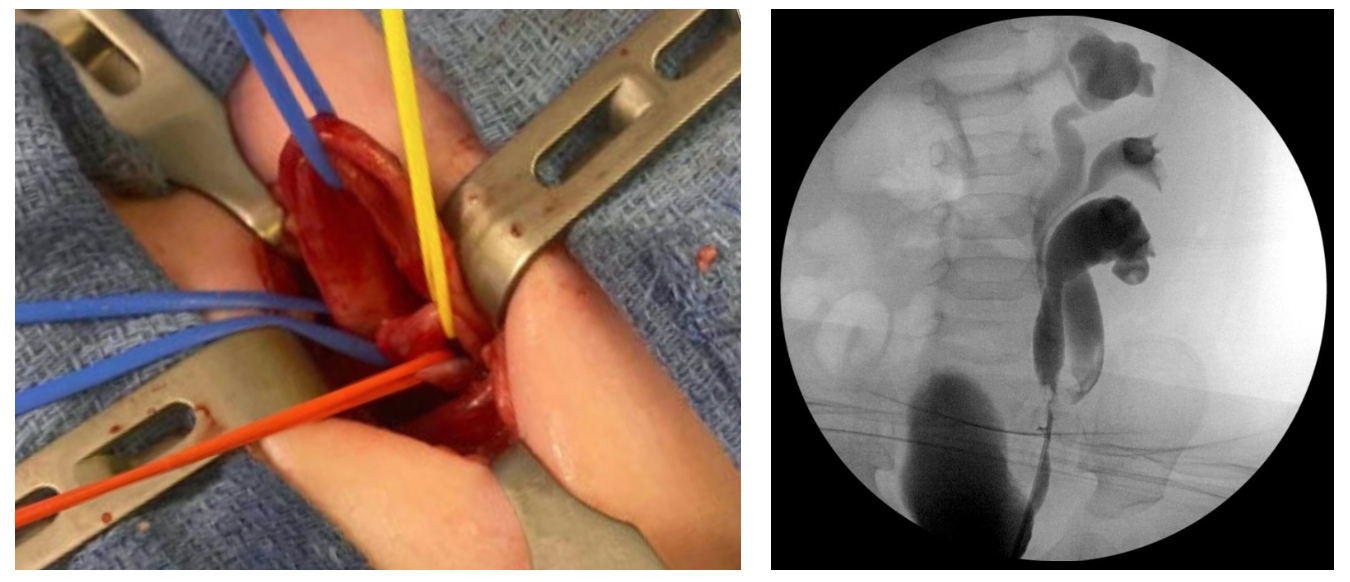
Figura 1 Triplicación ureteral. A: Foto intraoperatoria durante ureteroureterostomía superior a inferior. El lazo vascular azul proximal rodea el uréter del hemisistema superior, que es ectópico al cuello vesical con obstrucción. Los lazos vasculares rojo y amarillo rodean los uréteres de los hemistemas medio e inferior, que están duplicados de forma incompleta. B: Ureteropielograma retrógrado después de la ureteroureterostomía.
Uréter ectópico
Un uréter ectópico típicamente se implanta caudal a la localización normal en el reborde trigonal, a lo largo del trayecto del sistema mesonéfrico en desarrollo. En los niños, los uréteres ectópicos pueden abrirse al cuello vesical, la uretra posterior (lo más frecuente), o a derivados del conducto mesonéfrico: epidídimo, vesícula seminal o conducto deferente.12 Cabe destacar que estas localizaciones son proximales al complejo esfinteriano uretral y, por tanto, nunca producen incontinencia. Por el contrario, en las niñas los uréteres ectópicos pueden terminar distal al esfínter externo y causar incontinencia urinaria continua. Los sitios terminales más frecuentes de los uréteres ectópicos en niñas son el cuello vesical y la uretra proximal (1/3), el vestíbulo vaginal (1/3), la vagina (1/4), y el cuello uterino, útero o recto (raros).12,13 Aproximadamente el 75–90% de los uréteres ectópicos se asocian con la hemiunidad superior de un riñón dúplex.14,15
Los problemas que resultan de la ectopia ureteral incluyen obstrucción ureteral con la consiguiente displasia renal y/o riesgo de infección, incontinencia urinaria continua en niñas y, ocasionalmente, epididimitis por reflujo de orina hacia el conducto deferente en niños.
Ureterocele
Un ureterocele es una dilatación quística de la porción terminal del uréter. La embriología no se comprende por completo. Una teoría plantea que los ureteroceles resultan de la persistencia de la membrana de Chwalle, la membrana ureteral primitiva que separa la yema ureteral del seno urogenital en desarrollo. De forma alternativa, algunos piensan que un ureterocele resulta de una inducción anómala de la musculatura del trígono vesical.16,17
Se han propuesto varios sistemas de clasificación basados en la localización del orificio ureteral, las características del orificio o la asociación con sistemas colectores únicos o duplicados. La clasificación adoptada por la Sección de Urología de la Academia Estadounidense de Pediatría es quizá la más simple y clasifica los ureteroceles según la localización del tejido ureteral dilatado.18 Los ureteroceles se clasifican en intravesicales (ortotópicos) o extravesicales (ectópicos). Los ureteroceles intravesicales están contenidos por completo dentro de la vejiga (Figura 2). Los ureteroceles que se extienden más allá del cuello vesical se clasifican como extravesicales. En ocasiones, distinguir entre ambas puede ser difícil. Por ejemplo, un ureterocele intravesical puede prolapsar a través del cuello vesical y un ureterocele extravesical puede tener un orificio ureteral intravesical (Figura 3). Los ureteroceles extravesicales con extensión submucosa más allá del cuello vesical se denominan a veces cecoureteroceles.
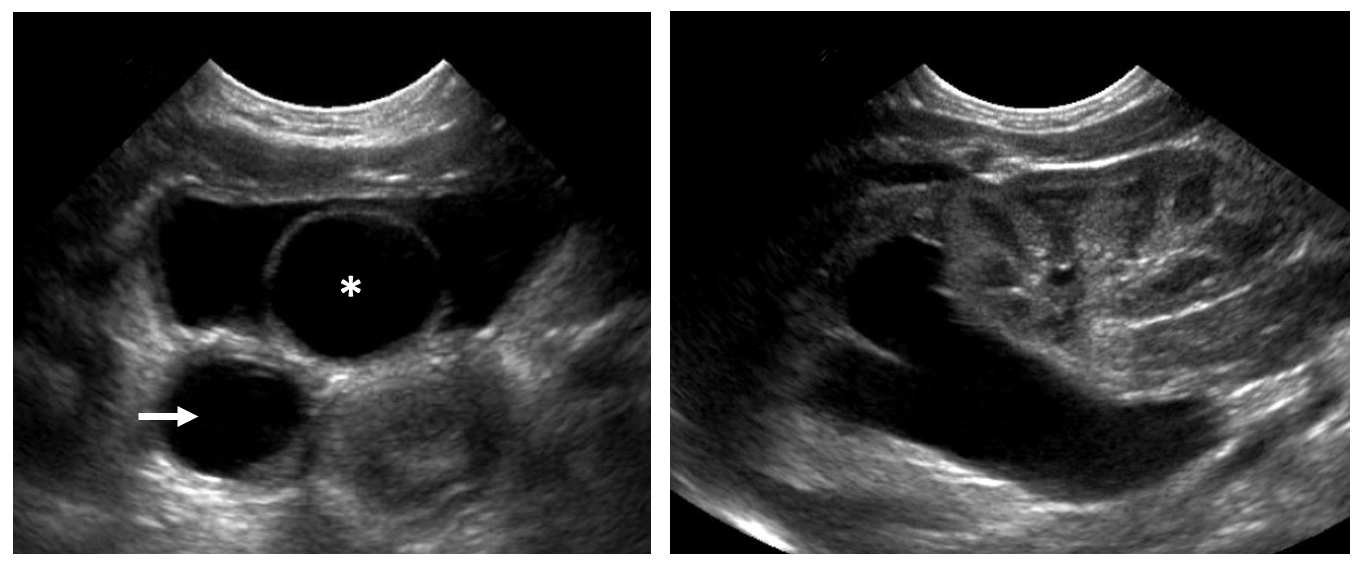
Figura 2 Ureterocele intravesical. A: Un ureterocele intravesical con su característica apariencia quística de pared delgada en la ecografía (asterisco). Se observa dilatación ureteral marcada (flecha). B: La hemiunidad superior asociada al ureterocele presenta hidronefrosis con adelgazamiento del parénquima renal. La hemiunidad inferior parece sana.
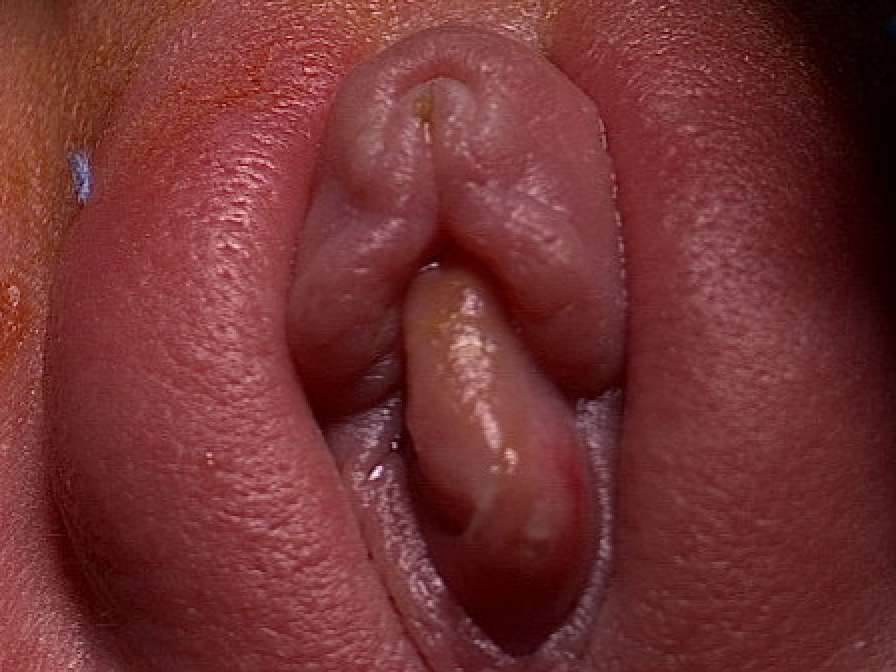
Figura 3 Prolapso de ureterocele en una lactante de sexo femenino.
En los sistemas duplicados, los ureteroceles se asocian casi siempre con el polo superior y el 60% son extravesicales.16 Los problemas incluyen obstrucción del polo superior y posiblemente del inferior, que en el contexto de una infección del tracto urinario puede presentarse como sepsis florida. La obstrucción suele asociarse con un polo superior displásico.19 Además, los ureteroceles que prolapsan pueden obstruir la salida vesical. Los ureteroceles grandes se asocian con una musculatura vesical subyacente deficiente, y los ureteroceles que se extienden más allá del cuello vesical pueden destruir el mecanismo de continencia femenino.20,21
Reflujo vesicoureteral
La combinación de duplicación ureteral y reflujo vesicoureteral es frecuente. Clásicamente, el reflujo se produce en el uréter del polo inferior, que tiene un orificio ureteral desplazado lateralmente y un túnel submucoso acortado, como predice la ley de Weigert-Meyer.22 El uréter del polo superior, con un orificio más medial y caudal, tiene un túnel submucoso más largo y es menos probable que presente reflujo. Cuando se produce reflujo en los uréteres de los polos superior e inferior, ambos orificios tienden a estar desplazados lateralmente o la duplicación es parcial. El reflujo hacia el uréter del polo superior también puede ocurrir si su orificio es ectópico al cuello vesical o la uretra, presumiblemente porque evita el trígono y tiene un túnel submucoso inadecuado. El reflujo en dichos uréteres solo ocurre durante la micción, y el cuello vesical tónicamente cerrado puede causar obstrucción durante la fase de almacenamiento (Figura 4). Como en los sistemas no duplicados, las secuelas del reflujo vesicoureteral en sistemas duplicados incluyen nefropatía por reflujo y pielonefritis.
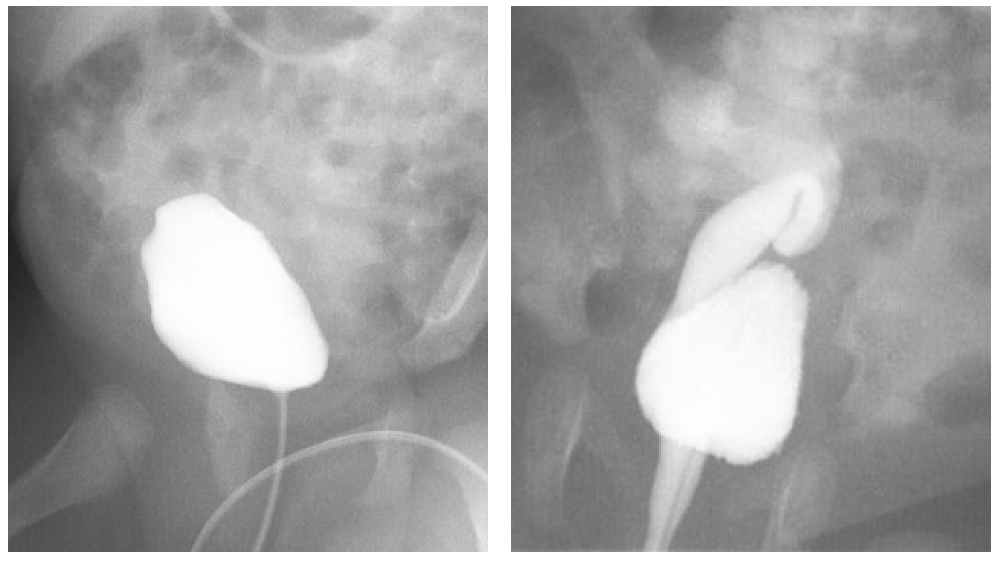
Figura 4 Uréter ectópico que se inserta en el cuello vesical. A: Durante el llenado vesical, no se observa reflujo vesicoureteral. B: El reflujo vesicoureteral se hace evidente durante la micción. Se observa que este riñón está localizado de manera ectópica en la pelvis.
Anomalías de migración y fusión
La ectopia renal se refiere a un riñón que no alcanza su posición normal y resulta de una migración y/o fusión renales anómalas durante el desarrollo embrionario.23 Los riñones ectópicos pueden clasificarse como simples, en herradura o cruzados. La incidencia comunicada varía de 1 en 500 a 1 en 5.000.24,25
Ectopia simple
La ectopia renal simple se refiere a un riñón ubicado en cualquier punto a lo largo del trayecto embrionario ipsilateral de ascenso desde la pelvis hasta la fosa renal. Los riñones pélvicos no ascienden más allá del reborde pélvico y son los riñones ectópicos más frecuentes, representando el 60% de todos los casos.26 En el 90% de los casos, la anomalía es unilateral y presenta una ligera preponderancia del lado izquierdo.26 Se pueden presentar otras posiciones ectópicas del riñón, ubicándose en algún punto entre la pelvis y la posición normal o, en raras ocasiones, dentro del tórax.27
Riñón en herradura
El riñón en herradura es la anomalía de fusión renal más común, con una incidencia entre 1:400 y 1:1800.28,29 En el 95% de los casos, los polos inferiores de ambos riñones están unidos por un istmo de tejido renal, que por lo general consiste en parénquima vascularizado, pero a veces es displásico o fibroso.29 En alrededor del 40% de los casos, el istmo se sitúa a nivel de L4, donde queda atrapado por debajo del origen de la arteria mesentérica inferior durante el ascenso renal. Rara vez, el istmo puede situarse posterior a la aorta y/o a la vena cava inferior.30 Una pequeña proporción de riñones en herradura están fusionados en sus polos superiores.31 La presencia del istmo impide la rotación medial completa de las pelvis renales y los uréteres deben arquearse anteriormente para pasar por encima del istmo, lo que explica la incidencia relativamente alta de anomalías ureteropélvicas (20%) asociadas con los riñones en herradura (Figura 5).32,33,34
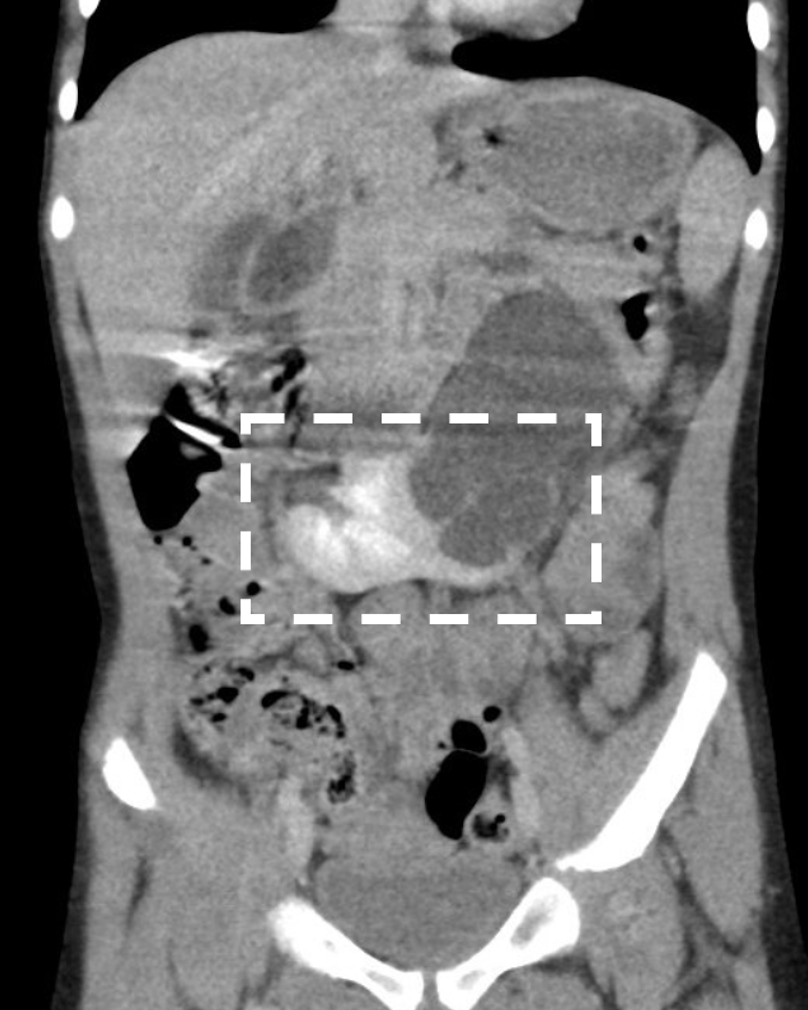
Figura 5 Riñón en herradura con obstrucción de la unión pieloureteral izquierda. El istmo (polos inferiores fusionados) está marcado por un cuadrado. Se observa hidronefrosis en el lado izquierdo.
Ectopia renal cruzada
La ectopia renal cruzada ocurre cuando uno o ambos riñones cruzan la línea media durante la migración. Hay cuatro variedades de ectopia renal cruzada: ectopia renal cruzada fusionada, en la que el riñón ectópico se fusiona con el riñón contralateral (85% de los casos), ectopia renal cruzada sin fusión, ectopia cruzada bilateral y ectopia cruzada solitaria. Existe un leve predominio masculino, y el cruce de izquierda a derecha ocurre con mayor frecuencia que de derecha a izquierda. En la ectopia renal cruzada fusionada, el punto de fusión suele estar entre el polo superior del riñón cruzado y el polo inferior del riñón en posición normal (tipo fusionado unilateral (Figura 6).35,36,37,38
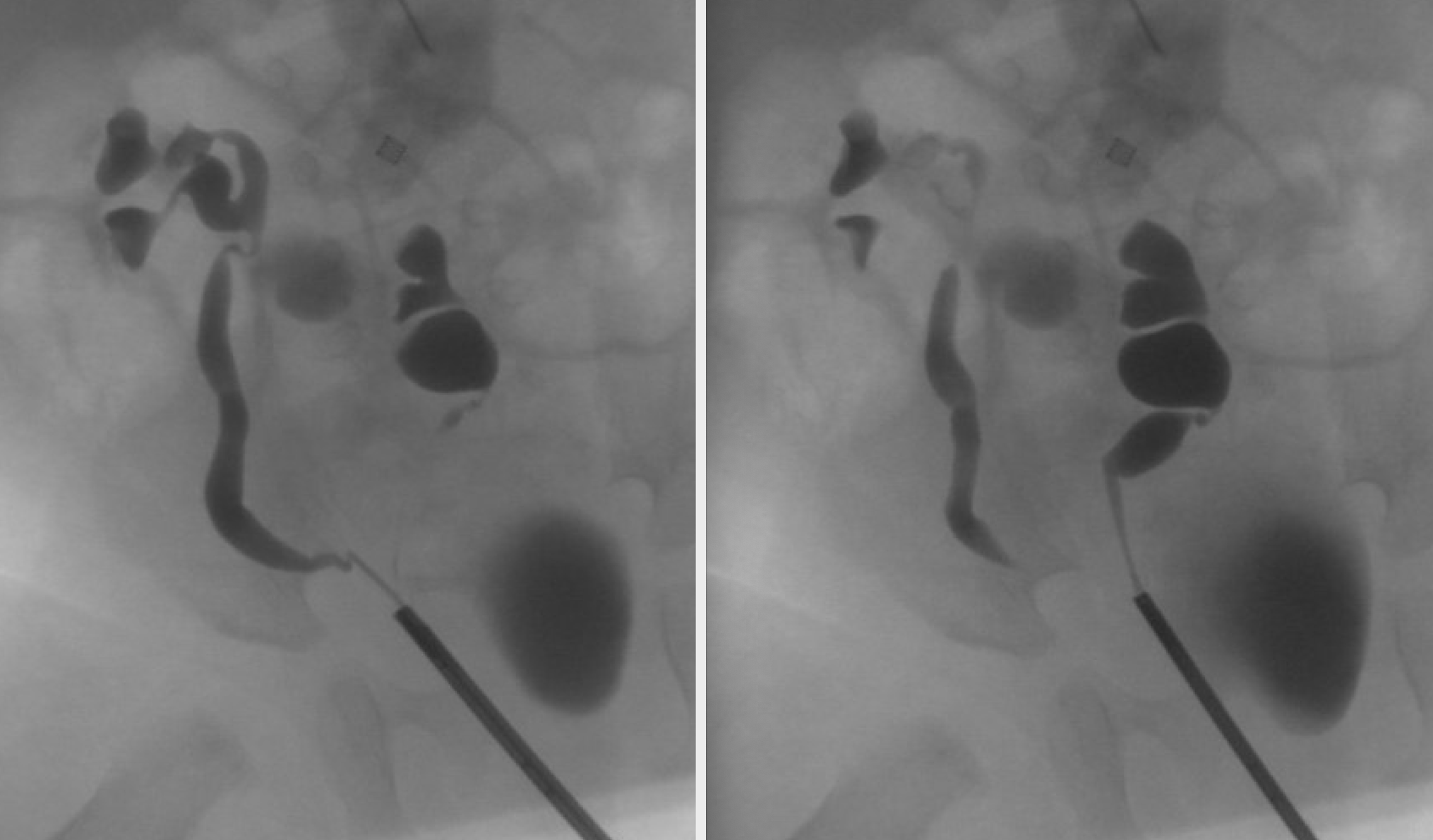
Figura 6 Ectopia renal cruzada con fusión. La pielografía retrógrada demuestra uréteres independientes. El riñón izquierdo se encuentra a la derecha de la línea media. Su polo superior está fusionado con el polo inferior del riñón derecho.
Los riñones ectópicos suelen ser asintomáticos. Cuando se detectan, suelen ser hipoplásicos o de forma irregular. El reflujo vesicoureteral es la anomalía asociada más frecuente, que se ha reportado en el 30–70% de los riñones en niños con ectopia renal. Curiosamente, en los casos de ectopia renal simple, el reflujo unilateral se observa con frecuencia en el riñón ortotópico.32,33,39 La malrotación y la inserción alta del uréter pueden conducir a obstrucción de la unión ureteropiélica, estasis urinaria y nefrolitiasis asociada.32,33,34 Además, dado que los riñones obtienen su vascularización de vasos cercanos durante el ascenso embrionario, el aporte vascular es predeciblemente anómalo en la ectopia renal y en las anomalías de fusión. En un estudio de 90 riñones en herradura, se identificaron 387 arterias, y casi una cuarta parte de los pacientes presentaba anatomía anómala de la vena renal.40
Las anomalías de migración y de fusión también se asocian comúnmente con anomalías no urológicas. El riñón en herradura, por ejemplo, está presente en el 14–20% de las niñas con síndrome de Turner.29 La ectopia renal y los riñones en herradura se observan con frecuencia en niños con malformaciones anorrectales y anomalías genitales, a veces como parte del síndrome VACTERL.41 En varones, las anomalías genitales pueden incluir hipospadias y criptorquidia.32 Si bien la agenesia renal es el hallazgo genitourinario clásico en mujeres con anomalías müllerianas, también se han descrito la duplicación uterina y la agenesia vaginal en asociación con anomalías de migración y de fusión.42,43
Evaluación y diagnóstico
Duplicación
Las anomalías de duplicación pueden diagnosticarse prenatalmente mediante ecografía de rutina. Posnatalmente, la duplicación se diagnostica con frecuencia durante la evaluación de una infección del tracto urinario (ITU); aproximadamente el 8% de los niños que consultan por una ITU presentan duplicación.44,45,46 En niñas que ya controlan esfínteres, la duplicación puede manifestarse con incontinencia si un uréter ectópico desemboca distal al complejo esfinteriano urinario.13,47 Los niños también pueden presentar síntomas de obstrucción del tracto urinario, como dolor intermitente o falta de medro.48 Frecuentemente, la duplicación se presenta como un hallazgo incidental en los estudios de imagen.49
La evaluación de la duplicación ureteral se centra en el diagnóstico, la prevención y el manejo de la hidronefrosis, la infección del tracto urinario, la disfunción renal y los trastornos miccionales. La duplicación detectada de forma incidental en ausencia de estos hallazgos no requiere estudios adicionales. Cada vez con mayor frecuencia, las anomalías de duplicación se detectan en el período prenatal cuando se observan hidronefrosis o ureteroceles en ecografías obstétricas.50 En raras ocasiones, puede estar presente displasia renal bilateral o una cicatrización renal significativa, con el consiguiente deterioro de la función renal. Como en todos los niños con displasia renal, se deben monitorizar la función renal y los niveles de electrolitos, y se debe realizar cribado de hipertensión y proteinuria. Diversas pruebas pueden aportar información valiosa respecto a la duplicación ureteral completa o incompleta, la ectopia, la dilatación ureteral, los ureteroceles y la función renal relativa.
Ecografía
Prácticamente todos los pacientes con duplicación renal requieren una ecografía de los riñones y la vejiga. Esto detectará hidronefrosis del polo superior y/o inferior, hidroureter, ureteroceles, y proporcionará información sobre la función de cada unidad renal según el aspecto del parénquima renal. La hidronefrosis del sistema superior puede ocurrir secundaria a un uréter ectópico obstruido o a un ureterocele. Con frecuencia se observa hidroureter asociado (Figura 2) Es común observar displasia marcada secundaria a nefropatía obstructiva. Un parénquima renal normal sin dilatación ureteral puede sugerir una duplicación incidental o un uréter ectópico que no está obstruido (Figura 7). En niñas, esto debe motivar preguntas sobre incontinencia continua para evaluar uréteres ectópicos distales al esfínter uretral (p. ej., “¿alguna vez ha tenido un período de sequedad?”). La hidronefrosis del sistema inferior puede deberse a reflujo vesicoureteral, obstrucción de la unión pieloureteral (UPJ), u obstrucción ureteral secundaria a un ureterocele del polo superior. Ecográficamente, un ureterocele aparece como una estructura intravesical quística de pared delgada (Figura 2), que puede contener detritos sugestivos de estasis urinaria o infección.
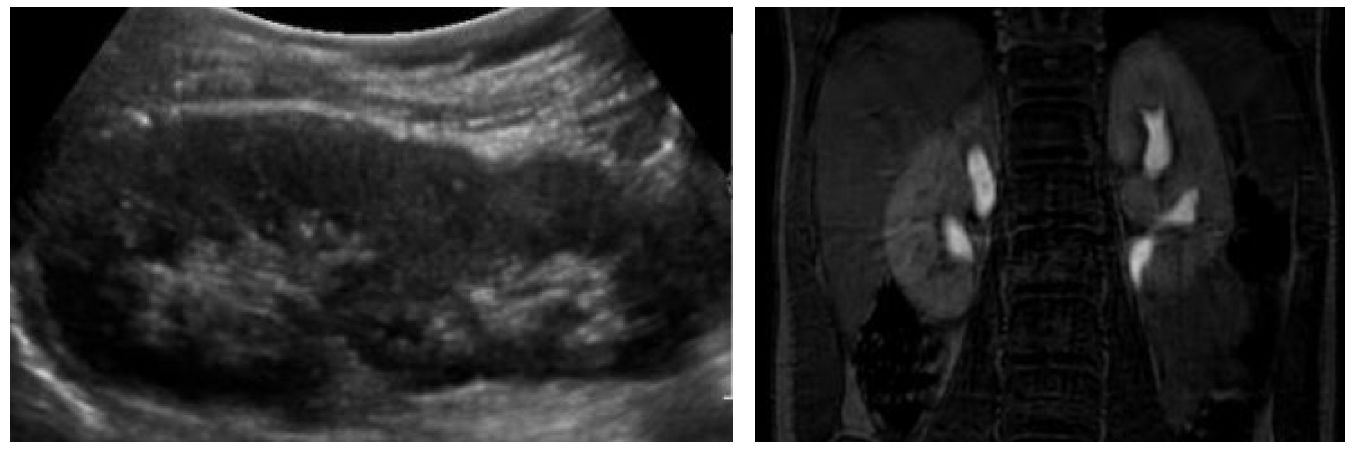
Figura 7 Incontinencia urinaria continua en una paciente de sexo femenino debida a uréteres ectópicos bilaterales. A: La ecografía renal demuestra dos hemirriñones sanos sin hidronefrosis. B: La urografía por resonancia magnética delinea la anatomía ureteral. En este caso, ambos uréteres de los polos superiores desembocaban en la vagina.
Cistouretrografía miccional
La VCUG es el estudio de elección para detectar y caracterizar el reflujo vesicoureteral. En los sistemas duplicados, el reflujo hacia el uréter inferior solo afecta a los cálices inferolaterales, un patrón radiográfico conocido como el “signo del lirio caído” (Figura 8). La VCUG también puede evidenciar ureteroceles, que aparecen como defectos de llenado en la vejiga y se observan mejor durante el llenado inicial (Figura 8) La naturaleza dinámica de esta modalidad proporciona información sobre la obstrucción del cuello vesical, el prolapso del ureterocele y la eversión del ureterocele, que se presenta como un divertículo vesical cuando la vejiga está distendida y sugiere una musculatura trigonal deficiente. Debe realizarse una VCUG si se observa dilatación del tracto urinario en la ecografía para evaluar reflujo vesicoureteral de alto grado, y antes de la reconstrucción del tracto urinario, ya que la presencia de reflujo vesicoureteral influirá en la toma de decisiones quirúrgicas.
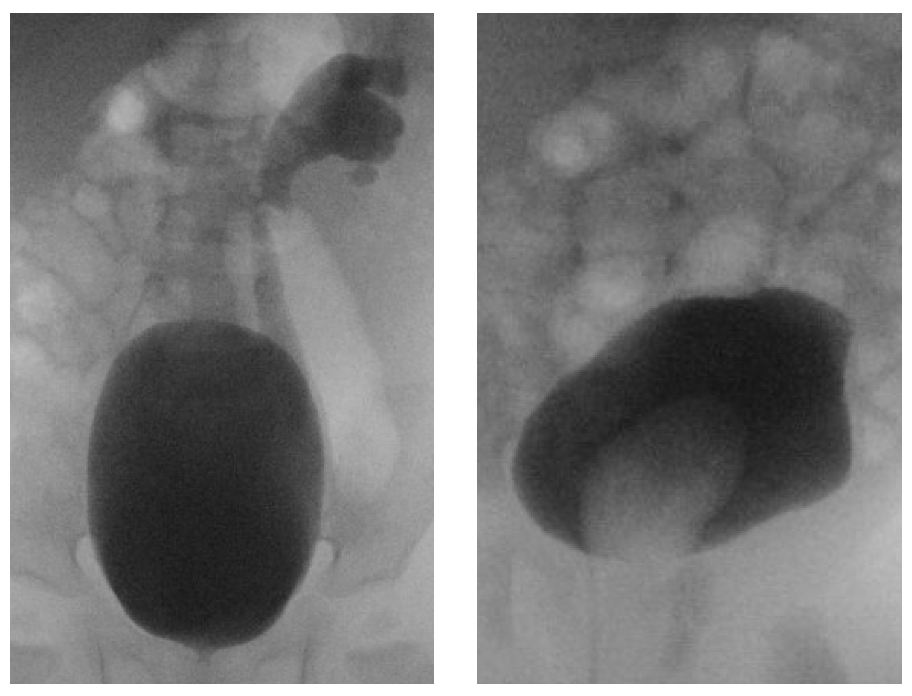
Figura 8 A: “Signo del lirio caído.” El reflujo vesicoureteral hacia el uréter inferior llena los cálices inferiores e interpolares. El sistema colector del hemirriñón superior no se opacifica. B: Un ureterocele se presenta como un defecto de llenado en la vejiga.
Gammagrafía renal
Las escintigrafías renales demuestran la función relativa y el drenaje de los hemirriñones. En el riñón dúplex no patológico, la función relativa de los hemirriñones superior e inferior es de un tercio y dos tercios, respectivamente.51
Urografía por resonancia magnética
La MRU proporciona un detalle anatómico preciso en pacientes con anatomía compleja que puede ser difícil de visualizar con otras modalidades. La MRU puede ser especialmente útil para delinear el trayecto de uréteres ectópicos (Figura 7). La MRU también es útil en pacientes con ureteroceles que ocultan otros aspectos de la vejiga, o en pacientes con anomalías uterinas, vaginales u otras anomalías vesicales.52 El software computacional permite calcular la función renal diferencial y los tiempos de tránsito urinario; sin embargo, este software es costoso, complejo y aún no está ampliamente disponible.53
Urografía intravenosa
Con otras modalidades disponibles, la IVU actualmente se realiza rara vez. Puede revelar duplicación y el nivel de confluencia, ectopia ureteral y ureteroceles, entre otras anomalías. Cuando la función de la porción superior es deficiente, la patología asociada no siempre se demuestra y las imágenes tardías pueden ser útiles.
Anomalías de migración y fusión
Las anomalías del ascenso y la fusión son hallazgos incidentales frecuentes. Los riñones ectópicos pélvicos no dilatados pueden ser difíciles de visualizar en la ecografía, y la ausencia del riñón en la fosa renal puede interpretarse erróneamente como agenesia renal.24,54 En tales casos, la presencia de tejido renal ectópico funcionante se demuestra mejor mediante gammagrafía renal con ácido dimercaptosuccínico (DMSA).54 La VCUG no es obligatoria en ausencia de infecciones del tracto urinario e hidronefrosis, ya que el reflujo vesicoureteral en este contexto tiende a ser de bajo grado y clínicamente insignificante.55 Síntomas como el cólico renal y la infección del tracto urinario generalmente denotan patología adicional, como reflujo vesicoureteral de alto grado, nefrolitiasis u obstrucción de la unión pieloureteral (UPJ), todas las cuales son más frecuentes con la ectopia renal y deben motivar una evaluación adicional (Figura 5)32,33,34
Opciones de tratamiento, resultados y complicaciones
Duplicación
El manejo de la duplicación renal depende de la patología en cuestión y abarca desde la observación con o sin profilaxis antibiótica hasta la cirugía mínimamente invasiva o la reconstrucción abierta del tracto urinario. En ausencia de infección, obstrucción o incontinencia urinaria, las anomalías de duplicación pueden observarse de forma segura. La profilaxis antibiótica está indicada en pacientes con duplicación y dilatación ureteral debida a un uréter ectópico obstructivo, un ureterocele o un reflujo vesicoureteral dilatante.56,57 La reconstrucción definitiva del tracto urinario puede realizarse a nivel vesical (tracto inferior) o a nivel renal (tracto superior). Rara vez es necesario un abordaje combinado del tracto superior e inferior. La función de los hemirriñones y la presencia de reflujo vesicoureteral son factores críticos para formular un plan quirúrgico. Si un paciente presenta patología a nivel vesical, incluyendo reflujo vesicoureteral o un ureterocele grande o ectópico, es preferible un abordaje del tracto inferior.
Incisión del uréterocele
La descompresión endoscópica del ureterocele se sugirió inicialmente a principios de la década de 1960. El abordaje inicial consistía en el destechamiento del ureterocele, que resultaba eficaz pero conllevaba una tasa elevada de reflujo vesicoureteral de novo. En 1962, Zielinski propuso una incisión baja, medial y transversal en la base del ureterocele.58 Esto proporciona una descompresión ureteral adecuada, mientras que el tejido residual del ureterocele crea una válvula de colgajo funcional que previene el reflujo vesicoureteral de novo. La abertura creada debe ser intravesical para evitar la obstrucción por el cuello vesical tónicamente cerrado. La incisión puede completarse con electrocauterio, láser o bisturí frío. La descompresión inicial exitosa se observa en más del 90% de los pacientes.59 Más recientemente, se ha promovido un abordaje “regadera” en ausencia de infección, en el cual el ureterocele se punciona 10–20 veces con láser.60 Esta técnica brinda una descompresión duradera con una reducción marcada del reflujo iatrogénico; sin embargo, la necesidad de procedimientos secundarios está menos estudiada. Para los ureteroceles intravesicales, independientemente de la técnica, la descompresión endoscópica primaria es con frecuencia la única intervención necesaria.61,62,63 Para los ureteroceles ectópicos, los resultados a largo plazo no son tan buenos; la descompresión del ureterocele puede considerarse una operación temporal, ya que entre el 50% y el 80% requerirá reconstrucción secundaria debido a reflujo sintomático o incontinencia urinaria.59,62,63
Inyección subureteral
Al igual que en los sistemas simples, el reflujo vesicoureteral en los sistemas duplicados puede tratarse mediante inyección subureteral de agentes de relleno. Inicialmente se informó que el éxito era inferior al de los sistemas simples, aunque análisis posteriores reportan tasas de éxito comparables.64 El fabricante del dextranómero-ácido hialurónico (Deflux®) ha eliminado recientemente la duplicación como contraindicación. La resolución del reflujo vesicoureteral tras la inyección subureteral se relaciona con la edad, el sexo, el grado y el momento del reflujo, y se analiza en detalle en el siguiente capítulo dedicado al reflujo vesicoureteral.
Clipaje ureteral
El clipaje ureteral es una técnica recientemente descrita para el manejo del hemisistema superior obstruido y no funcionante.65,66 El uréter del hemisistema superior se secciona, se drena y se liga por vía laparoscópica cerca del riñón, con la expectativa de que el parénquima renal no funcionante se atrofie. Esta técnica evita el riesgo de reflujo de novo asociado con la punción del ureterocele y es menos invasiva que la reconstrucción tradicional para un uréter ectópico, pero requiere vigilancia continua y conlleva riesgos que incluyen obstrucción sintomática, infección e incluso pionefrosis, y no ha sido ampliamente adoptada.
Reconstrucción del tracto urinario inferior
El tracto urinario inferior puede abordarse de forma intra- o extravesical. Las operaciones extravesicales, como la ureteroureterostomía, pueden realizarse mediante una incisión inguinal o de Gibson, dependiendo de la edad del paciente. Se abre el suelo del conducto inguinal, se desarrolla el espacio perivesical y los uréteres se encuentran posterolaterales al ligamento umbilical medial. Algunos colocan previamente stents ureterales en uréteres no dilatados para facilitar su identificación. En varones, se tiene cuidado de evitar lesionar el conducto deferente, que se arquea por encima de los uréteres. Las operaciones intravesicales, como el reimplante ureteral trans-trigonal, se realizan mediante una incisión de Pfannenstiel. Los abordajes laparoscópicos implican la colocación tradicional de puertos para la cirugía pélvica, incluyendo un puerto umbilical para la cámara y puertos de trabajo infraumbilicales izquierdo y derecho.
Ureteroureterostomía
La ureteroureterostomía es una excelente solución cuando existe patología aislada del polo superior o inferior, pero no de ambos.67 Si existe reflujo vesicoureteral inferior aislado, el uréter inferior puede anastomosarse al uréter superior en una anastomosis término-lateral. Con mayor frecuencia, se realiza una ureteroureterostomía superior a inferior en el contexto de un uréter ectópico que causa incontinencia urinaria u obstrucción. Los uréteres del polo superior que están obstruidos por ureteroceles intravesicales pequeños también pueden tratarse con ureteroureterostomía; los ureteroceles grandes o los ureteroceles ectópicos pueden requerir una operación intravesical para la escisión formal del ureterocele y la reconstrucción del suelo vesical.68 La ureteroureterostomía tiene varias ventajas. Evita la disección compleja alrededor del hilio renal que se requiere con la heminefrectomía, y evita la necesidad de abrir la vejiga o estrechar uréteres dilatados, como se requiere con el reimplante ureteral tradicional. Si no hay dilatación ureteral, el uréter debe espatularse para permitir una anastomosis ampliamente permeable. Las tasas de éxito a largo plazo superan el 90% en manos experimentadas.69,70,71,72 Los riesgos incluyen reflujo ureteroureteral (“yo-yo”), lesión del uréter receptor, estenosis anastomótica, fuga de orina e infección del muñón ureteral, todos los cuales son infrecuentes. Una anastomosis proximal con escisión del muñón ureteral distal puede disminuir la infección del muñón ureteral, y se realiza fácilmente por vía laparoscópica.73,74 Si los uréteres comparten una vaina común, no debe perseguirse agresivamente la escisión del muñón ureteral distal para evitar dañar la irrigación sanguínea compartida. Los stents, si se colocan, pueden atravesar la anastomosis o permanecer en el uréter receptor.
Reimplante ureteral
El reflujo vesicoureteral aislado en sistemas duplicados se trata de la misma manera que en sistemas simples. Cuando el reflujo vesicoureteral está presente además de un uréter ectópico u obstruido que requiere intervención, la reimplantación ureteral es tradicionalmente el procedimiento de elección. Distalmente, los uréteres duplicados suelen estar ubicados dentro de una vaina común y comparten un aporte vascular distal. En este caso, los uréteres deben movilizarse y reimplantarse en bloque.75 Un uréter ectópico no necesariamente compartirá el aporte vascular con el uréter del polo inferior. En este caso, los uréteres pueden reimplantarse de forma independiente.76 Como alternativa, puede realizarse una combinación de ureteroureterostomía del uréter superior al inferior y reimplante del uréter inferior.77,78
Los uréteres pueden reimplantarse mediante un abordaje intravesical o extravesical, con o sin taperización ureteral. Se evita la reimplantación extravesical bilateral debido al riesgo de lesión del plexo pélvico y de retención urinaria. La reimplantación de uréteres obstruidos o ectópicos, la taperización ureteral y la exéresis de ureteroceles asociados requieren desmembrar el uréter y abrir la mucosa vesical, y muchos cirujanos prefieren un abordaje intravesical en este contexto. Se han descrito abordajes vesicoscópicos para la reimplantación ureteral, pero aún no se han comunicado los resultados a largo plazo y el abordaje laparoscópico tradicional es extravesical.79 La tasa de éxito de la reimplantación en vaina común es superior al 95%, similar a la reimplantación de sistema único.80,81
Exéresis del ureterocele y reconstrucción vesical
Los ureteroceles no descomprimidos endoscópicamente, o los ureteroceles grandes con musculatura subyacente anormal, a veces se tratan con cirugía abierta. Esto puede incluir la escisión o marsupialización del ureterocele, la reconstrucción del suelo vesical y/o la reconstrucción del cuello vesical.82 La escisión del ureterocele se realiza con frecuencia en el momento del reimplante ureteral. Se reseca la mucosa redundante, cuidando de no dejar un colgajo mucoso que pueda obstruir la vejiga. El defecto del detrusor puede reconstruirse para asegurar un soporte adecuado para el uréter reimplantado. Puede producirse reflujo contralateral, presumiblemente debido a distorsión del trígono, aunque el significado clínico no está claro. Cuando el ureterocele se extiende más allá del cuello vesical, existe un riesgo significativo de incontinencia urinaria de esfuerzo y disfunción vesical. Se requiere una disección prudente; sin embargo, incluso con una técnica meticulosa, los resultados miccionales y de continencia son pobres y se consideran intrínsecos más que adquiridos.68,83
Ureterostomía cutánea
Este es un procedimiento temporal realizado en el contexto de infección del tracto urinario en el paciente pediátrico.16 Está indicado para el uréter del polo superior infectado y obstruido o para el uréter del polo inferior con reflujo vesicoureteral con infecciones urinarias de escape a pesar de profilaxis antibiótica. El abordaje quirúrgico abierto es similar a una ureteroureterostomía. El(los) uréter(es) patológico(s) se inciden o se seccionan y se marsupializan a la incisión cutánea inguinal. En lactantes, la combinación de vejigas pequeñas y uréteres dilatados impide un túnel ureteral adecuado, y la reimplantación suele diferirse. La dilatación ureteral a menudo mejora con la derivación, lo que puede obviar la necesidad de estrechamiento (taperización) en el momento de la reimplantación. La reconstrucción definitiva se realiza cuando el paciente tiene entre 1 y 2 años de edad. Cabe señalar que la ureterostomía derivativa es una excelente opción para el manejo de los ureteroceles cuando no se dispone de equipo endoscópico.
Enfoque del tracto urinario superior
La reconstrucción de la vía urinaria superior es apropiada cuando no existe patología de la vía urinaria inferior que deba abordarse. Aun así, a menudo se considera un abordaje de la vía urinaria inferior debido a su relativa facilidad y éxito.84,85
Heminefrectomía
La indicación para la heminefrectomía superior es un polo superior con mala función asociado a incontinencia urinaria o a patología del polo superior (p. ej., pielonefritis, cálculos). Se secciona el uréter del polo superior y se utiliza para tracción. Se ligan los vasos hiliares del polo superior, y el parénquima renal no viable se delimita y se reseca. Alternativamente, puede resecarse el parénquima del polo superior, que a menudo está pobremente vascularizado y se delimita con facilidad del polo inferior sano, y seguirse hasta los vasos hiliares del polo superior y el uréter, que luego se seccionan. Esto simplifica la disección hiliar pero conlleva riesgo de sangrado. El borde de corte del parénquima renal se cubre con la fascia de Gerota y grasa perirrenal. Se retira el uréter redundante del polo superior, pero el muñón ureteral residual se deja in situ. Se extrema el cuidado para no dañar la irrigación del parénquima ni del uréter del polo inferior.
La pérdida de la función del polo inferior es la complicación más importante relacionada con la heminefrectomía superior, con tasas reportadas de hasta nueve por ciento.84,86 Esto puede deberse a una lesión hiliar o a torsión posoperatoria del riñón remanente, y es más frecuente en niños menores de 1 año. En riñones completamente movilizados, considerar nefropexia para prevenir la torsión del remanente renal. Complicaciones adicionales incluyen sangrado posoperatorio (<5%), urinoma (5–20%, más común en procedimientos laparoscópicos, rara vez clínicamente significativo).87,88 Si bien la heminefrectomía superior fue en su momento el procedimiento de elección para el polo superior no funcional sin reflujo del polo inferior, los patrones de práctica están cambiando hacia la ureteroureterostomía, que es una operación más sencilla con tasas de éxito similares y menor riesgo para la función del polo inferior.84,85
La heminefrectomía inferior es menos frecuente. Se realiza por patología clínicamente significativa (p. ej., infección, obstrucción sintomática, litiasis) en el contexto de una unidad renal inferior no funcional con preservación del polo superior. Esto ocurre secundariamente a una obstrucción de la unión ureteropélvica (UPJ) o a un reflujo vesicoureteral de alto grado. Esta operación puede ser más compleja que la heminefrectomía superior, ya que el polo inferior se imbrica con el polo superior, lo que dificulta su separación. Las complicaciones y los resultados son similares a los de la heminefrectomía superior.89
Pieloplastia, Pieloureterostomía, Ureteroureterostomía proximal
Como se señaló previamente, la ureteroureterostomía con una anastomosis proximal ofrece algunas ventajas frente a una anastomosis distal. Con un abordaje laparoscópico, se evita una incisión en el flanco y hay una morbilidad adicional insignificante en comparación con una anastomosis distal.73,74 La obstrucción de la UPJ del polo inferior obliga a un abordaje del tracto superior. La técnica particular depende de la configuración del sistema colector. Con duplicación incompleta, el uréter inferior puede ser corto, lo que impide una pieloplastia tradicional. Se realiza una pieloureterostomía creando una anastomosis latero-lateral o término-lateral entre la pelvis renal del polo inferior obstruida y el uréter del polo superior.9,90 Si existe reflujo concomitante del polo inferior, lo mejor es una anastomosis término-lateral con drenaje y ligadura del muñón ureteral. En casos de duplicación completa, la pieloplastia tradicional es una alternativa que evita riesgos para el sistema de drenaje superior.9 Las complicaciones y los resultados son similares a los de la ureteroureterostomía y la pieloplastia tradicional.
Anomalías de migración y fusión
La evaluación de un riñón ectópico o en herradura no complicado puede razonablemente limitarse a la ecografía. Si no hay hidronefrosis y el niño está asintomático, no se requieren estudios adicionales. En el contexto de una fosa renal vacía, una gammagrafía renal con DMSA puede localizar tejido renal ectópico.50 Se indican estudios adicionales si hay hidronefrosis o antecedentes de infección documentada que susciten preocupación por reflujo vesicoureteral. Es importante recalcar que la mayoría de los pacientes no padecen problemas derivados de su riñón en posición anómala, y la intervención quirúrgica debe limitarse a corregir la patología concomitante, la obstrucción o el reflujo. Cuando la cirugía está indicada, los cirujanos deben anticipar la vasculatura renal anómala.
Puntos clave
- Las anomalías de duplicación y migración son frecuentes, y la mayoría son clínicamente insignificantes.
- La patología clínicamente significativa varía ampliamente. El manejo se centra en el diagnóstico, la prevención y el tratamiento de la obstrucción del tracto urinario, la infección, la disfunción renal y los trastornos miccionales.
- En la duplicación completa, el uréter superior es propenso a la obstrucción debido a un ureterocele o a una inserción ectópica, mientras que el uréter inferior es propenso al reflujo vesicoureteral.
- El manejo de los ureteroceles extravesicales es complejo y puede haber problemas persistentes relacionados con la micción y la continencia.
- Considere la función del hemirriñón superior e inferior y la presencia de reflujo vesicoureteral al formular un plan quirúrgico en sistemas dúplex.
- Cuando la cirugía esté indicada en casos de ectopia renal, tenga en cuenta que la vascularización renal será aberrante.
Lecturas recomendadas
- Ross JH, Kay R. Ureteropelvic junction obstruction in anomalous kidneys. Urol Clin North Am 1998; 25 (2): 219–225. DOI: 10.1016/S0094-0143(05)70010-0.
- Le H-K, Chiang G. Long-term Management of Ureterocele in Duplex Collecting Systems: Reconstruction Implications. Curr Urol Rep 2018; 19 (2). DOI: 10.1007/s11934-018-0758-3.
- Stanasel I, Peters CA. Ectopic Ureter, Ureterocele, and Ureteral Anomalies. 12th ed., Philadelphia, PA: Elsevier Saunders; 2020.
- VanderBrink BA, Reddy PP. Anomalies of the Upper Urinary Tract. 12th ed., Philadelphia, PA: Elsevier Saunders; 2020, DOI: 10.1016/b978-1-4160-6911-9.00117-1.
Referencias
- Moore KL, Persaud MD, Torchia MG. Urogenital System. The Developing Human: Clinically Oriented Embryology. 11th ed. Philadelphia, PA: Elsevier; 2020. DOI: 10.5005/jp/books/10691_7.
- Tanagho EA. Embryologic basis for lower ureteral anomalies: A hypothesis. Urology 1976; 7 (5): 451–464. DOI: 10.1016/0090-4295(76)90179-5.
- Decter RM. Renal duplication and fusion anomalies. Pediatr Clin North Am 1997; 44 (5): 1323–1341. DOI: 10.1016/S0031-3955(05)70559-9.
- Williams H. Renal revision: from lobulation to duplication—what is normal? Arch Dis Child. Educ Pract 2007; 92 (5). DOI: 10.1136/adc.2007.126680.
- Kaplan WE, Nasrallah P, King LR. Reflux in complete duplication in children. J Urol 1978; 120 (2): 220–222. DOI: 10.1016/s0022-5347(17)57116-5.
- Whitaker J, Danks DM. A Study of the Inheritance of Duplication of the Kidneys and Ureters. J Urol 1966; 95 (2): 176–178. DOI: 10.1016/s0022-5347(17)63429-3.
- Atwell JD, Cook PL, Howell CJ. Familial incidence of bifid and double ureters. Arch Dis Child 1974; 49 (5): 390–393. DOI: 10.1136/adc.49.5.390.
- Joseph DB, Bauer SB, Colodny AH. Lower pole ureteropelvic junction obstruction and incomplete renal duplication. J Urol 1989; 141 (4): 896–899. DOI: 10.1016/s0022-5347(17)41044-5.
- Ross JH, Kay R. Ureteropelvic junction obstruction in anomalous kidneys. Urol Clin North Am 1998; 25 (2): 219–225. DOI: 10.1016/S0094-0143(05)70010-0.
- Weigert C. Ueber einige Bildungsfehler der Ureteren. Arch Für Pathol Anat Physiol Für Klin Med. 1877; 70 (4): 490–501. DOI: 10.1007/BF01935232.
- Meyer R. Zur Anatomie und Entwicklungsgeschichte der Ureterverdoppelung. Virchows Arch Path Anat 1907; 187: 408–434. DOI: 10.1007/BF01946114.
- Stanasel I, Peters CA. Ectopic Ureter, Ureterocele, and Ureteral Anomalies. 12th ed., Philadelphia, PA: Elsevier Saunders; 2020.
- Duicu C, Kiss E, Simu I. A Rare Case of Double-System With Ectopic Ureteral Openings Into Vagina. Front Pediatr 2018; 6 (176). DOI: 10.3389/fped.2018.00176.
- Gillenwater JY, Grayhack JT, Howards SS, editors. Adult and Pediatric Urology. 4th ed., Lippincott Williams & Wilkins; 2002, DOI: 10.1001/jama.1991.03460240115044.
- Plaire JC, Pope JCI, Kropp BP. Management of Ectopic Ureters: Experience With the Upper Tract Approach. J Urol 1997; 158 (3): 1245–1247. DOI: 10.1016/s0022-5347(01)64442-2.
- Coplen DE, Duckett JW. The Modern Approach to Ureteroceles. J Urol 1995; 153 (1): 166–171. DOI: 10.1097/00005392-199501000-00068.
- Chwalle R. The process of formation of cystic dilatation of the vesical end of the ureter and of diverticula at the ureteral ostium. Urol Cutan Rev 1927; 31: 499–504.
- Glassberg KI, Braren V, Duckett JW. Suggested Terminology for Duplex Systems, Ectopic Ureters and Ureteroceles. J Urol 1984; 132 (6): 1153–1154. DOI: 10.1016/S0022-5347(17)50072-5.
- Bolduc S, Upadhyay J, Restrepo R. The predictive value of diagnostic imaging for histological lesions of the upper poles in duplex systems with ureteroceles. BJU Int 2003; 91 (7): 678–682. DOI: 10.1046/j.1464-410x.2003.04247.x.
- Abrahamsson K, Hansson E, Hermansson G. Bladder dysfunction: an integral part of the ectopic ureterocele complex. J Urol 1998; 160 (4): 1468–1470. DOI: 10.1016/s0022-5347(01)62593-x.
- Fehrenbaker LG, Kelalis PP, Stickler GB. Vesicoureteral Reflux and Ureteral Duplication in Children. J Urol 1972; 107 (5): 862–864. DOI: 10.1016/S0022-5347(17)61160-1.
- Campbell MF. Renal Ectopy. J Urol 1930; 24 (2): 187–198. DOI: 10.1016/s0022-5347(17)72892-3.
- Yuksel A, Batukan C. Sonographic Findings of Fetuses with an Empty Renal Fossa and Normal Amniotic Fluid Volume. Fetal Diagn Ther 2004; 19 (6): 525–532. DOI: 10.1159/000080166.
- Sheih CP, Liu MB, Hung CS. Renal abnormalities in schoolchildren. Pediatrics 1989; 84 (6): 1086–1090. DOI: 10.1542/peds.84.6.1086.
- Mouriquand P, Panait N. Renal Fusions and Ectopia. Pediatric Surgery 2012 (12). DOI: 10.1016/b978-0-323-02842-4.50113-3.
- N’Guessen G, Douglas Stephens F. Congenital superior ectopic (thoracic) kidney. Urology 1984; 24 (3): 219–228. DOI: 10.1016/0090-4295(84)90346-7.
- Natsis K, Piagkou M, Skotsimara A. Horseshoe kidney: a review of anatomy and pathology. Surg Radiol Anat 2014; 36 (6): 517–526. DOI: 10.1007/s00276-013-1229-7.
- Kirkpatrick JJ, Leslie SW. Horseshoe Kidney. 2021. DOI: 10.53347/rid-18142.
- Dajani AM. Horseshoe kidney: a review of twenty-nine cases. Br J Urol 1966; 38 (4): 388–402. DOI: 10.1111/j.1464-410x.1966.tb09725.x.
- Khougali HS, Alawad OAMA, Farkas N. Bilateral pelvic kidneys with upper pole fusion and malrotation: a case report and review of the literature. J Med Case Reports 2021; 15 (1). DOI: 10.1186/s13256-021-02761-1.
- Guarino N, Tadini B, Camardi P. The incidence of associated urological abnormalities in children with renal ectopia. J Urol 2004; 172 (4, Supplement): 1757–1759. DOI: 10.1097/01.ju.0000138376.93343.74.
- Kramer SA, Kelalis PP. Ureteropelvic junction obstruction in children with renal ectopy. J Urol (Paris 1984; 90 (5): 331–336. DOI: 10.1016/s0094-0143(05)70009-4.
- Gleason PE, Kelalis PP, Husmann DA. Hydronephrosis in Renal Ectopia: Incidence, Etiology and Significance. J Urol 1994; 151 (6): 1660–1661. DOI: 10.1016/S0022-5347(17)35338-7.
- McDonald JH, McClellan DS. Crossed renal ectopia. Am J Surg 1957; 93 (6): 995–1002. DOI: 10.53347/rid-26773.
- Marshall FF, Freedman MT. Crossed Renal Ectopia. J Urol 1978; 119 (2): 188–191. DOI: 10.53347/rid-26773.
- Abeshouse BS, Bhisitkul I. Crossed Renal Ectopia With and Without Fusion. Urol Int 1959; 9 (2): 63–91. DOI: 10.1159/000277442.
- VanderBrink BA, Reddy PP. Anomalies of the Upper Urinary Tract. 12th ed., Philadelphia, PA: Elsevier Saunders; 2020, DOI: 10.1016/b978-1-4160-6911-9.00117-1.
- CMA B, JAE W, GMA B. Urological and Nephrological Findings of Renal Ectopia. J Urol 2010; 183 (4): 1574–1578. DOI: 10.1016/j.juro.2009.12.041.
- Glodny B, Petersen J, Hofmann KJ. Kidney fusion anomalies revisited: clinical and radiological analysis of 209 cases of crossed fused ectopia and horseshoe kidney. BJU Int 2009; 103 (2): 224–235. DOI: 10.1111/j.1464-410X.2008.07912.x.
- Cunningham BK, Khromykh A, Martinez AF. Analysis of Renal Anomalies in VACTERL Association. Birt Defects Res A Clin Mol Teratol 2014; 100 (10): 801–805. DOI: 10.1002/bdra.23302.
- Je B-K, Kim HK, Horn PS. Incidence and Spectrum of Renal Complications and Extrarenal Diseases and Syndromes in 380 Children and Young Adults With Horseshoe Kidney. Am J Roentgenol 2015; 205 (6): 1306–1314. DOI: 10.2214/AJR.15.14625.
- Creatsas G, Malhotra N, Malhotra J. Vaginal agenesis associated with renal ectopia. Adolesc Pediatr Gynecol 1990; 3 (2): 103–105. DOI: 10.1016/S0932-8610(12)80191-5.
- Afshar K, Papanikolaou F, Malek R. Vesicoureteral reflux and complete ureteral duplication. conservative or surgical management? J Urol 2005; 173 (5): 1725–1727. DOI: 10.1097/01.ju.0000154164.99648.ee.
- Bisset G, Strife J. The duplex collecting system in girls with urinary tract infection: prevalence and significance. Am J Roentgenol 1987; 148 (3): 497–500. DOI: 10.2214/ajr.148.3.497.
- Stokland E, Jodal U, Sixt R. Uncomplicated duplex kidney and DMSA scintigraphy in children with urinary tract infection. Pediatr Radiol 2007; 37 (8): 826–828. DOI: 10.1007/s00247-007-0518-x.
- Hanson GR, Gatti JM, Gittes GK. Diagnosis of ectopic ureter as a cause of urinary incontinence. J Pediatr Urol 2007; 3 (1): 53–57. DOI: 10.1016/j.jpurol.2005.06.009.
- Gao Z, Wu J, Lin C, Men C. Transperitoneal Laparoscopic Heminephrectomy in Duplex Kidney: Our Initial Experience. Urology 2011; 77 (1): 231–236. DOI: 10.1016/j.urology.2010.02.002.
- Connolly JO, Chan MMY, Neild GH. Congenital Anomalies of the Kidney and Urinary Tract. Comprehensive Clinical Nephrology. 6th ed. Elsevier;2019:50,607-625.e1; . DOI: 10.1007/978-3-319-29219-9.
- Vergani P, Ceruti P, Locatelli A. Accuracy of prenatal ultrasonographic diagnosis of duplex renal system. J Ultrasound Med 1999; 18 (7): 463–467. DOI: 10.7863/jum.1999.18.7.463.
- Privett JTJ, Jeans WD, Roylance J. The incidence and importance of renal duplication. Clin Radiol 1976; 27 (4): 521–530. DOI: 10.1016/s0009-9260(76)80121-3.
- Dickerson EC, Dillman S JR, E.A.. Pediatric MR Urography: Indications, Techniques, and Approach to Review. Radiogr Rev Publ Radiol Soc N Am Inc 2015; 35 (4): 1208–1230. DOI: 10.1148/rg.2015140223.
- Jones RA, Perez-Brayfield MR, Kirsch AJ. Renal transit time with MR urography in children. Radiology 2004; 233 (1): 41–50. DOI: 10.1148/radiol.2331031117.
- Chow JS, Benson CB, Lebowitz RL. The Clinical Significance of an Empty Renal Fossa on Prenatal Sonography. J Ultrasound Med 2005; 24 (8): 1049–1054. DOI: 10.7863/jum.2005.24.8.1049.
- Elmacı AM, Dönmez Mİ, Soran M. Is voiding cystourethrography necessary for evaluating unilateral ectopic pelvic kidney? Turk J Urol 2019; 45 (Suppl 1). DOI: 10.5152/tud.2019.72798.
- Visuri S, Jahnukainen T, Taskinen S. Prenatal complicated duplex collecting system and ureterocele - important risk factors for urinary tract infection. J Pediatr Surg 2018; 53 (4): 813–817. DOI: 10.1016/j.jpedsurg.2017.05.007.
- Bascietto F, Khalil A, Rizzo G. Prenatal imaging features and postnatal outcomes of isolated fetal duplex renal collecting system: A systematic review and meta-analysis. Prenat Diagn 2020; 40 (4): 424–431. DOI: 10.1002/pd.5622.
- Zielínski J. Avoidance of Vesicoureteral Reflux after Transurethral Ureteral Meatotomy for Ureterocele. J Urol 1962; 88 (3): 386–386. DOI: 10.1016/S0022-5347(17)64805-5.
- Meir DB, CJTDAE S, Rao P. Does the endoscopic technique of ureterocele incision matter? J Urol 2004; 172 (2): 684–686. DOI: 10.1097/01.ju.0000129228.92805.31.
- Palmer BW, Greger H, Mannas DB. Comparison of Endoscopic Ureterocele Decompression Techniques. Preliminary Experience—Is the Watering Can Puncture Superior? J Urol 2011; 186 (4S): 1700–1704. DOI: 10.1016/j.juro.2011.04.007.
- Jelloul L, Berger D, Frey P. Endoscopic management of ureteroceles in children. Eur Urol 1997; 32 (3): 321–326. DOI: 10.1159/000019704.
- Cooper CS, Passerini-Glazel G, Hutcheson JC. Long-term followup of endoscopic incision of ureteroceles: intravesical versus extravesical. J Urol 2000; 164(3. DOI: 10.1097/00005392-200009020-00045.
- Renzo DD, Ellsworth PI, Caldamone AA. Transurethral Puncture for Ureterocele—Which Factors Dictate Outcomes? J Urol 1620; 2010;184(4. DOI: 10.1016/j.juro.2010.04.023.
- Hensle TW, Reiley EA, Ritch C. The clinical utility and safety of the endoscopic treatment of vesicoureteral reflux in patients with duplex ureters. J Pediatr Urol 2010; 6 (1): 15–22. DOI: 10.1016/j.jpurol.2009.05.015.
- Lopes RI, Fernandez N, Koyle MA. Clinical Outcomes of the Upper Urinary Tract after Ureteral Clipping for Treatment of Low Functioning or Nonfunctioning Renal Moieties. J Urol 2018; 199 (2): 558–564. DOI: 10.1016/j.juro.2017.09.080.
- Lopes RI, Mello MF, Koyle MA. Ureteral clipping for the treatment of a non-functioning upper kidney moiety associated with a massive ureterocele: step-by-step description of a novel technique. J Pediatr Urol 2019; 15 (3): 284–285. DOI: 10.1016/j.jpurol.2019.03.005.
- Buchtel HA. Uretero-ureterostomy. J Urol 1965; 93 (2): 153–157. DOI: 10.1016/s0022-5347(17)63740-6.
- Le H-K, Chiang G. Long-term Management of Ureterocele in Duplex Collecting Systems: Reconstruction Implications. Curr Urol Rep 2018; 19 (2). DOI: 10.1007/s11934-018-0758-3.
- Bracci U, Miano L, Lauretti C. Ureteroureterostomy in Complete Ureteral Duplication. Eur Urol 1979; 5: 347–351. DOI: 10.1159/000473152.
- Huisman TK, Kaplan GW, Brock WA. Ipsilateral Ureteroureterostomy and Pyeloureterostomy: A Review of 15 Years of Experience With 25 Patients. J Urol 1987; 138 (5): 1207–1210. DOI: 10.1016/S0022-5347(17)43551-8.
- Grimsby GM, Merchant Z, Jacobs MA. Laparoscopic-Assisted Ureteroureterostomy for Duplication Anomalies in Children. J Endourol 2014; 28 (10): 1173–1177. DOI: 10.1089/end.2014.0113.
- Abdelhalim A, Chamberlin JD, Truong H. Ipsilateral ureteroureterostomy for ureteral duplication anomalies: predictors of adverse outcomes. J Pediatr Urol 2019; 15 (5). DOI: 10.1016/j.jpurol.2019.05.016.
- Lee YS, Hah YS, Kim M-J. Factors Associated with Complications of the Ureteral Stump After Proximal Ureteroureterostomy. J Urol 2012; 188 (5): 1890–1894. DOI: 10.1016/j.juro.2012.07.015.
- Ellison JS, Lendvay TS. Robot-assisted ureteroureterostomy in pediatric patients: current perspectives. Robot Surg Res Rev 2017; 4: 45–55. DOI: 10.2147/RSRR.S99536.
- Belman AB, Filmer RB, King LR. Surgical Management of Duplication of the Collecting System. J Urol 1974; 112 (3): 316–321. DOI: 10.1016/S0022-5347(17)59718-9.
- Marshall S. Reimplantation of the Dilated Ectopic Ureter of the Duplex System as a Separate Unit. J Urol 1986; 135 (3): 574–575. DOI: 10.1016/S0022-5347(17)45743-0.
- Amar AD, Egan RM, Das S. Ipsilateral Ureteroureterostomy Combined with Ureteral Reimplantation for Treatment of Disease in both Ureters in a Child with Complete Ureteral Duplication. J Urol 1981; 125 (4): 581–582. DOI: 10.1016/s0022-5347(17)55111-3.
- Lashley DB, McAleer IM, Kaplan GW. Ipsilateral ureteroureterostomy for the treatment of vesicoureteral reflux or obstruction associated with complete ureteral duplication. J Urol 2001; 165 (2): 552–554. DOI: 10.1097/00005392-200102000-00067.
- Kanojia R, Pandey A, Bawa M. Vesicoscopic common sheath cohen’s reimplant in duplex ureters. J Urol 2021; 206 (Supplement 3, V03-03). DOI: 10.1097/JU.0000000000001991.03.
- Ellsworth PI, Lim DJ, Walker RD. Common sheath reimplantation yields excellent results in the treatment of vesicoureteral reflux in duplicated collecting systems. J Urol 1996; 155 (4): 1407–1409. DOI: 10.1097/00005392-199604000-00090.
- Barrieras D, Lapointe S, Houle A-M. Is Common Sheath Extravesical Reimplantation an Effective Technique to Correct Reflux in Duplicated Collecting Systems? J Urol 2003; 170 (4 Part 2): 1545–1547. DOI: 10.1097/01.ju.0000084149.02826.64.
- Lewis JM, Cheng EY, Campbell JB. Complete Excision or Marsupialization of Ureteroceles: Does Choice of Surgical Approach Affect Outcome? J Urol 2008; 180 (4S): 1819–1823. DOI: 10.1016/j.juro.2008.04.078.
- Holmes NM, Coplen DE, Strand W. Is Bladder Dysfunction and Incontinence Associated with Ureteroceles Congenital or Acquired? J Urol 2002; 168 (2): 718–719. DOI: 10.1016/S0022-5347(05)64732-5.
- Michaud JE, Akhavan A. Upper Pole Heminephrectomy Versus Lower Pole Ureteroureterostomy for Ectopic Upper Pole Ureters. Curr Urol Rep 2017; 18 (3). DOI: 10.1007/s11934-017-0664-0.
- Sheth KR, White JT, Janzen N. Evaluating Outcomes of Differential Surgical Management of Nonfunctioning Upper Pole Renal Moieties in Duplex Collecting Systems. Urology 2019; 123: 198–203. DOI: 10.1016/j.urology.2018.06.028.
- Gundeti MS, Ransley PG, Duffy PG. Renal outcome following heminephrectomy for duplex kidney. J Urol 2005; 173 (5): 1743–1744. DOI: 10.1097/01.ju.0000154163.67420.4d.
- Wallis MC, Khoury AE, Lorenzo AJ. Outcome Analysis of Retroperitoneal Laparoscopic Heminephrectomy in Children. J Urol 2006; 175 (6): 2277–2282. DOI: 10.1016/S0022-5347(06)00338-7.
- You D, Bang JK, Shim M. Analysis of the late outcome of laparoscopic heminephrectomy in children with duplex kidneys. BJU Int 2010; 106 (4): 250–254. DOI: 10.1111/j.1464-410x.2009.09038.x.
- Singh RR, Wagener S, Chandran H. Laparoscopic management and outcomes in non-functioning moieties of duplex kidneys in children. J Pediatr Urol 2010; 6 (1): 66–69. DOI: 10.1016/j.jpurol.2009.04.005.
- Leclair M-D, Vidal I, Suply E. Retroperitoneal Laparoscopic Heminephrectomy in Duplex Kidney in Infants and Children: A 15-Year Experience. Eur Urol 2009; 56 (2): 385–391. DOI: 10.1016/j.eururo.2008.07.015.
- Lowe GJ, Canon SJ, Jayanthi VR. Laparoscopic reconstructive options for obstruction in children with duplex renal anomalies. BJU Int 2008; 101 (2): 227–230. DOI: 10.1111/j.1464-410X.2007.07106.x.
Última actualización: 2025-09-21 13:35