17: Megaureter
This chapter will take approximately 21 minutes to read.
Introduction
Congenital anomalies of the ureterovesical junction (UVJ) often present with a dilated or ‘mega’-ureter (Figure 1). Many synonyms for this condition exist, including hydroureter and megaloureter, but all represent a ureter with a diameter larger than normal with or without associated renal pelvis dilatation. Smith classified megaureters into four categories, obstructed, refluxing, refluxing with obstruction, and non-refluxing/non-obstructing, later subdivided into primary and secondary by King.1,2 The Pfister-Hendren classification, established in 1978, was based on the morphological appearance: type I involved the distal ureter without associated hydronephrosis; type II extended to both ureter and pelvis; and type III was associated with severe hydroureteronephrosis and ureteric tortuosity.3 Congenital megaureters are twice as common in males, and present with a left-to-right ratio of 1.8:1.4 As the refluxing UVJ anomaly is discussed in detail elsewhere, the focus of this chapter will be the obstructed, and non-obstructed non-refluxing, megaureters.
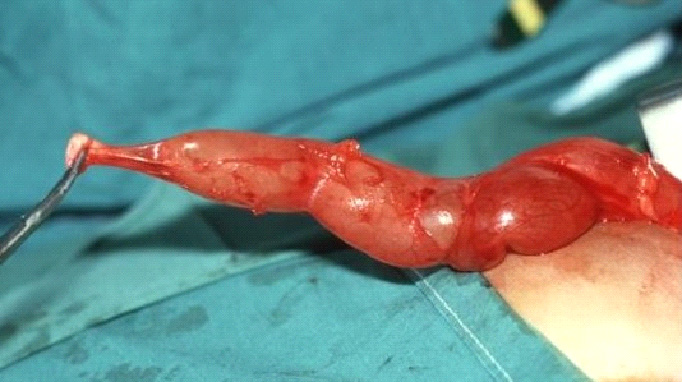
Figure 1 Intra-operative view of megaureter (MU) and narrowed, aperistaltic juxta-vesical segment (AS).
The definition of a dilated ureter is not well established in the literature. Cussen examined ureters of fetuses over 20 weeks’ gestation and children up to the age of 12 years and established mean ureteral dimensions for fetuses at 30 weeks’ gestation, and for infants and children at 3 months and 3, 6, and 12 years.5 The upper limit of the range of diameter of the mid-section of the ureter from birth to 12 years was 0.5–0.65 cm; therefore, the upper limit of normal was regarded as closely approximating 0.7 cm. Hellstrom et al provided radiological data of normal ureteric diameter in 194 children aged 0 to16 years, and also came to the conclusion that a ureteric diameter over 7 mm was abnormal in this age group.6
Embryology and Genetics of the Ureterovesical Junction
UVJ anomalies form part of the congenital anomalies of the kidney and urinary tract (CAKUT). CAKUT includes a spectrum of anomalies affecting urinary tract development, including renal agenesis, dysplasia or hypoplasia; proximal or distal ureteric obstruction (ureteropelvic junction (UPJ) or UVJ obstruction); dilatation of the ureter (megaureter); vesicoureteric reflux (VUR) and urethral obstruction (e.g., due to posterior urethral valves or urethral atresia).7 Embryologically, the mammalian kidney derives from two components: the ureteric bud, an epithelium that branches recurrently to form the collecting ducts, and the metanephric mesenchyme. The ureteric bud also gives rise to the epithelium of the renal pelvis and ureter, which is known as the urothelium.
To date, our knowledge of normal ureteral development is limited to work on animal models including transgenic mice with abnormal renal/ ureteral phenotypes. These studies suggest that several genes, transcription factors, as well as secreted proteins, contribute to normal ureteral development, that when deranged, manifest various forms of CAKUT.8 A key player in ureteric development is the glial cell line-derived neurotrophic factor and the transmembrane c-ret protein tyrosine kinase (GDNF/c-ret) axis.9 Ureteric bud sprouting and branching is dependent on the interaction between GDNF and its co-receptor glycosyl-phosphaditylinositol-anchored co-receptor 1 (Gfrα1), which bind to and activate c-ret. GDNF is primarily expressed in the metanephric mesenchyme, the trigonal wedge of the distal ureter and the urogenital sinus. In c-ret -/-mutants, renal agenesis, severe hypodysplasia and blind-ending ureters were observed; GDNF -/- and Gfrα1 -/- mutants were also found to have similar phenotypes, suggesting that the GDNF-c-ret axis is required for ureteric bud initiation and early branching morphogenesis.
C-ret expression is retinoic acid (a metabolite of Vitamin A) dependent.9 Batourina et al showed that Vitamin A and the Ret proto-oncogene are required for formation of a proper connection between the bladder and ureter.10 Mouse mutants lacking Ret or Rara and Rarb, two retinoid receptor family members, had hydronephrosis and megaureter, both of which were linked to defects at early stages of ureter maturation, when “common nephritic duct” (CND—the name given by Batourina et al to the part of the mesonephric duct proximal to the origin of the ureteric bud) remodeling induces ureteric orifices to separate from the mesonephric ducts. Their studies suggested that Vitamin A and Ret were required for the expansion of the CND into a structure called the wedge, which they proposed was the likely precursor of the trigone.11,12
The timing of smooth muscle differentiation in the distal ureter is unknown, but may be key to understanding why VUR or obstruction improve postnatally. Pirker et al studied bladder specimens form porcine fetuses, newborn and 6-month old piglets.13 They found that at 60 days, bladder detrusor muscle was well-developed and innervated, whereas the intra- and extra-vesical ureter had only just started to differentiate. At day 90, the extravesical ureter was well-innervated, but the intravesical ureter did not mature until after birth. All smooth muscle components showed a striking increase in bulk between neonatal and later sections. The time-course of muscular development in this study correlates with findings in humans, in whom smooth muscle bundles of the extramural ureter first appear in weeks 17 to 22 of gestation, and ureteral muscle development proceeds in a craniocaudal direction.14 The authors also noticed that periureteral sheath development precedes ureteral smooth muscle development, which was also reported in humans. The periureteral sheath showed considerable postnatal neuromuscular development. Its muscle fibers grew significantly, and almost formed a continuous layer at 6 months.13 Tanagho also observed that the distal ureter is the last portion to develop its muscular coat, and that early muscular differentiation is primarily of the circular muscles . Other studies have suggested possible explanations for the development of UVJ obstruction.15 These include a thick sleeve of muscle around the distal portion of the ureter, increased deposition of collagen fibres and dysregulated neuromodulation in the juxtavesical segment of obstructed megaureters.11,16,17
Prenatal Diagnosis
Ureteral dilation is associated with antenatal hydronephrosis in 5–10% of cases.18 Currently, nearly 79% of patients with hydroureteronephrosis present on perinatal ultrasound scan (USS), with the remainder presenting after a febrile urinary tract infection (UTI).19 The detection of a dilated ureter in-utero by ultrasound is a difficult endeavor. The fetus may not be in the proper orientation, or the ureter may be obscured by overlying structures or maternal bowel gas. Furthermore, technicians may not be trained to specifically look for dilated ureters on routine screening. As a result, the prenatal detection of dilated ureters is likely to be unreliable. In a study of 101 patients with postnatally-identified hydroureteronephrosis referred to Great Ormond Street Hospital, only 20% had a dilated ureter documented prenatally. Forty-eight (47.5%) infants were found to have VUR or other pathology, and 53 (52.5%) were suspected of having a UVJ obstruction.4
Postnatal management
Patients with ureteral dilation over 7mm are at increased risk for urinary tract infection (UTI) and thus continuous antibiotic prophylaxis (CAP) is recommended.20 A recent prospective multi-center study of 237 patients with ureteral dilation concluded that patients with ureters 7 mm or greater had nearly three times the risk of UTI adjusting for sex, circumcision status, antibiotic prophylaxis and hydronephrosis grade. The higher risk was also present in patients investigated with a voiding cystourethrogram (VCUG) and found not to have VUR. CAP was significantly protective against UTI.21
A postnatal ultrasound (US) scan is recommended between 48 hours and 4 weeks (UTD consensus), where the dilatation is unilateral (Figure 2).22 Bilateral hydroureteronephrosis should be investigated more urgently, and bladder outlet obstruction excluded prior to postnatal discharge. Presence of a dilated ureter(s) is an indication for a VCUG, in order to exclude urethral obstruction or VUR. The presence of VUR confirms a “refluxing megaureter”. It is important to ensure upper tract drainage following bladder emptying on the MCUG. Poor drainage on VCUG, especially where there is significant upper tract dilatation on USS, may raise the suspicion of a “refluxing and obstructive” megaureter.20
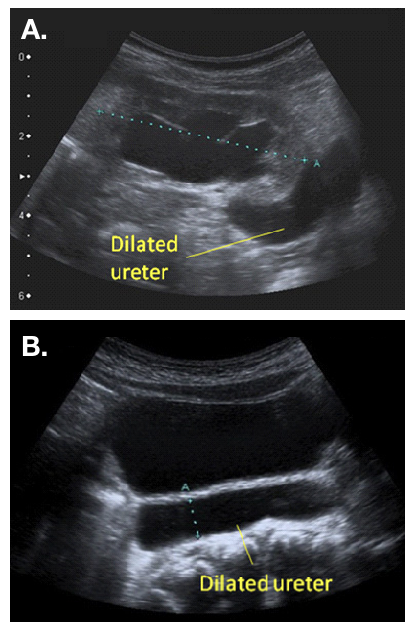
Figure 2 Ultrasound appearance of a dilated ureter, seen exiting a dilated renal pelvis (a) and visible behind the bladder (b)
In the absence of VUR or urethral obstruction, a dynamic renogram, such as 99mTc-MAG-3 diuretic renography, and a repeat ultrasound scan should be performed after 6 weeks of age. These investigations will assist with differentiating between an “obstructive”, or “non-obstructive-non-refluxing”, megaureter. Babies presenting postnatally (e.g., with a UTI) are also investigated in a similar fashion, although it is best to wait for recovery from urosepsis before embarking on a VCUG or MAG-3 renogram.20
According to the European Association of Nuclear Medicine (EANM) guidelines for standard and diuretic renography in children, renography allows estimation of two aspects of renal function.23 The first aspect is renal clearance, or differential renal function (DRF). DRF estimation is best undertaken between one and two minutes after tracer injection. The second aspect is the excretion, or disappearance, of the tracer from the kidney. Traditionally, an important delay in excretion is suggested by a continuously ascending curve and delayed transit, i.e. a Tmax (time to reach the maximum, or peak, of the curve) over 20 minutes (Figure 3). However, delayed transit may be caused by the increased capacity afforded by a dilated pelvis and ureter. Hence, furosemide is administered in order to distinguish between obstruction of the effect of a dilated system. Poor drainage may also be apparent because the bladder is full, or because the effect of gravity on drainage is incomplete. For all these reasons, the interpretation of “impaired drainage” on diuretic renography is controversial.24
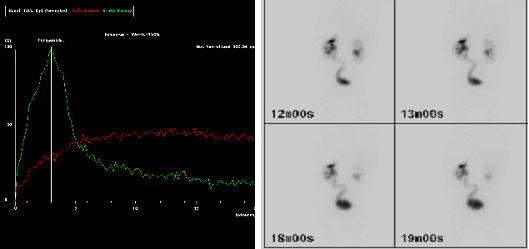
Figure 3 MAG-3 images showing delayed drainage of tracer (red curve) at 20 minutes (a) and visible left (L) ureter on delayed imaging (b), suggesting a hold-up at the left UVJ.
A combination of clinical and radiological findings must be taken into account when designating a megaureter as “obstructed” or “non-obstructed”. The presence of symptoms, such as febrile UTI, or pain, are clinical indicators of possible obstruction. In the asymptomatic patient, the presence of a DRF below 40%, and/or increasing dilatation on serial ultrasound scans, are also suggestive of obstruction and indications for intervention. Delayed transit on MAG-3 in the presence of stable or improving dilatation, and a DRF above 40%, in an asymptomatic patient, may be managed conservatively.20 Liu et al and McLellan et al suggested that children with a retrovesical ureteral diameter greater than 10 mm were more likely to require surgical intervention.4,25
It is important to be aware that ipsilateral UPJ and UVJ obstruction may co-exist. The pre-operative MAG-3 may suggest dual pathology when the obstructed ureter fills slowly then fails to drain. In this scenario, a retrograde pyelogram can be helpful in making a diagnosis.26
Management
Management of primary megaureters has changed over the past 30 years. In 1989, Peters et al reported that 89% of patients required surgery before 8 months of age.27 That same year, Keating et al reported that when the decision to intervene was based on absolute renal function, up to 87% of patients could be followed up conservatively.28 A further publication from the same unit confirmed stability or improvement at long-term follow-up of the same group of patients who managed conservatively.29 Liu et al also suggested that the majority of megaureters may be followed up conservatively, and showed that at a mean follow-up of 3.1 years, 34% of primary megaureters resolved spontaneously. Of the remaining cases, 17% were re-implanted due to break-through infection or deteriorating renal function, whereas 49% had persistent dilatation at follow-up, and were still being monitored conservatively.4
Non-Obstructed Non-Refluxing Megaureters
Megaureters associated with a DRF >40% in an asymptomatic patient, may be managed conservatively. Close follow-up with interval USS and antibiotic prophylaxis are recommended, especially where the ureteral dilation is over 7mm21 Song et al showed that the risk of UTI is higher with UVJ than UPJ obstruction, and that UTIs tend to occur within the first six months of life.30 Once the hydronephrosis is noted to be improving, follow-up appointments may be reduced to six-monthly and then annually, and antibiotic prophylaxis discontinued. Resolution of the hydroureteronephrosis may take several years. McLellan et al showed that hydronephrosis had resolved completely in 72% of cases at a median follow-up of 25.8 months.25
Long-term follow-up is advisable where dilatation is persistent. A long-term follow-up study by Shukla et al reported late deterioration in a 14-year old boy with bilateral primary megaureters, who presented with increased dilatation and functional deterioration six years after he was discharged from conservative follow-up.31 Hemal et al. 30 reported the outcome of 55 patients with congenital megaureters presenting for the first time in the third decade of life, of whom 20 patients were found to have renal calculi. Five patients with bilateral megaureters were in chronic renal failure at diagnosis, of whom two eventually died of the disease.32 Hence early resolution following conservative management is not a life-long guarantee, and follow-up into adulthood is advisable.
Obstructed Megaureters
Obstruction is diagnosed based on a reduced DRF <40% at diagnosis, or a drop in DRF >10% on repeated interval renography. Increasing dilatation on USS, and symptoms (UTI/pain) are also suggestive of obstruction. The choice of surgical intervention depends on the age of the child (below or over 1 year), and availability of equipment and expertise where an endoscopic option is considered. Surgical options include:
- Ureteral re-implantation (open/ laparoscopic or robotic-assisted)
- Temporary JJ-stenting
- Endoscopic balloon dilatation ± endoureterotomy
- Refluxing ureterocystotomy
- Cutaneous ureterostomy
Ureteral Reimplantation
The aim of ureteral re-implantation is to excise the aperistaltic, narrow UVJ segment, and tunnel the ureter into the bladder in an anti-reflux fashion. The tunnel should be at least five times the ureteral diameter to be effective, data based on a study by Paquin in 1959.33 In order to obtain this ratio when the ureteral diameter is above 10 mm, the distal 10 cm of the ureter may need to be tapered. A psoas hitch may also be required in order to ensure a straight entry of the ureter into the bladder. The two most popular techniques used for tailoring megaureters are plication as reported by Starr and Kalicinski et al., and tapering as modified by Hendren.34,35,36 Kalicinski et al described a folding technique whereby the lateral, avascular part of the ureter was excluded from the lumen by a longitudinal running suture, and then folded posteriorly prior to re-implantation. Starr modified this procedure, introducing many interrupted Lembert sutures that folded the ureteral wall inwards, thus preserving its blood supply, but making the subsequent re-implantation more cumbersome. Hendren advised excisional tapering, whereby a longitudinal strip of the redundant ureter is excised, and the ureter sutured in two layers.35,36 Ben-Meir et al compared the outcome of megaureter reimplantation with and without tailoring, and showed that there was no significant difference in outcome (94% success rate in the tailored group, and 96% in the non-tailored group).37 An example of reimplantation with tapering is provided in (Figure 4 and Figure 5).
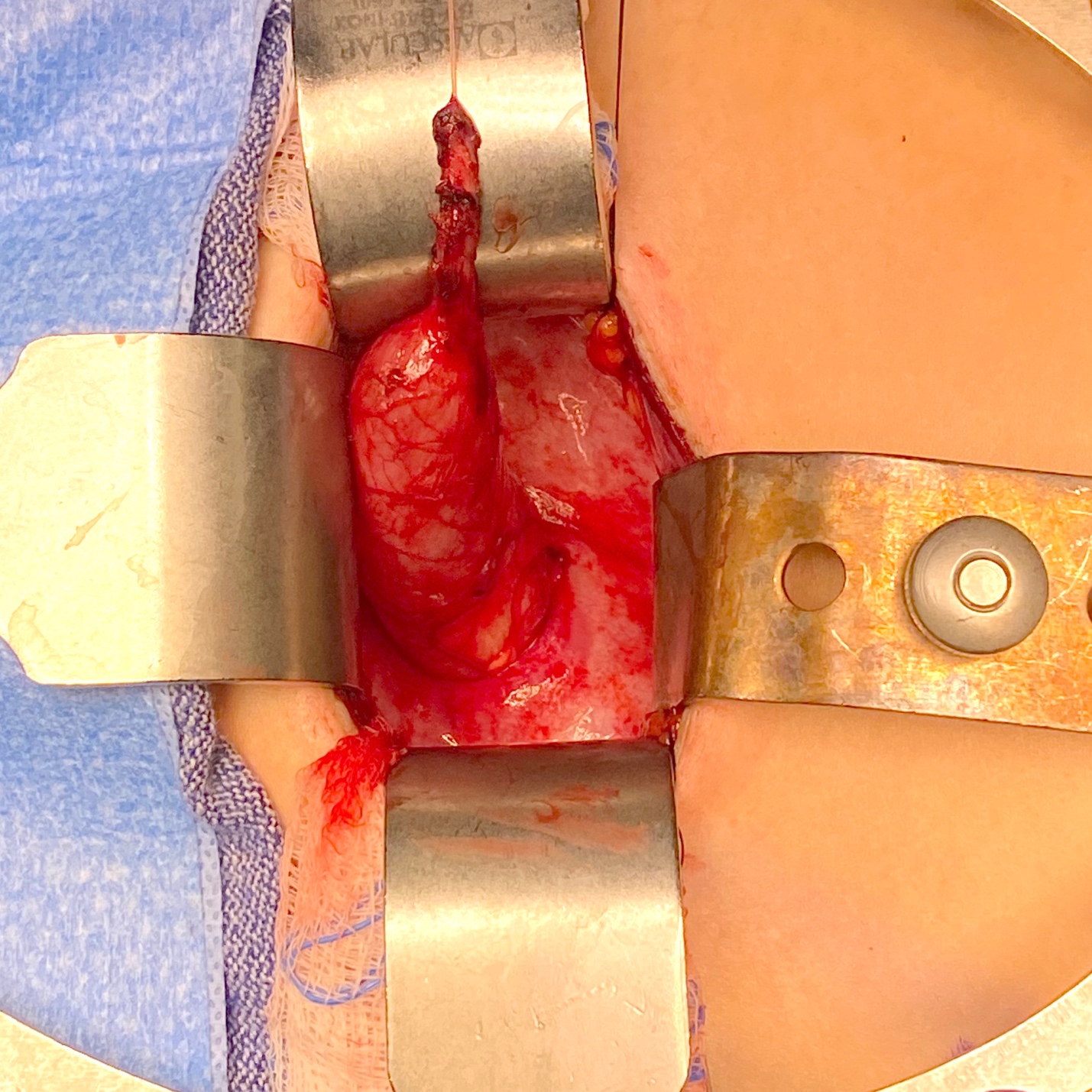
Figure 4 Intraoperative view of a megaureter with aperistaltic segment and proximal dilation.
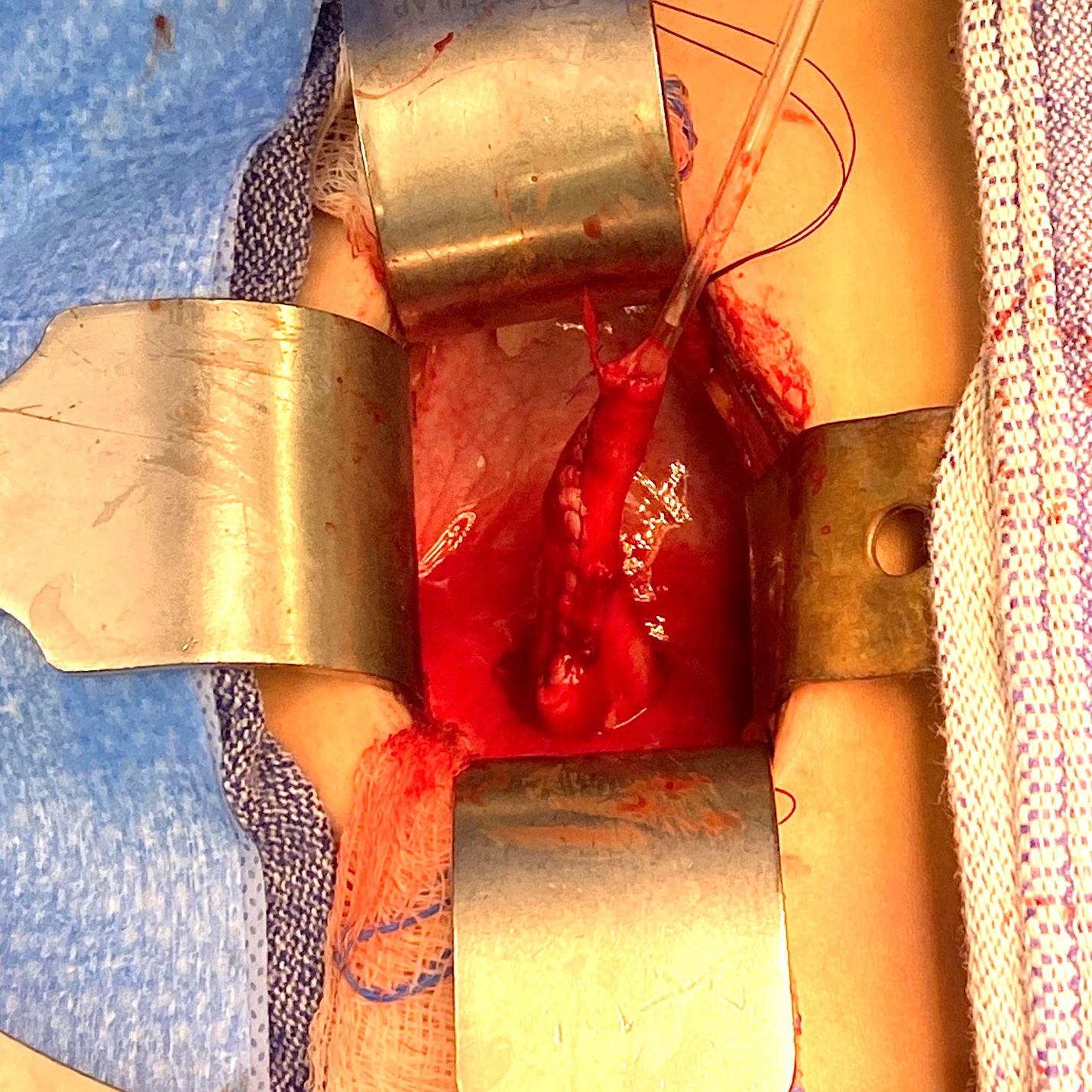
Figure 5 Intraoperative view of same ureter from Figure 4 after tapering over a 10 Fr catheter. It was subsequently reimplanted by creating a cross-trigonal tunnel, passing it through the tunnel and securing it. A ureteral stent was left in place for 2 weeks.
An extravesical Lich–Gregoir reimplantation is commonly preferred when laparoscopic or robotic-assisted (RALUR) techniques are used.38,39,40 The dilated ureter is identified below the iliac vessels, and the peritoneum dissected along the ureter to the level of the crossing of the vas deferens. The ureter is mobilized with blunt dissection to minimize the risk of nervous and blood supply injuries, and is then transected at the level of stenosis. The sero-muscular tunnel is marked out using hook electrocautery and then the detrusor fibers are sharply divided with scissors until the bladder mucosa bulges freely. Vesical mucosa is opened at the end of the tunnel and the ureterovesical anastomosis is performed with interrupted sutures. The ureter is then installed into the tunnel, which is approximated by continuous or interrupted sutures.
VUR resolution rates after extravesical RALUR reported in the literature range from 66.7 to 100% in multiple relatively small series.40 Overall success upon pooling these series is 91%. A multi-institutional retrospective study reported radiographic resolution in 87.9% of 280 ureters. More recently, a large prospective multi-institutional study reported 93.8% resolution in 199 ureters.40 RALUR may be performed bilaterally; however, there is concern that bilateral dissection of the posterior bladder may disrupt the pelvic nerve plexus, resulting in higher rates of postoperative urinary retention. Nerve-sparing dissection has been proposed to reduce this complication. In 2008, Casale et al reported a 97.6% success rate following bilateral nerve-sparing RALUR in 41 patients.41 There were no complications or instances of urinary retention. Herz et al reported a 91.7% success rate for unilateral RALUR but a success rate of only 77.8% of ureters (72.2% of children) for bilateral cases. In this study, complication rates (including ureteral obstruction, readmission, and urinary retention) were higher for bilateral cases. A nerve-sparing technique was not utilized.42
Ureteric reimplantation in infants below one year of age may be challenging due to the discrepancy between the grossly-dilated ureter and the small infantile bladder, and concern regarding possible iatrogenic bladder dysfunction. De Jong et al investigated the bladder functional outcome of 49 neonates and infants with primary obstructive or refluxing megaureters, who underwent a Politano-Leadbetter procedure, at a mean follow-up of 7.3 years.43 No onset of voiding dysfunction was noted at follow-up, and bladder capacity for age was significantly increased. No post-operative ureteral obstruction was observed. The author concluded that early major reconstructions of the lower urinary tract do not damage the urodynamic properties of the bladder and pelvic floor, provided that the surgery is performed by specialized pediatric urological surgeons. A further study from the same group 41 compared urodynamic outcome in patients who underwent ureteral reimplantation before, or after, one year of age. No significant difference in bladder capacity, compliance, or incidence of instability, was found.44
Temporary JJ Stenting
In 1999, Shenoy and Rance reported of the use of JJ stents as a temporizing measure in infants with primary obstructive megaureters.45 The procedure involves the insertion of a double pigtail stent, endoscopically where possible. A tight UVJ obstruction may preclude endoscopic insertion, in which case an open insertion via a cystostomy combined with dilatation of the ureteric orifice using a lachrymal probe may be performed. Stents are licensed to remain in-situ for six months, following which, they should be removed or replaced. The megaureter should be stented until the baby is over one year of age, by which time a ureteric reimplantation may be safely performed, if the dilatation has not resolved.45 The baby should remain on prophylactic antibiotics while the stent is in-situ, due to the risk of stent infection, and should be monitored by ultrasonography to ensure decompression of the system, and to check stent position.46 An ultrasound scan and MAG-3 renography performed three months after removal of stent will direct further management. In the presence of improved dilatation and drainage, and preserved function, no further intervention may be required. Persistent or worsening dilatation after a period of stenting, or further infection, is an indication for ureteric reimplantation. A long-term outcome study revealed that in 56% of cases, no further intervention was required after removal of the stent. However, two-thirds of patients required open stent insertion, and one-third suffered complications (migration, stone formation and infection).47
Endoscopic Balloon Dilatation
Endoscopic management has become accepted as a less invasive option for treatment. Advantages of the endoscopic approach include a minimally-invasive approach, no manipulation of the distal ureteral blood supply, and no need for prolonged catheterization.48 Furthermore, if endoscopic management should fail, ureteral reimplantation can still be performed. However, the potential need for a second anesthetic for stent removal must be factored into discussions regarding the risks of the endoscopic approach. The procedure is performed using a pediatric rigid cystoscope (8F–10.5F) and a flexible guidewire is placed up into the renal pelvis. For high-pressure balloon dilatation (HPBD), a balloon catheter (usually 3F–5F, 2–4 cm balloon length, 3–7 mm inflated balloon diameter) is used to dilate the ureterovesical junction, followed by placement of a Double-J ureteral stent. For incisional ureterotomy, both Kajbafzadeh et al and Shirazi and et al perform incisions at the 6 o’clock position.49,50,51
Doudt et al published a systematic review of the long-term outcome of endoscopic management.48 Eleven retrospective and 1 prospective, single institution case series were analyzed. Mean age at time of surgery was 24.6 months (range 3–84). Initial attempts at endoscopic management were not effective in 10.5%, either because of an inability to pass the stent endoscopically, or difficulty with advancement of the pediatric cystoscope through the urethra. Effectively completed endoscopic approaches were cystoscopy+high-pressure balloon dilation (HPBD)+ Double-J ureteral stent placement in 49.5%, cystoscopy+incisional ureterotomy+Double-J ureteral stent placement in 27.8%, cystoscopy+Double-J ureteral stent placement in 18.9% and cystoscopy+HPBD+ incisional ureterotomy+Double-J ureteral stent placement in 3.7%. Mean stent duration was 2.3 months (range 0.25–6). At a mean follow-up of 3.2 years, initial success rate was 76.7%. Endoscopic re-treatment was performed in 15.1% of cases with a 36.7% overall surgical re-intervention rate. The authors concluded that endoscopic management for persistent or progressive POM in children >12 months of age is a minimally invasive alternative to ureteral reimplantation with modest success rates. In infants, it may best be utilized as a temporizing procedure. Approximately one-third of patients require surgical re-intervention.48
Refluxing Re-Implantation
Kaefer proposed a novel approach for managing obstructing megaureters in infants, performed in two stages.52 The first stage remedies the obstruction without the concern of creating an anti-reflux mechanism, by simply dividing the ureter proximal to the obstruction and performing an end-to-side refluxing anastomosis to the bladder; then, as the bladder size increases during infancy, the second stage is performed, a standard tapered ureteral reimplantation with creation of an anti-reflux valve mechanism.52,53 Alyami et al published their experience with the refluxing reimplantation approach in 32 patients with distal ureteral obstruction diagnosed antenatally.54 Mean age at surgery was 3.7 months (range 0 to 33) and mean follow-up was 34.3 months (range 6 to 58). A circumcision was also performed in most of the males. Of 32 patients, 86% demonstrated significant improvement in dilation. Six patients underwent further procedures, including a circumcision and 2 ureteral reimplantations for recurrent infections. The authors concluded that despite the trade-off of relieving obstruction by creating reflux, this procedure can be considered a potentially definitive intervention in patients who remain infection-free. Close follow-up is critical to document satisfactory long-term results.54
Cutaneous Ureterostomy
A temporary cutaneous ureterostomy remains a safe and effective option, especially in the presence of urosepsis, or where endoscopic technology is not available. Shrestha et al published the outcome of 25 patients who underwent an end cutaneous ureterostomy (ECU) POM at a mean age of 7 months (range 23 days to 2.5 years). Following diversion, renal failure had resolved in all, and febrile urinary tract infection (UTI) developed in one while awaiting reimplantation.55 No stomal complications were reported in this study, although others have reported complications, including stomal stenosis and pyelonephritis.56
Conclusion
UVJ anomalies, associated with a megaureter, may be obstructive, refluxing, non-obstructed and non-refluxing, or both refluxing and obstructed. Only 10-20% of megaureters require intervention, whilst the remainder may be monitored conservatively. Surgical options include ureteric reimplantation, with or without ureteral tailoring, endoscopic procedures, refluxing ureterneocystostomy or a cutaneous ureterostomy. Persistent dilatation in non-obstructive non-refluxing megaureters is warranted due to the long-term risk of stone-formation.
Key Points
- Beware of the rare refluxing and obstructive megaureter: take note of upper tract drainage after bladder emptying when high-grade VUR is diagnosed on VCUG. In the presence of poor drainage associated with significant upper tract dilatation, a diuretic MAG-3 renogram is indicated. Ureteral reimplantation is the best option in this situation.
- UVJ and UPJ obstructions can co-exist. Look for discrepancies in renal pelvis and ureteral dilatation, especially where one appears to improve while the other does not. Inspect MAG-3 renogram imaging for drainage at both junctions. Where suspicion is raised, a retrograde pyelogram may assist with the diagnosis. A surgical strategy could be a pyeloplasty with double-J stent insertion in the first instance; followed by further imaging after stent removal.
- Where conservative management of the non-obstructive non-refluxing megaureter is undertaken, the current literature recommends antibiotic prophylaxis for ureteral dilation over 7mm; the greatest risk of UTI is in the first year of life.
- Long-term follow-up is advisable in cases where ureteral dilatation does not resolve in childhood, due to the long-term risk of stone-formation or renal functional deterioration in bilateral cases.
References
- Ferenci P, Lockwood A, Mullen K, Tarter R, Weissenborn K, Blei AT. Hepatic encephalopathy-Definition, nomenclature, diagnosis, and quantification: Final report of the Working Party at the 11th World Congresses of Gastroenterology, Vienna, 1998. Hepatology 1977; 35 (3): 716–721. DOI: 10.1053/jhep.2002.31250.
- King LR. Megaloureter: Definition, Diagnosis and Management. J Urol 1980; 123 (2): 222–223. DOI: 10.1016/s0022-5347(17)55867-x.
- Pfister RC, Hendren WH. Primary megaureter in children and adults Clinical and pathophysiologic features of 150 ureters. Urology 1978; 12 (2): 160–176. DOI: 10.1016/0090-4295(78)90327-8.
- Liu HYA, Dhillon HK, Yeung CK, Diamond DA, Duffy PG, Ransley PG. Clinical Outcome and Management of Prenatally Diagnosed Primary Megaureters. J Urol 1994; 152 (2 Part 2): 614–617. DOI: 10.1016/s0022-5347(17)32664-2.
- Cussen LJ. Normal Position of the Ureteral Orifice in Infancy and Childhood: A Quantitative Study. J Urol 1967; 121 (5): 646–647. DOI: 10.1016/s0022-5347(17)56920-7.
- Hellström M, Hjälmås K, Jacobsson B, Jodal U, Odén A. Normal Ureteral Diameter in Infancy and Childhood. Acta Radiol Diagn (Stockh) 1985; 26 (4): 433–439. DOI: 10.1177/028418518502600412.
- Stahl DA, Koul HK, Chacko JK, Mingin GC. Congenital anomalies of the kidney and urinary tract (CAKUT): A current review of cell signaling processes in ureteral development. J Pediatr Urol 2006; 2 (1): 2–9. DOI: 10.1016/j.jpurol.2005.04.006.
- Woolf AS. A molecular and genetic view of human renal and urinary tract malformations. Kidney Int 2000; 58 (2): 500–512. DOI: 10.1046/j.1523-1755.2000.00196.x.
- Takahashi M. The GDNF/RET signaling pathway and human diseases. Cytokine Growth Factor Rev 2001; 12 (4): 361–373. DOI: 10.1016/s1359-6101(01)00012-0.
- Batourina E, Choi C, Paragas N, Bello N, Hensle T, Costantini FD. Faculty Opinions recommendation of Distal ureter morphogenesis depends on epithelial cell remodeling mediated by vitamin A and Ret. Faculty Opinions – Post-Publication Peer Review of the Biomedical Literature 2002; 32. DOI: 10.3410/f.1009006.124259.
- Viana R, Batourina E, Huang H, Dressler GR, Kobayashi A, Behringer RR, et al.. The development of the bladder trigone, the center of the anti-reflux mechanism. Development 2007; 134 (20): 3763–3769. DOI: 10.1242/dev.011270.
- Batourina E, Tsai S, Lambert S, Sprenkle P, Viana R, Dutta S. Faculty Opinions recommendation of Apoptosis induced by vitamin A signaling is crucial for connecting the ureters to the bladder. Faculty Opinions – Post-Publication Peer Review of the Biomedical Literature 2005; 37. DOI: 10.3410/f.1029077.343528.
- Pirker ME, Rolle U, Shinkai T, Shinkai M, Puri P. Prenatal and Postnatal Neuromuscular Development of the Ureterovesical Junction. J Urol 2007; 177 (4): 1546–1551. DOI: 10.1016/j.juro.2006.11.081.
- Matsuno T, Tokunaka S, Koyanagi T. Muscular Development in the Urinary Tract. J Urol 1984; 132 (1): 148–152. DOI: 10.1016/s0022-5347(17)49508-5.
- Tanagho EA, Meyers FH, Smith DR. The Trigone: Anatomical and Physiological Considerations. 1. In Relation to the Ureterovesical Junction. J Urol 1968; 100 (5): 623–632. DOI: 10.1016/s0022-5347(17)62584-9.
- Tanagho EA. Intrauterine Fetal Ureteral Obstruction. J Urol 1973; 109 (2): 196–203. DOI: 10.1016/s0022-5347(17)60388-4.
- Lee BR, Silver RI, Partin AW, Epstein JI, Gearhart JP. A Quantitative Histologic Analysis of Collagen Subtypes: The Primary Obstructed and Refluxing Megaureter of Childhood. Urology 1998; 51 (5): 820–823. DOI: 10.1016/s0090-4295(98)00013-2.
- Nguyen HT, Herndon CDA, Cooper C, Gatti J, Kirsch A, Kokorowski P. The Society for Fetal Urology consensus statement on the evaluation and management of antenatal hydronephrosis. Yearbook of Urology 2010; 2010: 209. DOI: 10.1016/s0084-4071(10)79537-3.
- Rubenwolf P, Herrmann-Nuber J, Schreckenberger M, Stein R, Beetz R. Primary non-refluxive megaureter in children: single-center experience and follow-up of 212 patients. Int Urol Nephrol 2016; 48 (11): 1743–1749. DOI: 10.1007/s11255-016-1384-y.
- Farrugia M-K, Hitchcock R, Radford A, Burki T, Robb A, Murphy F. British Association of Paediatric Urologists consensus statement on the management of the primary obstructive megaureter. J Pediatr Urol 2014; 10 (1): 26–33. DOI: 10.1016/j.jpurol.2013.09.018.
- Holzman SA, Braga LH, Zee RS, Herndon CDA, Davis-Dao CA, Kern NG. Correspondence to the article ‘risk of urinary tract infection in patients with hydroureter: an analysis from the society of fetal urology prenatal hydronephrosis registry.’ J Pediatr Urol 2021; 18 (5): 722–723. DOI: 10.1016/j.jpurol.2022.07.031.
- Nguyen HT, Benson CB, Bromley B, Campbell JB, Chow J, Coleman B. Faculty Opinions recommendation of Multidisciplinary consensus on the classification of prenatal and postnatal urinary tract dilation (UTD classification system). Faculty Opinions – Post-Publication Peer Review of the Biomedical Literature 2014; 10: 982–998. DOI: 10.3410/f.725257762.793506733.
- Gordon I, Piepsz A, Sixt R. Guidelines for standard and diuretic renogram in children. Eur J Nucl Med Mol Imaging 2011; 38 (6): 1175–1188. DOI: 10.1007/s00259-011-1811-3.
- Gordon I. Diuretic renography in infants with prenatal unilateral hydronephrosis: an explanation for the controversy about poor drainage. BJU Int 2001; 87 (6): 551–555. DOI: 10.1046/j.1464-410x.2001.00081.x.
- McLELLAN DAWNL, RETIK ALANB, BAUER STUARTB, DIAMOND DAVIDA, ATALA ANTHONY, MANDELL JAMES, et al.. Rate and Predictors of Spontaneous Resolution of Prenatally Diagnosed Primary Nonrefluxing Megaureter. J Urol 2002: 2177–2180. DOI: 10.1097/00005392-200211000-00091.
- CAY ALI, IMAMOGLU MUSTAFA, BAHAT ELIF, SARIHAN HALUK. Diagnostic difficulties in children with coexisting pelvi-ureteric and vesico-ureteric junction obstruction. BJU Int 2006; 98 (1): 177–182. DOI: 10.1111/j.1464-410x.2006.06187.x.
- Peters CA, Mandell J, Lebowitz RL, Colodny AH, Bauer SB, Hendren WH. Congenital obstructed megaureters in early infancy: Diagnosis and treatment. J Pediatr Surg 1989; 25 (5): 576. DOI: 10.1016/0022-3468(90)90639-q.
- Keating MA, Escala J, Snyder HMC, Heyman S, Duckett JW. Changing Concepts in Management of Primary Obstructive Megaureter. J Urol 1989; 142 (2 Part 2): 636–640. DOI: 10.1016/s0022-5347(17)38841-9.
- Baskin LS, Zderic SA, Snyder HM, Duckett JW. Primary Dilated Megaureter: Long-Term Followup. J Urol 1994; 152 (2 Part 2): 618–621. DOI: 10.1016/s0022-5347(17)32665-4.
- Song S-H, Lee S-B, Park YS, Kim KS. Is Antibiotic Prophylaxis Necessary in Infants With Obstructive Hydronephrosis? J Urol 2007; 177 (3): 1098–1101. DOI: 10.1016/j.juro.2006.11.002.
- SHUKLA ASEEMR, COOPER JEFFREY, PATEL RAKESHP, CARR MICHAELC, CANNING DOUGLASA, ZDERIC STEPHENA, et al.. Prenatally Detected Primary Megaureter: A Role For Extended Followup. J Urol 2005; 173 (4): 1353–1356. DOI: 10.1097/01.ju.0000152319.72909.52.
- Hemal AK, Ansari MS, Doddamani D, Gupta NP. Symptomatic and complicated adult and adolescent primary obstructive megaureter–indications for surgery: analysis, outcome, and follow-up. Urology 2003; 61 (4): 703–707. DOI: 10.1016/s0090-4295(02)02590-6.
- Paquin AJ. Ureterovesical Anastomosis: the Description and Evaluation of a Technique. J Urol 1959; 82 (5): 573–583. DOI: 10.1016/s0022-5347(17)65934-2.
- Villanueva CA. Extracorporeal ureteral tailoring during HIDES laparoscopic robotic-assisted ureteral reimplantation for megaureter. J Pediatr Urol 1979; 11 (6): 362–363. DOI: 10.1016/j.jpurol.2015.08.006.
- Kaliciński ZH, Kansy J, Kotarbińska B, Joszt WLELA. Surgery of megaureters–Modification of Hendren’s operation. J Pediatr Surg 1977; 12 (2): 183–188. DOI: 10.1016/s0022-3468(77)80005-5.
- Hendren WH. Operative Repair of Megaureter in Children. J Urol 1969; 101 (4): 491–507. DOI: 10.1016/s0022-5347(17)62370-x.
- Ben-Meir D, McMullin N, Kimber C, Gibikote S, Kongola K, Hutson JM. Reimplantation of obstructive megaureters with and without tailoring. J Pediatr Urol 2006; 2 (3): 178–181. DOI: 10.1016/j.jpurol.2005.05.010.
- Stanasel I, Atala A, Hemal A. Robotic Assisted Ureteral Reimplantation: Current Status. Curr Urol Rep 2013; 14 (1): 32–36. DOI: 10.1007/s11934-012-0298-1.
- LEE S, AKBAL C, KAEFER M, Chertin B, Pollack A, Koulikov D. Laparoscopic Ureteral Reimplantation With Extracorporeal Tailoring for Megaureter: A Simple Technical Nuance. J Urol 2006; 176 (6): 2640–2642. DOI: 10.1016/j.juro.2006.08.025.
- Bilgutay AN, Kirsch AJ. Robotic Ureteral Reconstruction in the Pediatric Population. Front Pediatr 2019; 7. DOI: 10.3389/fped.2019.00085.
- Casale P, Patel RP, Kolon TF. Nerve Sparing Robotic Extravesical Ureteral Reimplantation. J Urol 2008; 179 (5): 1987–1990. DOI: 10.1016/j.juro.2008.01.062.
- Herz D, Fuchs M, Todd A, McLeod D, Smith J. Robot-assisted laparoscopic extravesical ureteral reimplant: A critical look at surgical outcomes. J Pediatr Urol 2016; 12 (6): 402.e1–402.e9. DOI: 10.1016/j.jpurol.2016.05.042.
- Nelson CP. Obstructing congenital anomalies of the urinary tract: ureteropelvic junction obstruction, ureterocele, megaureter, and posterior urethral valves. The Brady Urology Manual 1997; 65: 207–220. DOI: 10.1201/9780429194993-21.
- KORT LAETITIAMO de, KLIJN AARTJ, UITERWAAL CUNOSPM, JONG TOMPVM de. Ureteral Reimplantation In Infants And Children: J Urol 2002; 167: 285–287. DOI: 10.1097/00005392-200201000-00084.
- Shenoy, Rance. Is there a place for the insertion of a JJ stent as a temporizing procedure for symptomatic partial congenital vesico-ureteric junction obstruction in infancy? BJU Int 1999; 84 (4): 524–525. DOI: 10.1046/j.1464-410x.1999.00256.x.
- Farrugia M-K, Steinbrecher HA, Malone PS. The utilization of stents in the management of primary obstructive megaureters requiring intervention before 1 year of age. J Pediatr Urol 2011; 7 (2): 198–202. DOI: 10.1016/j.jpurol.2010.04.015.
- Farrugia MK, Steinbrecher HA, Malone PS. Re: The Utilization of Stents in the Management of Primary Obstructive Megaureters Requiring Intervention Before 1 Year of Age. J Urol 2011; 186 (5): 2063–2063. DOI: 10.1016/j.juro.2011.07.142.
- Doudt AD, Pusateri CR, Christman MS. Endoscopic Management of Primary Obstructive Megaureter: A Systematic Review. J Endourol 2018; 32 (6): 482–487. DOI: 10.1089/end.2017.0434.
- Kajbafzadeh A-M, Payabvash S, Salmasi AH, Arshadi H, Hashemi S-M, Arabian S, et al.. Endoureterotomy for Treatment of Primary Obstructive Megaureter in Children. J Endourol 2007; 21 (7): 743–749. DOI: 10.1089/end.2006.0330.
- Tourchi A, Kajbafzadeh AM, Mazaheri T, Raad M. Endoscopic Management of Obstructive Refluxing Megaureter: a Novel Approach. J Pediatr Urol 2010; 6: S62. DOI: 10.1016/j.jpurol.2010.02.110.
- Shirazi M, Natami M, Hekmati P, Farsiani M. Result of Endoureterotomy in the Management of Primary Obstructive Megaureter in the First Year of Life: Preliminary Report. J Endourol 2014; 28 (1): 79–83. DOI: 10.1089/end.2013.0098.
- LEE SANGDON, AKBAL CEM, KAEFER MARTIN. Refluxing Ureteral Reimplant As Temporary Treatment Of Obstructive Megaureter In Neonate And Infant. J Urol 2005; 173 (4): 1357–1360. DOI: 10.1097/01.ju.0000152317.72166.df.
- Kaefer M, Misseri R, Frank E, Rhee A, Lee D. Re: Refluxing Ureteral Reimplantation: A Logical Method for Managing Neonatal UVJ Obstruction. J Urol 2014; 194 (2): 518–518. DOI: 10.1016/j.juro.2015.05.043.
- Alyami FA, Koyle MA, Bowlin PR, Gleason JM, Braga LH, Lorenzo AJ. Side-to-Side Refluxing Nondismembered Ureterocystotomy: A Novel Strategy to Address Obstructed Megaureters in Children. J Urol 2017; 198 (5): 1159–1167. DOI: 10.1016/j.juro.2017.05.078.
- Shrestha AL, Bal HS, Kisku SMC, Sen S. Commentary to “Outcome of end cutaneous ureterostomy (ECU) as a non conservative option in the management of primary obstructive megaureters (POM).” J Pediatr Urol 2018; 15 (3): 294–295. DOI: 10.1016/j.jpurol.2019.02.024.
- Kitchens DM, DeFoor W, Minevich E, Reddy P, Polsky E, McGregor A, et al.. End Cutaneous Ureterostomy for the Management of Severe Hydronephrosis. J Urol 2007; 177 (4): 1501–1504. DOI: 10.1016/j.juro.2006.11.076.
Last updated: 2025-09-25 12:10